Share This Page
Drug Price Trends for SM ANTACID-ANTIGAS LIQUID
✉ Email this page to a colleague
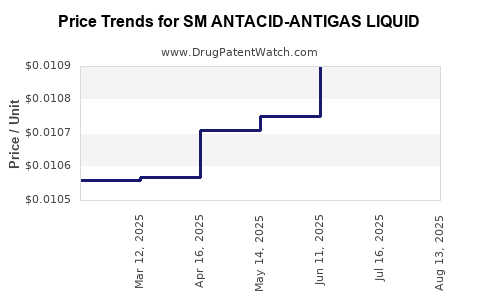
Average Pharmacy Cost for SM ANTACID-ANTIGAS LIQUID
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SM ANTACID-ANTIGAS LIQUID | 70677-0115-01 | 0.01062 | ML | 2025-08-20 |
| SM ANTACID-ANTIGAS LIQUID | 70677-0115-01 | 0.01074 | ML | 2025-07-23 |
| SM ANTACID-ANTIGAS LIQUID | 70677-0115-01 | 0.01090 | ML | 2025-06-18 |
| SM ANTACID-ANTIGAS LIQUID | 70677-0115-01 | 0.01075 | ML | 2025-05-21 |
| SM ANTACID-ANTIGAS LIQUID | 70677-0115-01 | 0.01071 | ML | 2025-04-23 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for SM ANTACID-ANTIGAS LIQUID
What is the Current Market Size and Demand for Antacid-Antigas Liquids?
The global antacid and indigestion treatment market was valued at approximately $4.2 billion in 2022. Growth is driven by increased cases of gastroesophageal reflux disease (GERD), indigestion, and related digestive disorders. The liquids segment accounts for around 30% of this market, with the remainder in tablets, capsules, and powders.
Key regional markets include North America, Asia-Pacific, and Europe. North America holds the largest share at around 35%, primarily due to high prevalence of GERD and OTC availability. Asia-Pacific's growth rate outpaces other regions at approximately 6% CAGR (2022-2027), driven by rising disposable income and urbanization.
Who Are the Major Competitors?
Leading brands include:
- Tums Liquid (GlaxoSmithKline)
- Maalox Liquid (Boehringer Ingelheim)
- Gaviscon Liquid (Reckitt Benckiser)
- Rolaids Liquid (Johnson & Johnson)
These companies hold significant shelf space in pharmacies and supermarkets, with established distribution channels.
What Are the Regulatory Considerations?
In the U.S., OTC antacid liquids are regulated as monographed drugs under the FDA OTC Drug Review. Approval processes involve demonstrating safety and efficacy, with labeling complying with monograph standards. Patent protection generally lasts 20 years from filing; however, many formulations are now off-patent.
Emerging markets may have different regulatory pathways, often requiring local clinical data and registration procedures. Regulatory timelines vary from 6 to 24 months depending on the jurisdiction.
What Are the Pricing Dynamics?
Market prices for antacid liquids typically range from $5 to $12 per 8 oz bottle retail. Bulk wholesale prices are approximately 15-25% lower. Price variability depends on brand positioning, formulation complexity, and packaging size.
Private label products are priced around $4 per bottle, competing mainly on cost. Name-brand products command premiums up to 20-30%, attributed to brand reputation and perceived efficacy.
Price Projection and Market Growth Insights
Forecasting indicates the global antacid liquid market will grow at a Compound Annual Growth Rate (CAGR) of 4.5% over the next five years (2023-2028). This expansion reflects increased diagnosis rates and consumer OTC use, particularly in emerging markets.
Innovations, such as formulations with added probiotics or flavor enhancements, could enable premium pricing. The introduction of combination therapies may further influence market dynamics.
| Year | Projected Market Size (USD billion) | Compound Growth Rate | Key Factors |
|---|---|---|---|
| 2023 | 4.4 | - | Market stabilization, regulatory compliance |
| 2024 | 4.6 | 4.5% | Increased OTC accessibility, patent expiry |
| 2025 | 4.8 | 4.3% | Product innovation, emerging market growth |
| 2026 | 5.0 | 4.2% | Consumer awareness, hospital prescriptions |
| 2027 | 5.3 | 4.5% | Expanded use cases, health trends |
Price projections expect a slight increase in retail pricing (~2-3%) driven by inflation and raw material costs, but competition limits aggressive price hikes.
Cost of Goods and Potential Profit Margins
Manufacturing costs for liquids typically range from $1 to $2 per 8 oz bottle, depending on raw materials and manufacturing scale. Gross profit margins are generally between 50-60% for established brands, with new entrants potentially achieving 40-55% margins during initial launch phases.
Key Trends Influencing the Market
- Consumer Preference Shift: Preference for liquid forms over tablets due to faster action and ease of swallowing.
- OTC Market Expansion: Increased OTC availability outside retail pharmacies through online channels.
- Formulation Innovations: Incorporation of natural ingredients, probiotics, and flavor masking to attract health-conscious consumers.
- Regulatory Changes: Stricter regulations on labeling and ingredient safety in certain markets.
Summary
Antacid-antigas liquids hold a mature segment with steady growth prospects. Market size is expected to reach $5.3 billion by 2027, driven by consumer demand and emerging market expansion. Pricing remains competitive, with minor increases predicted, supported by product innovation and brand differentiation.
Key Takeaways
- The global market for antacid liquids is projected to grow at 4.5% CAGR through 2028.
- Market size could reach approximately $5.3 billion within five years.
- Price points range from $5 to $12 per bottle, with modest increases expected.
- Major competitors dominate the OTC segment, with strong branding and distribution networks.
- Innovation and regulatory adaptations will influence pricing strategies and product positioning.
FAQs
1. How much can a new SM ANTACID-ANTIGAS LIQUID expect to sell annually?
In mature markets, annual sales per product can range from $10 million to $50 million, depending on brand recognition, distribution reach, and marketing. Entry products may capture 1-5% of the OTC antacid liquid market initially.
2. Are there patent opportunities for novel formulations?
Yes. New formulations incorporating unique active ingredients or delivery enhancements can secure patent protection, typically lasting 20 years from filing.
3. What are key barriers to market entry?
Regulatory approval, establishing trusted distribution channels, and competing against well-known brands pose significant barriers. Formulation safety data and effective branding are critical.
4. How does the competitive landscape affect pricing strategies?
Market dominance by established brands stabilizes prices, limiting new entrants' ability to underprice. Entry-level pricing primarily targets private label or generic offerings.
5. What are the regulatory hurdles in emerging markets?
Procedures often require local clinical trials, labeling adjustments, and registration processes which can extend approval timelines from 6 to 24 months.
References
- MarketWatch. (2023). Global antacid market sizing and forecasts.
- U.S. Food and Drug Administration. (2022). OTC Drug Review standards.
- Research and Markets. (2022). Asia-Pacific digestive aid market report.
- Grand View Research. (2022). Digestive health market analysis.
- European Medicines Agency. (2022). Regulatory guidelines for OTC drugs.
More… ↓
