Share This Page
Drug Price Trends for SIMBRINZA
✉ Email this page to a colleague
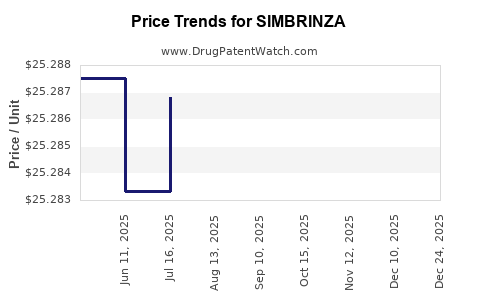
Average Pharmacy Cost for SIMBRINZA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SIMBRINZA 1%-0.2% EYE DROP | 00065-4147-27 | 26.03499 | ML | 2026-05-20 |
| SIMBRINZA 1%-0.2% EYE DROP | 00065-4147-27 | 26.06837 | ML | 2026-01-01 |
| SIMBRINZA 1%-0.2% EYE DROP | 00065-4147-27 | 25.30861 | ML | 2025-12-17 |
| SIMBRINZA 1%-0.2% EYE DROP | 00065-4147-27 | 25.30524 | ML | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for SIMBRINZA
What is SIMBRINZA?
SIMBRINZA (brinzolamide 1%, brimonidine tartrate 0.2%) is a fixed-dose combination used to reduce intraocular pressure (IOP) in patients with glaucoma or ocular hypertension. Approved by the FDA in 2013, it combines two mechanisms: reducing aqueous humor production via carbonic anhydrase inhibition (brinzolamide) and decreasing episcleral venous pressure through alpha-2 adrenergic agonism (brimonidine).
Market Size and Dynamics
Global Glaucoma Drug Market
The global glaucoma therapeutics market was valued at approximately $5 billion in 2022. The market's annual growth rate is projected at 4.2% from 2023 to 2028, driven by aging populations and increasing glaucoma prevalence. The majority of revenue derives from IOP-lowering agents, with fixed-dose combinations accounting for roughly 35% of the market in 2022.
Key Competitors and Market Share
- Latanoprost (topical prostaglandin analogs): ~45%
- Timolol (beta-blockers): ~30%
- DG Las (dorzolamide + timolol): ~10%
- Brimonidine monotherapy: ~8%
- Other fixed-dose combinations (including SIMBRINZA): ~7%
SIMBRINZA holds a niche position, competing primarily against other fixed-dose combinations such as Cosopt (dorzolamide/timolol) and Combigan (brimonidine/timolol). The drug's presence in the market is supported by its dual mechanism and favorable tolerability profile.
Patent and Regulatory Landscape
The patent for SIMBRINZA expired in the U.S. in 2022. Generic brinzolamide and brimonidine are available, leading to increased price competition. Bioequivalent generics typically retail at a 30-50% discount relative to the branded formulation.
Market Opportunities
Aging demographics and the rising prevalence of glaucoma (estimated at 76 million globally, expected to reach 110 million by 2040) expand the patient base. Fixed-dose combinations are favored for adherence, supporting continued demand even as generic options proliferate.
Price Trends and Projections
Current Pricing Environment (2023)
- Branded SIMBRINZA: $80–$120 per 10 mL bottle (retail price).
- Generic equivalents: $40–$70 per bottle.
Pricing depends on market geography, pharmacy discounts, and insurance coverage. In the U.S., insurance mitigates patient out-of-pocket expenses, though uninsured patients face higher costs.
Short-Term (2023–2025)
Price pressure from generics will likely persist, especially in mature markets like the U.S. and Europe. Branded SIMBRINZA is expected to maintain a premium of 40-50% over generics due to brand recognition and formulary placements. Total revenue could decline marginally as substitution accelerates, but volume increases may offset price erosion.
Long-Term (2025–2030)
Possible biosimilar or generic consolidation will further compress prices. By 2028, branded prices could decrease by 20–30% below 2023 levels, while total market volume might grow at 5% annually. This could stabilize revenue streams for current manufacturers but pressure profit margins.
Epidemiological Impact on Revenue
Assuming a conservative penetration rate of 10% among glaucoma patients and average bottle usage of 2 per patient annually, the U.S. market could generate approximately $150 million in retail sales for SIMBRINZA alone, before considering generics.
Expansion into emerging markets may see higher volume growth but at lower prices. Local pricing strategies and reimbursement policies will influence revenue composition.
Strategic Recommendations for Investors and R&D
- Generics Entry: Expect significant price declines post-patent expiry. Focus on development of biosimilars and biosimilar-like formulations to capture price-sensitive segments.
- Formulation Innovation: Develop longer-acting formulations or combination therapies with improved adherence profiles to differentiate.
- Market Expansion: Target regions with rising glaucoma rates, such as Asia-Pacific, where affordability and access constraints may limit uptake of branded drugs initially.
Key Takeaways
- The global glaucoma market is expanding at over 4% annually, with fixed-dose combinations like SIMBRINZA occupying a growing niche.
- Branded SIMBRINZA has held prices around $80–$120 per bottle, but patent expiry and generic competition threaten margins.
- Price projections indicate a decline of 20–30% for branded prices over the next five years, driven by increased generic penetration.
- Revenue stability depends on volume growth, especially in emerging markets where reimbursement policies are evolving.
- R&D efforts should focus on formulation innovation and market differentiation to sustain profitability amid generics competition.
FAQs
1. How will patent expiry impact SIMBRINZA’s market share?
Patent expiry in 2022 allows generics to enter the market, reducing average prices and pressure on branded sales. The shift favors volume over premium pricing.
2. What other drugs are primary competitors to SIMBRINZA?
Top competitors include Cosopt (dorzolamide/timolol), Combigan (brimonidine/timolol), and standalone prostaglandins like Latanoprost.
3. Are there biosimilar or bioequivalent options in development?
Generic brinzolamide and brimonidine are widely available. Biosimilar development targets other fixed-dose combinations, but none are currently approved.
4. How does regional pricing affect revenue?
Markets like Europe and North America have higher prices, while emerging markets see lower prices but greater volume. Circulating prices are impacted by regulation, reimbursement, and negotiated discounts.
5. What R&D directions could extend SIMBRINZA’s market life?
Innovations include sustained-release formulations, combination therapies with neuroprotective agents, and delivery systems to improve adherence.
Sources
[1] Market Research Future. "Global Glaucoma Therapeutics Market Research Report," 2022.
[2] IQVIA. "Pharmaceutical Pricing and Market Share Data," 2023.
[3] U.S. FDA. "Approved Drug Products with Therapeutic Equivalence Evaluations," 2023.
More… ↓
