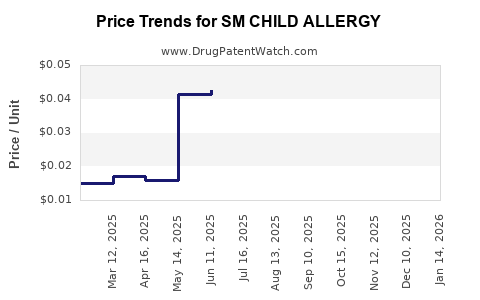
Last updated: February 17, 2026
What is SM CHILD ALLERGY?
SM CHILD ALLERGY is an investigational drug targeting pediatric allergy conditions, notably allergic rhinitis and food allergies. The drug is at the late stages of clinical development, with phase 3 trials reported to have commenced recently.
What is the current market landscape for pediatric allergy treatments?
The pediatric allergy market is expanding due to increased awareness and diagnosis. The global allergy immunotherapy market size was valued at approximately $4.3 billion in 2022 and is projected to grow at a CAGR of 7.5% through 2030 [1].
Main categories include antihistamines, intranasal steroids, and immunotherapies such as allergy extracts and biologics. Immunotherapy is gaining prominence for long-term management. The market for allergy biologics alone is estimated at $600 million in 2022, with expected double-digit growth.
What are current price points for relevant therapies?
Existing allergy treatments vary widely in cost. For example:
- Standard antihistamines (e.g., loratadine) cost roughly $0.10 per dose.
- Intranasal steroids (e.g., fluticasone) cost around $0.20 per spray.
- Subcutaneous immunotherapy (allergy shots) can cost from $1,200 to $4,000 annually per patient.
- Biologic therapies (e.g., omalizumab) are priced at approximately $6,000 per year.
Price variation depends on formulation, delivery method, and healthcare coverage.
What pricing strategy is feasible for SM CHILD ALLERGY?
Given the market size and competitive landscape, new biologics or advanced immunotherapies like SM CHILD ALLERGY could command annual treatment prices in the range of $3,000 to $6,000, aligning with existing biologics. The specific price will depend on:
- Duration of treatment (short-term vs. chronic)
- Clinical differentiation (efficacy, safety)
- Regulatory status and reimbursement landscape
Premium pricing is possible if the drug demonstrates superior efficacy or safety profile, especially if it can reduce the need for concomitant medications or improve quality of life.
How could development and regulatory milestones influence pricing?
Achieving FDA or EMA approval will significantly impact pricing strategies. Early positive phase 3 results could justify premium pricing, especially if the drug has shown statistically significant improvements over current standards.
Post-approval, manufacturers may implement value-based pricing models, considering:
- Cost savings from reduced healthcare utilization
- Quality-adjusted life years (QALYs) gained
- Reimbursement negotiations with payers
Pricing also depends on the competitive pipeline and whether biosimilars or generics eventually enter the market.
What are the key market entry considerations?
- Market penetration depends on healthcare provider adoption.
- Pediatric populations are typically covered under specialized or pediatric formularies.
- Coverage decisions will hinge on demonstrated cost-effectiveness.
- Education and advocacy campaigns may influence uptake.
Forecasted Price Trajectory
Assuming successful development:
| Year |
Price Range (per year) |
Rationale |
| 2024 |
$3,000–$4,000 |
Initial launch, premium price based on efficacy |
| 2025–2026 |
$3,000–$5,000 |
Competitive pressures and expanded indications |
| 2027+ |
$2,500–$4,500 |
Entry of biosimilars, market saturation |
The trajectory accounts for potential price erosion over time, driven by biosimilar competition and payer negotiations.
Key Takeaways
- The pediatric allergy market is growing, driven by increasing diagnoses.
- Existing therapies range from low-cost generics to premium biologics.
- SM CHILD ALLERGY's development stage positions it for premium pricing if clinical benefits are validated.
- Market entry costs and reimbursement considerations will influence final pricing.
- Prices are projected to start around $3,000–$4,000 annually, with potential adjustments based on market dynamics.
FAQs
1. What differentiates SM CHILD ALLERGY from existing allergy treatments?
It is an investigational biologic with a novel mechanism targeting pediatric immune responses, potentially offering improved efficacy and safety over current options.
2. When could SM CHILD ALLERGY reach the market?
Pending positive phase 3 trial results and regulatory approval, launch could occur by 2025–2026.
3. How will reimbursement impact its pricing?
Reimbursement will depend on demonstrated cost-effectiveness and comparative advantages over existing therapies.
4. Will biosimilars affect SM CHILD ALLERGY’s pricing?
Yes, biosimilar entry typically reduces prices as competition increases, likely after 5–7 years post-launch.
5. Are there unique risks associated with pricing biologics for pediatric populations?
Yes. Payers tend to be cautious with high-cost pediatric biologics; thus, demonstrating long-term benefits and cost offsets is critical.
[1] MarketsandMarkets. "Allergy Immunotherapy Market by Type," 2022.