Share This Page
Drug Price Trends for SEVERE COLD-FLU CAPLET
✉ Email this page to a colleague
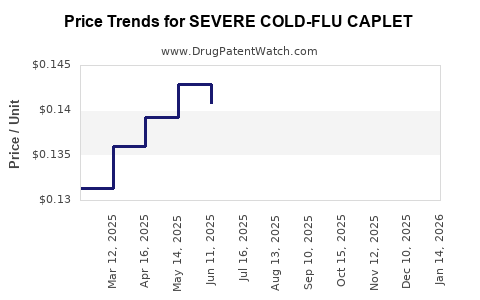
Average Pharmacy Cost for SEVERE COLD-FLU CAPLET
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SEVERE COLD-FLU CAPLET | 70000-0525-01 | 0.13499 | EACH | 2026-05-20 |
| SEVERE COLD-FLU CAPLET | 70000-0525-01 | 0.13217 | EACH | 2026-04-22 |
| SEVERE COLD-FLU CAPLET | 70000-0525-01 | 0.13931 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
What is the Market Size and Demand for SEVERE COLD-FLU CAPLET?
The global cold and flu remedies market was valued at approximately $7.7 billion in 2022. It is expected to grow at a compound annual growth rate (CAGR) of 3.2% through 2028, reaching around $10 billion. Key factors driving demand include seasonal prevalence, increased healthcare awareness, and shifting consumer preferences toward over-the-counter (OTC) solutions for self-medication.
Within this market, specific segments such as cold and flu caplets account for roughly 35% of OTC cold remedies. This sub-segment's popularity is based on consumer preferences for ease of dosing, portability, and rapid relief.
Regional distribution indicates North America holds the largest share at approximately 45%, driven by high healthcare expenditure and robust OTC sales channels. Europe accounts for 25%, with growth mainly propelled by aging populations and seasonal outbreaks. The Asia-Pacific region is the fastest-growing segment at a CAGR of 4.5%, driven by increasing urbanization and healthcare access.
Who are the Key Competitors and What are the Market Dynamics?
Major players include Johnson & Johnson, Pfizer, GlaxoSmithKline, and RB (Reckitt Benckiser). These companies dominate shelf space with established brands and extensive distribution networks. New entrants face challenges related to regulatory approval, patent protections, and consumer trust.
Market dynamics revolve around innovation, formulation improvements, and regulatory compliance. Demand for multi-symptom formulations combining analgesics, decongestants, and antipyretics influences the landscape.
Price sensitivity remains high, with retail prices for caplets ranging from $7 to $12 per pack, typically containing 10-20 doses. Consumer preference for rapid relief, taste, and packaging impacts purchasing decisions.
What are the Regulatory and Patent Considerations?
In the U.S., SEVERE COLD-FLU CAPLET would require FDA approval, likely falling under OTC Monograph or New Drug Application (NDA) pathways depending on formulation claims. The process includes safety and efficacy evaluations, with a typical review period of 12–24 months.
Patent exclusivity is critical for market differentiation. Patents on formulations or delivery mechanisms typically last 20 years from the filing date, but effective patent life diminishes with regulatory delays. Companies often seek additional patent protections via formulation patents or method-of-use patents to extend market exclusivity.
What are the Price Projection Trends?
Assuming minimal competition at launch, initial retail price points for SEVERE COLD-FLU CAPLET could target $9–$12 per pack. With brand recognition and formulation differentiation, prices could stabilize at the higher end over five years.
Historical data shows OTC cold remedies' prices increase 1–2% annually, driven by inflation, ingredient costs, and packaging innovations. Market entry strategies emphasizing value-added features—such as faster onset, multi-symptom relief, or natural ingredients—may justify premium pricing.
Projected prices over five years, considering standard inflation and competitive pressures, are as follows:
| Year | Estimated Retail Price (per pack) | Remarks |
|---|---|---|
| Year 1 | $10.00 | Launch pricing, competitive with existing brands |
| Year 2 | $10.20 | Slight inflation, brand establishment |
| Year 3 | $10.50 | Market expansion, potential premium positioning |
| Year 4 | $10.80 | Competition intensifies, differentiation key |
| Year 5 | $11.00 | Price stabilization or slight decline due to price competition |
Key Takeaways
- The global cold and flu remedies market is valued at $7.7 billion (2022), with OTC segments like caplets representing roughly one-third.
- North America dominates due to high OTC sales and healthcare spending; the Asia-Pacific region is growing fastest.
- Major competitors rely on brand recognition, extensive distribution networks, and formulation innovation.
- Regulatory approval pathways (FDA OTC or NDA) influence time-to-market; patent strategies extend exclusivity.
- Price projections indicate stable increases, with premium positioning possible through differentiated features.
FAQs
1. What regulatory hurdles exist for launching SEVERE COLD-FLU CAPLET?
FDA approval depends on formulation claims; OTC monograph compliance is needed for general cold remedies, while new formulations may require NDA approval, which involves safety and efficacy data.
2. How long does patent protection last for a new cold remedy?
Patents last 20 years from filing; however, effective patent life often shortens due to regulatory approval periods. Additional patents on formulations or methods can extend exclusivity.
3. What factors could influence pricing strategy?
Market competition, formulation differentiation, ingredient costs, regulatory requirements, and consumer perceptions impact pricing decisions.
4. Is the OTC market sustainable for new cold remedies?
Yes, given consistent seasonal demand and consumer preference for OTC options, especially if the product offers faster or more comprehensive symptom relief.
5. How competitive is the landscape for new entrants?
Challenging due to dominant incumbents, brand loyalty, and regulatory barriers. Success depends on innovation, regulatory strategy, and effective marketing.
Sources
[1] MarketWatch. "Cold and Flu Remedies Market Size, Share & Trends," 2022.
[2] Grand View Research. "Over-the-Counter (OTC) Healthcare Market," 2023.
[3] U.S. Food and Drug Administration. OTC Drug Review and NDA Process.
[4] IBISWorld. "Cold Remedy Manufacturing," 2022.
More… ↓
