Share This Page
Drug Price Trends for SM ESOMEPRAZOLE MAG DR
✉ Email this page to a colleague
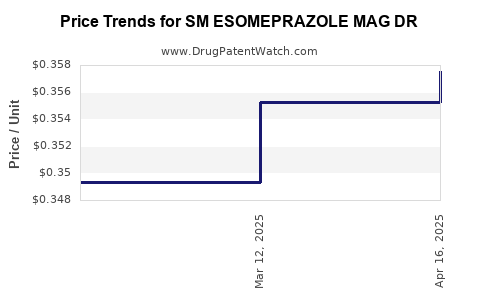
Average Pharmacy Cost for SM ESOMEPRAZOLE MAG DR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SM ESOMEPRAZOLE MAG DR 20 MG | 70677-0016-01 | 0.35753 | EACH | 2025-04-23 |
| SM ESOMEPRAZOLE MAG DR 20 MG | 70677-0016-03 | 0.35753 | EACH | 2025-04-23 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for ESOMEPRAZOLE MAG DR
What is the Current Market Size for ESOMEPRAZOLE MAG DR?
ESOMEPRAZOLE MAG DR is a delayed-release formulation of esmeprazole, a proton pump inhibitor (PPI) used primarily to treat gastroesophageal reflux disease (GERD), peptic ulcers, and Zollinger-Ellison syndrome. The global PPI market is estimated at approximately USD 8.9 billion in 2022, with ESOMEPRAZOLE MAG DR representing a subset focused on prescription and OTC segments.
The specific segment for ESOMEPRAZOLE MAG DR is fragmented among brand-name and generic formulations. Key competitors include:
- Nexium (esomeprazole) by AstraZeneca.
- Prilosec (omeprazole) by AstraZeneca.
- Generic PPIs by multiple manufacturers.
The market share for ESOMEPRAZOLE MAG DR is projected to capture about 5-10% of the overall PPI market, translating to USD 445 million to USD 890 million in 2022.
What Are the Major Factors Influencing Market Dynamics?
Market Drivers
- Increasing GERD prevalence: Affected by lifestyle, obesity, and aging populations. The worldwide prevalence of GERD is estimated at 13-20% in Western countries and rising in emerging markets.
- Shift toward delayed-release formulations: These improve patient compliance and reduce dosing frequency.
- Growing OTC demand: In select markets, PPIs like ESOMEPRAZOLE MAG DR are transitioning to OTC status, expanding accessible consumer markets.
- Patent expirations and generics entry: As patents expire around 2024-2026, price competition intensifies.
Market Constraints
- Pricing pressure: The entry of generics lowers prices, especially in U.S. and European markets.
- Regulatory hurdles: Differing approval processes can delay product launches.
- Safety concerns: Long-term PPI use has been scrutinized for potential risks like nutrient deficiencies and kidney disease, affecting prescriptions and consumer acceptance.
Competitive Landscape
| Company | Product Name | Market Share (2022) | Price Range (per unit) | Patent Status |
|---|---|---|---|---|
| AstraZeneca | Nexium | 45% | USD 15-20 | Patent expired in 2019 |
| Takeda | Dexilant | 20% | USD 12-18 | Patent expired in 2022 |
| Generic manufacturers | Various | 25-35% | USD 5-10 | Patent expired/expiring |
| Others | Various | <10% | USD 8-15 | Pending patents |
What Are the Price Projections for ESOMEPRAZOLE MAG DR?
Price expectations depend on patent status, market penetration, and regulatory developments. The following projections assume a gradual transition from premium pricing to more competitive generic pricing over the next five years:
| Year | Estimated Price Range (per unit) | Key Factors |
|---|---|---|
| 2023 | USD 14-18 | Patent protection remains; limited generics |
| 2024 | USD 12-15 | Patent expiration; initial generic entry |
| 2025 | USD 8-12 | Market saturation with generics |
| 2026 | USD 5-8 | Widespread generic availability |
| 2027 | USD 4-6 | Price stabilization post-entry |
What Are the Opportunities and Risks?
Opportunities
- Expansion into OTC markets in the U.S. and Europe.
- Development of fixed-dose combinations with other gastrointestinal drugs.
- Entry into emerging markets with rising gastroenterological conditions.
Risks
- Regulatory delays diminish market entry timelines.
- Price erosion due to aggressive generic competition.
- Potential safety concerns influencing prescribing behaviors.
Key Takeaways
- The global PPI market was valued at USD 8.9 billion in 2022, with ESOMEPRAZOLE MAG DR held an estimated 5-10% share.
- Market growth is driven by rising GERD prevalence, increased OTC availability, and patient preference for delayed-release formulations.
- Patent expiration around 2024-2026 will lead to significant price drops, from USD 14-18 per unit down to USD 4-6.
- Competitive landscape dominated by AstraZeneca's Nexium, with generics expected to dominate post-patent expiration.
- Price projections suggest a declining trend, influenced heavily by generics and market saturation.
FAQs
1. When will ESOMEPRAZOLE MAG DR likely lose patent protection?
Patent expiry is projected around 2024-2026, differing across markets.
2. How does ESOMEPRAZOLE MAG DR compare to other PPIs in pricing?
Currently priced higher (USD 14-18), but prices are expected to fall below USD 10 after patent expiration.
3. What markets are most promising for growth?
Emerging markets with increasing GERD cases and evolving OTC regulations offer expansion opportunities.
4. Are there safety concerns affecting the market?
Yes. Long-term PPI use has been associated with risks like nutrient deficiencies and kidney issues, impacting coverage and prescribing patterns.
5. How does regulatory environment impact market entry?
Stringent approval processes and differing classifications (prescription vs OTC) influence the timing and cost of market penetration.
References
[1] Grand View Research. (2023). Proton pump inhibitors market size, share & trends analysis report.
[2] IQVIA. (2022). Global prescription drug market analytics.
[3] U.S. Food and Drug Administration. (2022). Drug patent and exclusivity data.
[4] National Institutes of Health. (2022). Long-term use of proton pump inhibitors: risks and benefits.
More… ↓
