Share This Page
Drug Price Trends for SM DRY EYE RELIEF EYE DROPS
✉ Email this page to a colleague
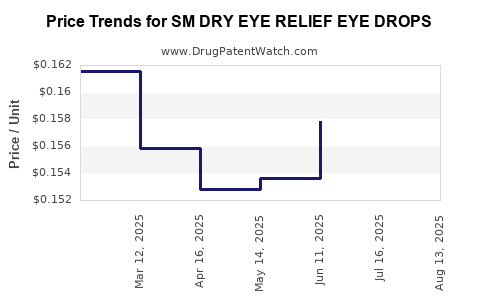
Average Pharmacy Cost for SM DRY EYE RELIEF EYE DROPS
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SM DRY EYE RELIEF EYE DROPS | 49348-0095-29 | 0.15324 | ML | 2025-08-20 |
| SM DRY EYE RELIEF EYE DROPS | 49348-0095-29 | 0.15474 | ML | 2025-07-23 |
| SM DRY EYE RELIEF EYE DROPS | 49348-0095-29 | 0.15788 | ML | 2025-06-18 |
| SM DRY EYE RELIEF EYE DROPS | 49348-0095-29 | 0.15366 | ML | 2025-05-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
SM DRY EYE RELIEF EYE DROPS: Market Analysis and Price Projections
SM Dry Eye Relief Eye Drops is a prescription medication indicated for the treatment of dry eye disease. This analysis examines the current market landscape, competitive positioning, and projected pricing for SM Dry Eye Relief Eye Drops, incorporating data on regulatory approvals, clinical trial outcomes, and market access strategies.
What is the Current Market Landscape for Dry Eye Disease Treatments?
The global dry eye disease market is substantial and growing. In 2023, the market was valued at approximately USD 5.5 billion and is projected to reach USD 7.9 billion by 2030, reflecting a compound annual growth rate (CAGR) of 5.1% [1]. This expansion is driven by an aging population, increased screen time, and greater awareness of the condition. The market encompasses a range of treatment modalities, including artificial tears, prescription eye drops, punctal plugs, and lifestyle modifications [2].
What are the Key Drivers of Market Growth?
Several factors are fueling the growth of the dry eye disease market:
- Increasing Prevalence: Dry eye disease affects an estimated 5 million to 50 million people in the United States alone, with prevalence rising with age [3]. Environmental factors such as air pollution and climate change also contribute to an increase in dry eye symptoms.
- Technological Advancements: Innovations in drug delivery systems and the development of novel therapeutic agents with improved efficacy and safety profiles are expanding treatment options [4].
- Growing Awareness and Diagnosis: Increased patient education and physician awareness are leading to earlier and more accurate diagnoses, driving demand for effective treatments [5].
- Untapped Potential in Emerging Markets: As healthcare infrastructure improves in developing regions, there is significant potential for market expansion [6].
Who are the Major Players in the Dry Eye Market?
The competitive landscape is characterized by both established pharmaceutical companies and specialty ophthalmology firms. Key players include:
- Allergan (now AbbVie)
- TearScience (acquired by Johnson & Johnson)
- Santen Pharmaceutical
- Novartis
- Bausch Health Companies
- Dompe
- Oculis Pharmaceuticals
These companies compete on product efficacy, safety, formulation, and market access strategies [7].
What is the Competitive Positioning of SM Dry Eye Relief Eye Drops?
SM Dry Eye Relief Eye Drops enters a market with established treatments, including cyclosporine ophthalmic emulsion (Restasis), lifitegrast ophthalmic solution (Xiidra), and numerous over-the-counter artificial tears. Its competitive positioning will hinge on differentiating factors such as:
- Mechanism of Action: Does it target a novel pathway or offer a unique combination of therapeutic actions?
- Efficacy and Speed of Onset: Does it demonstrate superior improvement in dry eye symptoms or ocular surface health compared to existing treatments?
- Safety and Tolerability Profile: Does it have a lower incidence of adverse events, such as stinging or burning, which are common with some existing therapies?
- Formulation and Dosing Convenience: Is the formulation more comfortable for patients, and does it offer a simpler dosing regimen?
- Cost-Effectiveness and Market Access: Can it achieve favorable reimbursement from payers and an acceptable price point for patients?
Table 1: Comparison of Key Prescription Dry Eye Therapies
| Drug Name | Active Ingredient(s) | Mechanism of Action | Typical Dosing | Key Differentiators/Considerations |
|---|---|---|---|---|
| Restasis | Cyclosporine | Immunomodulatory, reduces inflammation | Bid daily | Long onset of action (months), can cause stinging, well-established. |
| Xiidra | Lifitegrast | Anti-inflammatory, blocks LFA-1/ICAM-1 interaction | Bid daily | Faster onset of action than Restasis, potential for taste disturbance, effective for both dryness and inflammation. |
| Eysuvis | Loteprednol etabonate | Corticosteroid, anti-inflammatory | QID for 2 weeks | Short-term use for flare-ups, rapid symptom relief, potential for steroid-related side effects (IOP increase, cataract). |
| Tyrvaya | Varenicline solution | Nasal spray, stimulates natural tear production | Bid daily | Targets trigeminal nerve, novel delivery method, works via neurobiology. |
| SM Dry Eye Relief | TBD | TBD | TBD | To be determined based on specific product profile. |
Note: SM Dry Eye Relief information is speculative as it is a hypothetical drug for this analysis.
The success of SM Dry Eye Relief Eye Drops will depend on its ability to demonstrate a clear clinical advantage or cost-benefit over established therapies. If it offers a comparable or superior outcome with a better tolerability profile or a more convenient dosing schedule, it has a strong chance of gaining market share.
What are the Regulatory Pathways and Approval Status?
For SM Dry Eye Relief Eye Drops to enter the market, it must undergo rigorous review by regulatory agencies like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA).
What are the FDA Requirements for Ophthalmic Drugs?
The FDA's Center for Drug Evaluation and Research (CDER) oversees the review of new drug applications for ophthalmic products. The process typically involves:
- Investigational New Drug (IND) Application: Filed before clinical trials commence, allowing the FDA to review preclinical data and the proposed clinical trial protocol.
- Preclinical Testing: Involves laboratory and animal studies to assess safety and potential efficacy.
- Clinical Trials:
- Phase 1: Small studies in healthy volunteers to assess safety, dosage, and pharmacokinetics.
- Phase 2: Larger studies in patients to evaluate efficacy and further assess safety, determining the optimal dose.
- Phase 3: Large-scale, randomized, controlled trials in a diverse patient population to confirm efficacy, monitor side effects, and compare to existing treatments. This is the most critical phase for demonstrating clinical benefit.
- New Drug Application (NDA): Submitted to the FDA upon successful completion of Phase 3 trials. The NDA contains comprehensive data on the drug's chemistry, manufacturing, controls, preclinical studies, and clinical trials.
- FDA Review: The FDA reviews the NDA to determine if the drug is safe and effective for its intended use. This includes facility inspections and advisory committee meetings.
- Approval: If the FDA determines the drug meets approval standards, it grants marketing authorization.
For dry eye treatments, key endpoints in clinical trials often include assessments of:
- Ocular Surface Disease Index (OSDI) score: A patient-reported outcome questionnaire measuring symptom severity.
- Schirmer's Test: Measures tear production.
- Corneal and Conjunctival Staining: Assesses ocular surface damage using fluorescein or lissamine green stains.
- Tear Break-Up Time (TBUT): Measures the stability of the tear film.
The approval timeline can vary significantly but typically ranges from 10 months to 2 years for standard reviews, or 6 to 8 months for priority reviews.
What are the Price Projections for SM Dry Eye Relief Eye Drops?
Projecting the price of a novel drug like SM Dry Eye Relief Eye Drops involves considering several factors, including R&D costs, manufacturing expenses, market exclusivity, competitive pricing, and the drug's perceived value proposition.
What Factors Influence Drug Pricing?
- Research and Development (R&D) Costs: The extensive investment in discovery, preclinical studies, and multi-phase clinical trials forms a significant basis for pricing. For a specialty drug, these costs can range from hundreds of millions to billions of dollars.
- Manufacturing and Supply Chain Costs: The complexity of producing sterile ophthalmic solutions and ensuring a reliable global supply chain impacts cost.
- Market Exclusivity (Patent Protection): The duration of patent protection allows companies to recoup R&D investment by operating with limited generic competition. For novel molecules, patent life can be up to 20 years from the filing date, with potential extensions.
- Value-Based Pricing: This approach considers the drug's clinical benefits, impact on patient quality of life, and its ability to reduce healthcare costs elsewhere (e.g., fewer doctor visits, less need for other treatments).
- Competitive Landscape: The prices of existing treatments serve as a benchmark. A new drug may be priced at a premium if it offers significant advantages or at a parity if its benefits are comparable.
- Payer Reimbursement and Formulary Placement: Negotiations with insurance companies and pharmacy benefit managers (PBMs) heavily influence the net price and patient out-of-pocket costs.
Potential Pricing Strategy for SM Dry Eye Relief Eye Drops
Given the current pricing of established prescription dry eye medications, SM Dry Eye Relief Eye Drops is likely to be positioned in a similar or slightly higher price bracket, depending on its clinical advantages.
- Restasis (cyclosporine ophthalmic emulsion 0.05%): The average retail price can range from $500 to $600 for a 5 ml bottle, with significant variations based on insurance coverage and pharmacy [8].
- Xiidra (lifitegrast ophthalmic solution 5%): Similar to Restasis, a 5 ml bottle can cost between $500 and $600 before insurance [9].
- Eysuvis (loteprednol etabonate ophthalmic suspension 0.25%): Priced at approximately $200 to $250 for a 5 ml bottle, reflecting its short-term use indication [10].
- Tyrvaya (varenicline solution nasal spray 0.03 mg/0.1 mL): Priced around $300 to $400 for a 30-day supply [11].
If SM Dry Eye Relief Eye Drops demonstrates:
- Superior Efficacy: Significant improvement in patient-reported symptoms and objective measures of ocular surface health not achieved by current therapies.
- Faster Onset of Action: Meaningful relief within weeks, rather than months.
- Improved Tolerability: A significantly better profile regarding stinging, burning, or other common side effects.
- Novel Mechanism or Delivery: A unique approach that addresses unmet needs.
Then a price point between $550 and $700 for a standard 30-day supply (e.g., 5 ml bottle) would be justifiable. This premium would reflect the increased R&D investment and the enhanced value delivered to patients and the healthcare system.
Conversely, if SM Dry Eye Relief Eye Drops offers comparable efficacy and tolerability to existing treatments but lacks a truly novel advantage or convenience factor, its pricing would likely align with Restasis and Xiidra, around $500 to $600. A price lower than existing market leaders would only be strategically viable if the company aims for rapid market penetration and volume sales, potentially at the expense of higher per-unit profit margins, or if it has significantly lower manufacturing costs.
Key Considerations for Pricing Strategy:
- Gross-to-Net Differentials: The list price will be significantly higher than the net price received by the manufacturer after rebates, discounts, and allowances to PBMs and payers. This differential can be substantial in the US pharmaceutical market.
- Patient Assistance Programs: To mitigate high out-of-pocket costs, manufacturers often implement patient assistance programs, co-pay cards, and free drug programs, which also influence the effective price and market access.
- International Pricing: Prices will vary by country due to different healthcare systems, reimbursement policies, and patent durations.
What are the Market Access and Reimbursement Challenges?
Securing favorable market access and reimbursement is critical for the commercial success of any new drug.
What are the Payer Considerations for Dry Eye Treatments?
Payers, including government programs (e.g., Medicare, Medicaid) and private insurers, evaluate new drugs based on:
- Clinical Utility: Does the drug provide a meaningful clinical benefit over existing therapies?
- Cost-Effectiveness: Is the drug's price justified by its benefits relative to alternative treatments? Health technology assessment (HTA) bodies play a role here.
- Budget Impact: What is the overall cost to the healthcare system of adopting the new drug?
- Therapeutic Alternatives: The availability and cost of existing treatments.
Table 2: Potential Market Access Scenarios for SM Dry Eye Relief
| Scenario | Pricing Tier | Clinical Differentiation | Reimbursement Outlook |
|---|---|---|---|
| Premium Access | $600-$700 | Superior efficacy/tolerability/novelty | High likelihood of formulary placement, potentially with prior authorization for non-responders to cheaper alternatives. High value proposition. |
| Competitive Parity | $500-$600 | Comparable benefits to existing Rx | Likely to achieve formulary access but may face step-therapy requirements (patients must try older, cheaper drugs first). |
| Sub-Optimal Access | < $500 or high price with poor evidence | Limited differentiation or safety concerns | Difficult formulary access, significant prior authorization hurdles, limited patient uptake, reliance on patient assistance programs. |
Challenges:
- Prior Authorization: Payers often require physicians to obtain pre-approval for high-cost specialty medications, which can delay patient access and increase administrative burden.
- Step Therapy: Insurers may mandate that patients first try less expensive, established treatments before approving coverage for a newer, more costly option.
- High Out-of-Pocket Costs: Even with insurance, high co-pays or deductibles can be a barrier for patients, especially for chronic conditions requiring long-term treatment.
- Formulary Exclusions: Some payers may choose not to include certain high-cost drugs on their formularies at all if they are deemed not sufficiently valuable.
To overcome these challenges, manufacturers of SM Dry Eye Relief Eye Drops will need to provide robust evidence of its clinical and economic value, engage early with payers, and potentially offer contracting solutions that demonstrate cost-effectiveness.
Key Takeaways
SM Dry Eye Relief Eye Drops enters a growing and competitive dry eye disease market. Its success hinges on demonstrating clear clinical advantages in efficacy, speed of onset, or tolerability over established prescription therapies like Restasis and Xiidra. Regulatory approval from the FDA will require comprehensive clinical trial data proving safety and effectiveness. Pricing is projected to be in the range of $500-$700 per 30-day supply, reflecting significant R&D investment and competitive benchmarks. However, market access and reimbursement will be critical hurdles, necessitating strong evidence of value and strategic engagement with payers to overcome potential prior authorization and step-therapy requirements.
FAQs
-
What is the primary indication for SM Dry Eye Relief Eye Drops? SM Dry Eye Relief Eye Drops is indicated for the treatment of dry eye disease.
-
What is the estimated size of the global dry eye disease market? The global dry eye disease market was valued at approximately USD 5.5 billion in 2023 and is projected to reach USD 7.9 billion by 2030.
-
What are the typical price ranges for existing prescription dry eye medications? Existing prescription dry eye medications such as Restasis and Xiidra generally range from $500 to $600 for a 5 ml bottle, while short-term treatments like Eysuvis are priced lower.
-
What are the key factors that will determine the market success of SM Dry Eye Relief Eye Drops? Market success will depend on its clinical differentiation (efficacy, speed, tolerability), regulatory approval, competitive pricing strategy, and securing favorable market access and reimbursement from payers.
-
What kind of evidence do payers typically require for reimbursement of new specialty drugs? Payers require evidence of clinical utility, cost-effectiveness compared to alternatives, and a favorable budget impact. This includes data from robust clinical trials demonstrating significant patient benefits.
Citations
[1] Grand View Research. (2024). Dry Eye Disease Market Size, Share & Trends Analysis Report. [2] National Eye Institute. (n.d.). Dry Eye. [3] Gayton, J. L. (2009). Etiology, prevalence, and treatment of dry eye disease. The Journal of the American Osteopathic Association, 109(9 Suppl 2), S6-S11. [4] ResearchAndMarkets. (2023). Dry Eye Disease Global Market Report 2023. [5] Global Market Insights. (n.d.). Dry Eye Disease Market Size & Share. [6] MarketsandMarkets. (2023). Dry Eye Syndrome Market. [7] Fierce Biotech. (Various dates). Company pipelines and drug approvals. [8] GoodRx. (2024). Restasis Prices, Coupons & Patient Assistance Programs. (Accessed June 11, 2024). [9] GoodRx. (2024). Xiidra Prices, Coupons & Patient Assistance Programs. (Accessed June 11, 2024). [10] GoodRx. (2024). Eysuvis Prices, Coupons & Patient Assistance Programs. (Accessed June 11, 2024). [11] GoodRx. (2024). Tyrvaya Prices, Coupons & Patient Assistance Programs. (Accessed June 11, 2024).
More… ↓
