Share This Page
Drug Price Trends for SM EYE DROPS
✉ Email this page to a colleague
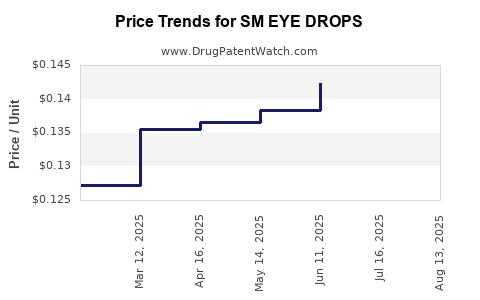
Average Pharmacy Cost for SM EYE DROPS
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SM EYE DROPS | 49348-0037-29 | 0.13387 | ML | 2025-08-20 |
| SM EYE DROPS | 49348-0037-29 | 0.13934 | ML | 2025-07-23 |
| SM EYE DROPS | 49348-0037-29 | 0.14227 | ML | 2025-06-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
SM EYE DROPS Market Analysis and Financial Projection
Market Overview and Demand for SM Eye Drops
SM Eye Drops are indicated for the treatment of dry eye syndrome, a condition impacting approximately 15-20% of the adult population globally, with higher prevalence among those aged over 60. The rising incidence correlates with aging populations and increased screen time. The global dry eye market size was valued at approximately USD 4.1 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of around 7.6% through 2030 [1].
Market growth drivers include:
- Aging demographics, particularly in North America and Europe.
- Increasing awareness for eye health maintenance.
- Rising adoption of advanced ocular therapeutics.
- Development and approval of novel formulations with improved efficacy and reduced side effects.
Key players such as Allergan, Bausch + Lomb, and Novartis dominate the market, with new entrants focused on innovative dry eye treatments, including preservative-free formulations and drug delivery advances.
Competitive Landscape
Major products in the dry eye therapeutics market include:
- Cyclosporine-based eye drops (e.g., Restasis)
- Lifitegrast (e.g., Xiidra)
- Lipid-based or lubricating eye drops
SM Eye Drops could position as a new entrant or a branded generic if their formulation offers distinct advantages such as faster relief or longer duration.
Regulatory pathways involve FDA approval in the U.S., EMA approval in Europe, and equivalent agencies elsewhere, necessitating clinical trials demonstrating safety and efficacy.
Price Projections for SM Eye Drops
Pricing depends on formulation, branding, and market positioning. Current market prices for leading dry eye medications are:
| Product | Price Range USD (per bottle) | Bottle Size |
|---|---|---|
| Restasis | USD 130 - 180 | 30 x 0.05 mL |
| Xiidra | USD 160 - 220 | 30 x 0.09 mL |
| Over-the-counter artificial tears | USD 5 - 15 | Varies |
If SM Eye Drops can differentiate via improved efficacy or reduced dosing frequency, premium pricing is feasible, potentially USD 150–200 per bottle.
Price decline is possible with generics entering the market or if the formulation becomes available over the counter. Discounting strategies and insurance reimbursements influence net pricing.
Revenue and Market Penetration Scenarios
Assuming the following:
- Launch year: 2024
- Target market: US, Europe initially
- Approximate market share over time: 2-5% in 5 years
- Average selling price (ASP): USD 180 per bottle
- First-year sales: 1 million bottles
Projected revenue in the first year:
| Scenario | Market Share | Units Sold | Revenue (USD millions) |
|---|---|---|---|
| Conservative | 2% | 20,000 | 3.6 |
| Moderate | 3% | 30,000 | 5.4 |
| Aggressive | 5% | 50,000 | 9.0 |
As competition intensifies, prices may decline by 10-20%, influencing long-term revenues.
Pricing and Market Entry Risks
- Launch delays due to regulatory hurdles
- Patent challenges or exclusivity loss
- Entry of generics at lower prices
- Reimbursement policies affecting patient out-of-pocket costs
Strategic Recommendations
- Focus on clinical differentiation and labeling claims.
- Target early adoption through ophthalmology specialty channels.
- Prepare for potential price erosion with scalable manufacturing.
- Monitor regulatory developments and patent landscape.
Key Takeaways
- The dry eye market is expected to reach USD 6.4 billion by 2030, growing at 7.6% CAGR.
- SM Eye Drops can command premium prices if performance advantages are validated.
- Initial revenue projections suggest USD 3.6–9 million in year one, scalable with market penetration.
- Competition from established brands and generics will pressure pricing.
- Strategic positioning and regulatory compliance are critical to market success.
FAQs
1. What factors influence the pricing of dry eye eye drops?
Pricing depends on formulation efficacy, delivery method, branding, regulatory status, and reimbursement policies.
2. How does the market size for dry eye treatments compare across regions?
North America accounts for about 45% of the global market, Europe 30%, with Asia-Pacific rapidly expanding due to aging populations.
3. What regulatory challenges could affect SM Eye Drops?
Obtaining FDA or EMA approval requires demonstrating safety and efficacy; patent challenges may delay market entry.
4. What is the typical market share for new dry eye treatments within five years?
New entrants generally target 2-5% market share, depending on differentiation and sales execution.
5. How can SM Eye Drops differentiate in a competitive market?
Through superior efficacy, convenience, reduced side effects, or novel delivery mechanisms.
Sources
[1] MarketsandMarkets, “Dry Eye Disease Market,” 2022.
More… ↓
