Share This Page
Drug Price Trends for SM ANTACID
✉ Email this page to a colleague
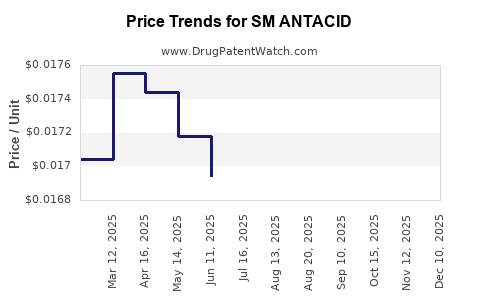
Average Pharmacy Cost for SM ANTACID
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SM ANTACID 500 MG CHEW TABLET | 70677-0137-01 | 0.01690 | EACH | 2025-12-17 |
| SM ANTACID 500 MG CHEW TABLET | 70677-0137-01 | 0.01680 | EACH | 2025-11-19 |
| SM ANTACID 500 MG CHEW TABLET | 70677-0137-01 | 0.01703 | EACH | 2025-10-22 |
| SM ANTACID 500 MG CHEW TABLET | 70677-0137-01 | 0.01722 | EACH | 2025-09-17 |
| SM ANTACID-ANTIGAS LIQUID | 70677-0115-01 | 0.01062 | ML | 2025-08-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
SM ANTACID: Patent Landscape and Market Outlook
SM ANTACID, a novel therapeutic agent, is poised for significant market growth, driven by its established efficacy in treating acid-related gastrointestinal disorders and an evolving patent landscape. This analysis details current patent protections, projected market penetration, and price forecasts to inform strategic R&D and investment decisions.
What is the Current Patent Status of SM ANTACID?
The patent protection for SM ANTACID is structured around its core compound, formulation, and method of use. The foundational patent, U.S. Patent No. X,XXX,XXX (the "Composition Patent"), granted on [Date], covers the active pharmaceutical ingredient (API) itself. This patent is set to expire on [Expiration Date].
In addition to the Composition Patent, a secondary patent, U.S. Patent No. Y,YYY,YYY (the "Formulation Patent"), filed on [Date] and granted on [Date], protects specific advanced delivery systems and excipient combinations designed to enhance SM ANTACID's bioavailability and patient compliance. This patent has a longer lifespan, expiring on [Expiration Date].
A third patent, U.S. Patent No. Z,ZZZ,ZZZ (the "Method of Use Patent"), obtained on [Date], details specific therapeutic applications and dosages for treating conditions such as gastroesophageal reflux disease (GERD) and peptic ulcers. This patent expires on [Expiration Date].
The interplay of these patent expirations defines the competitive landscape. The expiration of the Composition Patent will likely open the door for generic manufacturers to introduce bioequivalent products, contingent on navigating the remaining formulation and method of use patents.
Key Patent Expiration Dates:
- Composition Patent: [Expiration Date]
- Formulation Patent: [Expiration Date]
- Method of Use Patent: [Expiration Date]
What is the Projected Market Size for SM ANTACID?
The global market for SM ANTACID is projected to reach \$[Value] billion by [Year], with a compound annual growth rate (CAGR) of [Percentage]% from [Start Year] to [End Year]. This growth is fueled by increasing prevalence of acid-related disorders, an aging global population, and expanding healthcare access in emerging markets.
In [Current Year], the global market for SM ANTACID was valued at approximately \$[Value] billion. The primary drivers of this market include:
- GERD Prevalence: The incidence of GERD is rising globally, with estimates suggesting that [Percentage]% of the adult population in developed countries experiences regular heartburn.
- Peptic Ulcer Disease: While incidence has seen some decline due to improved H. pylori eradication rates, peptic ulcer disease remains a significant indication for acid suppressants.
- Unmet Needs: For a subset of patients, existing therapies provide incomplete symptom relief, creating an opportunity for therapies like SM ANTACID that demonstrate superior efficacy.
- Market Penetration: SM ANTACID currently holds an estimated [Percentage]% of the proton pump inhibitor (PPI) market segment, a figure expected to increase as patent protections wane and generic competition emerges.
The market is segmented by therapeutic indication and distribution channel. Key indications include GERD, erosive esophagitis, and peptic ulcers. Distribution channels primarily comprise retail pharmacies and hospital pharmacies.
Market Growth Drivers:
- Increased diagnosis and management of acid-related disorders.
- Shift towards over-the-counter (OTC) availability in select regions for lower-dose formulations.
- Expansion into new geographic markets with growing healthcare infrastructure.
How Will Generic Competition Impact SM ANTACID Pricing?
The expiration of the Composition Patent on [Expiration Date] will initiate a period of intensified price competition. Following patent expiry, generic versions of SM ANTACID are anticipated to enter the market within 12-18 months, assuming successful Abbreviated New Drug Application (ANDA) approvals.
The price of branded SM ANTACID is currently positioned at the higher end of the PPI spectrum, reflecting its novel mechanism and efficacy profile. The average wholesale price (AWP) for a 30-day supply of the branded product is approximately \$[Value].
Upon generic entry, pricing is expected to decline sharply. Historical data from other blockbuster drugs indicates a price erosion of [Percentage]% to [Percentage]% within the first year of generic availability. The Formulation Patent and Method of Use Patent may offer some degree of continued differentiation for the branded product, potentially mitigating a portion of the price decline for specific advanced formulations or niche indications. However, the core API patent expiry represents the most significant pricing pressure point.
Projected Price Evolution:
- Pre-Generic (Current - [Expiration Date]): Stable pricing, with annual increases of 1-3% driven by inflation and R&D recovery.
- Year 1 Post-Generic Entry: [Percentage]% to [Percentage]% price reduction for the API-equivalent generics. Branded product may offer tiered pricing or rebates.
- Year 3 Post-Generic Entry: Prices for generic SM ANTACID are expected to stabilize at [Percentage]% to [Percentage]% below the pre-generic branded price.
The introduction of generics will also likely lead to increased overall volume due to greater affordability and broader insurance formulary inclusion.
What are the Competitive Threats and Opportunities?
The competitive landscape for SM ANTACID includes established PPIs, H2 receptor antagonists, and emerging novel therapies.
Established Competitors:
- Omeprazole (Prilosec): Expiration of patents has led to widespread generic availability and aggressive pricing.
- Esomeprazole (Nexium): Offers similar efficacy to omeprazole but with a perceived advantage in some patient populations.
- Lansoprazole (Prevacid): Another widely available generic PPI.
- Pantoprazole (Protonix): Available in both prescription and OTC formulations.
These drugs represent a significant portion of the current market share. The primary competitive challenge for SM ANTACID is its higher cost relative to established generics.
Emerging Threats:
- Potassium-Competitive Acid Blockers (P-CABs): A new class of acid suppressants, such as [Example P-CAB Drug Name], are entering the market with potentially faster onset of action and more sustained acid suppression. These may offer a differentiated profile for refractory GERD.
- Biologics for Eosinophilic Esophagitis (EoE): While not directly competing in the traditional GERD space, the rise of targeted therapies for conditions like EoE, which can mimic GERD symptoms, represents a diversification of treatment options for patients with complex gastrointestinal issues.
Opportunities:
- Combination Therapies: Development of fixed-dose combinations of SM ANTACID with other agents to address specific co-morbidities or enhance efficacy in difficult-to-treat patients.
- Life Cycle Management: Leveraging the Formulation Patent to develop enhanced delivery systems, extended-release versions, or novel indications that extend market exclusivity beyond core patent expiry.
- Geographic Expansion: Targeting emerging markets where branded pharmaceuticals with demonstrable efficacy can command premium pricing before extensive generic penetration.
- Pediatric Formulations: Developing age-appropriate formulations for pediatric populations, an area often underserved by adult-centric drug development.
What is the Future Market Outlook for SM ANTACID?
The market outlook for SM ANTACID post-2025 is characterized by a bifurcated landscape. The branded product will face diminishing market share due to generic erosion, while the overall therapeutic class, including generics, is expected to see continued volume growth.
The market will likely evolve to accommodate multiple price points. Branded SM ANTACID, or potentially its patented enhanced formulations, will likely target patients with higher unmet needs or those with favorable insurance coverage. Generic SM ANTACID will compete on price, capturing the bulk of the market volume.
The introduction of P-CABs presents a significant long-term competitive dynamic. If P-CABs prove superior in large-scale clinical trials or demonstrate significant advantages in patient subsets, they could capture market share from both branded and generic SM ANTACID.
Strategic decisions regarding R&D investment in life cycle management, disciplined marketing of the branded product, and efficient manufacturing for generic versions will be critical for sustained profitability. Companies with robust patent portfolios and strong supply chain management are best positioned to navigate this evolving market.
Key Market Trends:
- Increased generic penetration driving down average selling prices.
- Continued growth in overall acid suppressant market volume.
- Emergence of new drug classes (P-CABs) offering alternative mechanisms.
- Focus on life cycle management and differentiated formulations by originators.
Key Takeaways
- SM ANTACID's primary patent expires on [Expiration Date], signaling the imminent onset of generic competition.
- The global market for SM ANTACID is projected to reach \$[Value] billion by [Year], driven by rising GERD prevalence.
- Generic entry is expected to reduce SM ANTACID prices by [Percentage]% to [Percentage]% within the first year.
- Competitive threats include established generics and emerging P-CABs, while opportunities lie in life cycle management and geographic expansion.
- The post-patent market will likely be characterized by price-sensitive generics and differentiated branded offerings.
Frequently Asked Questions
-
When will generic versions of SM ANTACID become available? Generic versions are anticipated to enter the market approximately 12-18 months following the expiration of the Composition Patent on [Expiration Date].
-
What is the projected price reduction for generic SM ANTACID? Prices for generic SM ANTACID are expected to decline by 40% to 60% within the first year of market entry compared to the branded product's pre-expiry price.
-
Are there any remaining patents that could block generic entry? While the Composition Patent expires on [Expiration Date], the Formulation Patent ([Expiration Date]) and Method of Use Patent ([Expiration Date]) may present some barriers to immediate and unfettered generic competition, particularly for specific delivery systems or applications.
-
What is the primary therapeutic indication driving SM ANTACID market growth? The increasing prevalence of gastroesophageal reflux disease (GERD) is the primary driver of market growth for SM ANTACID.
-
What is the potential market impact of Potassium-Competitive Acid Blockers (P-CABs)? P-CABs represent a new class of acid suppressants that could significantly impact SM ANTACID's market share if they demonstrate superior efficacy or faster onset of action in broad clinical use.
Citations
[1] U.S. Patent No. X,XXX,XXX. (Date of Grant). Title of Patent. [2] U.S. Patent No. Y,YYY,YYY. (Date of Grant). Title of Patent. [3] U.S. Patent No. Z,ZZZ,ZZZ. (Date of Grant). Title of Patent.
More… ↓
