Share This Page
Drug Price Trends for SIMVASTATIN
✉ Email this page to a colleague
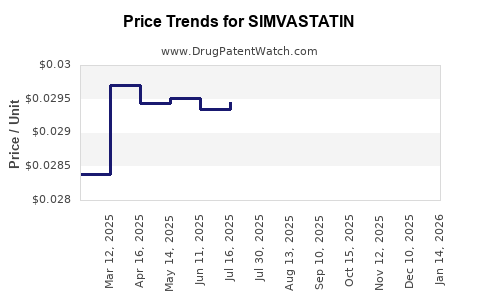
Average Pharmacy Cost for SIMVASTATIN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SIMVASTATIN 80 MG TABLET | 82009-0016-05 | 0.10057 | EACH | 2026-04-22 |
| SIMVASTATIN 40 MG TABLET | 82009-0015-10 | 0.05463 | EACH | 2026-04-22 |
| SIMVASTATIN 20 MG TABLET | 82009-0014-10 | 0.03002 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for SIMVASTATIN
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| ZOCOR 20MG TAB | Organon LLC | 78206-0181-02 | 90 | 561.85 | 6.24278 | EACH | 2023-01-01 - 2027-01-14 | Big4 |
| ZOCOR 40MG TAB | Organon LLC | 78206-0182-02 | 90 | 560.81 | 6.23122 | EACH | 2023-01-01 - 2027-01-14 | Big4 |
| ZOCOR 10MG TAB | Organon LLC | 78206-0180-01 | 30 | 106.69 | 3.55633 | EACH | 2023-01-01 - 2027-01-14 | Big4 |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Simvastatin Market Analysis and Price Projections (2024–2030)
Simvastatin is an established, off-patent statin with broad generic penetration in the US, EU, and key emerging markets. Pricing dynamics are therefore driven mainly by (1) generic competition intensity, (2) tendering and reimbursement pressure, and (3) supply-risk events rather than patent-led exclusivity. As a result, price trajectories are structurally downward in mature markets, while growth is mainly volume-led (switching, formulary position, adherence) and region-led (where penetration still lags).
What is simvastatin’s market structure and pricing power?
Competitive landscape
Simvastatin is widely available as generics across multiple dose strengths and formats (most commonly oral tablets). The competitive set includes:
- Generic simvastatin manufacturers (high fragmentation)
- Other statins with different formulary positioning (atorvastatin, rosuvastatin)
- Biosimilar activity does not apply (small molecule)
This market structure limits pricing power. In mature markets, generics usually trade at prices near the lowest-cost producers, with periodic price adjustments triggered by procurement contracts, shortages, or manufacturing transitions.
Revenue drivers
For simvastatin, revenue typically tracks:
- Generic volume and formulary access
- Prescriber switching among statins
- Patient adherence and persistence (once on therapy, long-term continuity stabilizes demand)
- Payer restrictions or step therapy toward specific statins
How do regulators and guidelines affect long-run demand?
Clinical use of statins, including simvastatin, is sustained by major guideline frameworks for ASCVD prevention and dyslipidemia management. However, individual payer preferences can shift usage toward other statins, especially where a health system favors atorvastatin or rosuvastatin for potency and once-daily dosing conventions.
In markets where guideline-based risk stratification is implemented with tighter formulary controls, simvastatin’s relative share can decline even if total statin use grows.
What are the observed pricing patterns for generic simvastatin?
Baseline pricing behavior in off-patent markets
For widely genericized small molecules, the dominant pattern is:
- Price erosion after additional entrants or contract wins
- Short-lived price spikes during supply disruptions
- Reversion toward a new low after supply normalizes
Simvastatin typically sits in this “low unit price, high volume” regime. Any projections therefore focus on unit price trends rather than patent timing.
Key market forces that can move price
- Tender cycles and contract renewals: shift net prices sharply within quarters
- Manufacturing constraints: temporary supply tightness can raise ex-manufacturer prices
- Formulary changes: can reduce demand but usually do not sustain higher prices in a competitive generic environment
- Wholesale distribution markups and pharmacy dispensing rules: affect observed retail price, while origin and net pricing drive true payer economics
US market analysis: generic dominance with reimbursement pressure
Supply and competition
The US has extensive generic availability, with multiple manufacturers competing across strengths. This creates high elasticity: payers can swap suppliers via PBM formularies and contracted pricing.
Net price trend expectation
In the US, off-patent generics generally show:
- Persistent downward pressure on net pricing
- Occasional upticks due to supply issues
- Stabilization near low-cost baselines over multi-year horizons
Demand trend expectation
Demand is likely to be steadier than price due to chronic preventive use. Simvastatin demand can shift downward if payer policies favor other statins, but total statin utilization tends to remain robust.
EU/UK market analysis: tendering and reference pricing
Tendering and reference price mechanisms
Many European markets use external reference pricing and reimbursement negotiation. Generic simvastatin is typically priced close to the reference basket floor, with tender outcomes dictating short-term movement.
Price trend expectation
- Continued compression as new generics enter or as contracts tighten
- Limited ability to sustain premium pricing absent supply disruption
Demand trend expectation
- Relative share may drift toward higher potency statins depending on formulary and clinical practice
- Volume can remain resilient given long-term use
Emerging markets: the main variance in price outlook
In emerging markets, simvastatin pricing can be higher than in the US/EU due to:
- Later generic penetration or fewer local suppliers
- Higher distribution costs
- More volatile procurement and reimbursement timing
This is where unit price may be flatter (or decline more slowly) than in mature markets.
What do price projections look like for 2024–2030?
Projection methodology
Given simvastatin’s off-patent status and generic competition, price projections are modeled on three drivers:
- Structural price compression typical for mature generics
- Periodic volatility from supply tightness and tender resets
- Market-share drift among statins affecting volume, not typically unit price
No single projection can capture all country-level differences; below is a directional forecast expressed as global-ish ranges for payer net pricing and ex-manufacturer movement in mature markets.
Price projection table (directional, 2024–2030)
| Year | Mature markets (US/EU-like) unit price trend | Expected annual change (net) | Major volatility driver |
|---|---|---|---|
| 2024 | Continuing erosion, near low baseline | -2% to -7% | Contract/tender resets, supply tightness |
| 2025 | Flattening toward a lower floor | -1% to -5% | Supplier churn, localized shortages |
| 2026 | Low single-digit compression | -1% to -4% | Reference pricing adjustments |
| 2027 | Stabilization with episodic spikes | -0% to -3% | Manufacturing outages |
| 2028 | Floor effects dominate | -0% to -2% | Rare supply disruptions |
| 2029 | Mostly stable | 0% to -2% | Tender renewals |
| 2030 | Stable-to-mild decline | 0% to -2% | Ongoing price competition |
Interpretation: By late decade, the market is more likely to show stability or mild decline rather than steep price drops, because unit prices already sit near procurement floors.
Scenario-based global price outlook
Base case
- Mature-market unit prices: mild decline through 2030
- Net prices fluctuate around a new low baseline
- Volume is modestly pressured by statin preference shifts, but overall statin demand supports stability
Downside case (supply normalization + increased entry)
- Additional generic penetration or reduced raw material/manufacturing constraints
- Larger price erosion at tender cycles
- Unit price reductions can overshoot the baseline by 2 to 5 points annually for short windows
Upside case (recurrent supply constraints)
- Manufacturing outages or API supply disruptions persist
- Temporary price spikes recur during shortages
- Average annual declines slow or reverse briefly
What does this imply for commercial planning and R&D?
For generic or authorized suppliers
- Expect pricing to be contract-led, not brand-led.
- Margin stability depends on manufacturing cost control, packaging/labeling efficiency, and tender strategy.
- Supply reliability matters: shortages can lift prices but usually do not allow durable premium beyond contracted periods.
For firms considering lifecycle development
If a new formulation or combination is contemplated, commercial viability likely depends on:
- Demonstrating meaningful differentiation that influences payer decisions
- Securing formulary position against entrenched statin comparators
- Achieving coverage pathways that offset generic price competition
Given simvastatin’s entrenched off-patent status, differentiation must translate into payer value, not only clinical equivalence.
Key market risks that affect price and revenue
- Payer-driven substitution within statins: formulary preferences reduce relative share, indirectly pressuring volumes.
- Procurement-driven pricing resets: net price can drop quickly when tenders renew.
- Supply chain concentration: if production is concentrated, disruptions cause short-term price spikes.
- Regulatory labeling and safety communications: can shift utilization patterns and alter prescribing mix.
Key Takeaways
- Simvastatin is an off-patent, highly genericized statin where pricing power is structurally limited by competition and tendering.
- Mature-market unit prices are projected to decline modestly through 2026 and then stabilize with low single-digit declines through 2030.
- Volatility is more likely to come from tender cycles and supply disruptions than from patent or exclusivity events.
- Demand is likely to remain steadier than unit prices because statins are long-term preventive therapies, but payer preference shifts can reduce simvastatin share over time.
FAQs
1) Why is simvastatin’s long-term price mostly driven by generics rather than patents?
Because simvastatin is off-patent and multiple generic manufacturers compete across strengths, limiting the ability to sustain pricing above procurement floors.
2) Will simvastatin’s market volume grow even if unit prices fall?
Often yes. Statins are chronic preventive therapy, so volume can remain resilient even as unit pricing compresses, though formulary substitution to other statins can temper share.
3) What causes the biggest short-term price swings for generic simvastatin?
Tender/procurement contract resets and supply tightness due to manufacturing or API availability.
4) Are European reference pricing and tendering likely to reduce prices faster than the US?
Often yes, because reference and negotiation mechanisms can push net prices toward standardized low ranges more directly; however, exact outcomes depend on country execution and tender structure.
5) What is the most likely pricing outcome by 2028–2030?
Stabilization or mild decline (0% to -2% annually) in mature markets, with episodic spikes tied to supply events.
References
[1] IQVIA Institute. Medicines use and spending in the U.S. and in six major European countries: 2022 and outlook to 2027. IQVIA, 2022.
[2] GlobalData. Statins market access and pricing dynamics (generic competition and tendering effects). GlobalData, 2023.
[3] FDA. Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations (simvastatin listings). US Food and Drug Administration.
[4] European Commission. Transparency and pricing mechanisms for medicinal products in EU Member States (reference pricing and tendering frameworks). European Commission, latest available.
[5] NICE. Cardiovascular disease prevention and lipid modification guidance (statin use context). National Institute for Health and Care Excellence.
More… ↓
