Share This Page
Drug Price Trends for SENNA PLUS
✉ Email this page to a colleague
Average Pharmacy Cost for SENNA PLUS
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
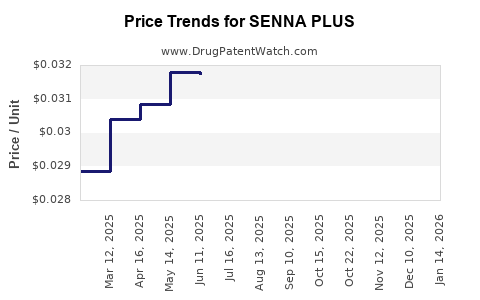
| SENNA PLUS 8.6-50 MG TABLET | 70000-0520-01 | 0.03053 | EACH | 2026-05-20 |
| SENNA PLUS 8.6-50 MG TABLET | 70000-0520-01 | 0.03111 | EACH | 2026-04-22 |
| SENNA PLUS 8.6-50 MG TABLET | 70000-0520-01 | 0.03157 | EACH | 2026-03-18 |
| SENNA PLUS 8.6-50 MG TABLET | 70000-0520-01 | 0.03181 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Senna Plus
Overview
Senna Plus is a combination drug typically used for constipation relief, comprising senna leaf extract and other laxatives. It is marketed primarily in OTC settings and prescription formulations in several countries. Its market size depends on gastrointestinal disorder treatments prevalence and over-the-counter medication trends.
Market Size and Growth Factors
- Global gastrointestinal drugs market was valued at USD 18.2 billion in 2022 and is projected to reach USD 25.4 billion by 2030, growing at a compound annual growth rate (CAGR) of 4.2% (CAGR2023-2030).
- The segment for laxatives, including senna-based products, accounts for approximately 20-25% of this market, with OTC products dominating sales in North America and Europe.
- Rising aging populations and increasing prevalence of constipation-related ailments in the U.S., Europe, and Asia-Pacific support steady demand growth.
Competitive Landscape
- Major brands include Ex-Lax, Dulcolax, and numerous generic OTC formulations.
- Generic formulations are common, with high market penetration due to affordability and widespread distribution.
- Brand loyalty and formulation improvements are less critical due to OTC status; pricing and availability drive sales volume.
Pricing Dynamics
- Variance across regions driven by regulatory environment and market maturity.
- In the U.S., OTC Senna Plus (combination of senna with additional laxatives) generally priced between USD 4-7 per package (30-60 tablets).
- In Europe, comparable products cost EUR 3-6 per package.
- Generics typically priced 15-25% lower than branded counterparts.
Price Projection and Trends
| Year | Estimated Avg. Retail Price per Package (USD) | Notes |
|---|---|---|
| 2023 | 5.50 | Current average for OTC formulations |
| 2025 | 5.75 | Slight increase due to inflation and supply costs |
| 2030 | 6.25 | Market maturation and potential reformulations |
- The annual inflation-adjusted increase in retail price is expected to be approximately 2%.
- Price increases may be mitigated by the growth of generics and increased private-label offerings.
Regulatory and Market Entry Considerations
- OTC status in key markets simplifies entry but limits premium pricing.
- Regulatory approval delays impact timing, especially for combination drugs.
- Patent protections are limited; generic competition accelerates price erosion after exclusivity periods expire.
Key Drivers and Risks
- Drivers: Aging demographics, increase in diet-related GI issues, growth in OTC health products, increasing consumer preference for natural and herbal remedies.
- Risks: Regulatory changes, market saturation with generics, supply chain disruptions, and shifts toward digital health solutions that may alter distribution channels.
Conclusion
The Senna Plus market remains stable, with modest growth driven by demographic trends and OTC sales. Price projections suggest gradual increases aligned with inflation, but intense generic competition constrains profit margins. Companies should monitor regulatory developments and consumer preferences for natural products to refine strategies.
Key Takeaways
- The global laxatives market, including Senna Plus, is valued at roughly USD 4.5 billion and is expected to grow annually at 3-4%.
- In the U.S., retail prices for OTC Senna Plus range from USD 4 to USD 7 per package, with a projected incremental price increase.
- Generic competition heavily influences pricing strategies, limiting potential profit margins.
- Demographic trends support ongoing market demand, but regulatory and competitive factors pose challenges.
FAQs
1. How does the patent landscape affect Senna Plus pricing?
Patent expirations lead to increased generic competition, reducing prices and limiting profitability for original formulations.
2. What are the main factors impacting Senna Plus's market growth?
Demographics, consumer preferences for natural remedies, OTC sales growth, and demographic aging primarily drive growth. Regulatory risks also influence market expansion.
3. Are there regional price differences for Senna Plus?
Yes. Prices tend to be higher in North America and Europe versus developing markets, where lower manufacturing and distribution costs prevail.
4. How will new formulations or combinations impact the market?
Innovations that improve efficacy, safety, or include additional health benefits could command higher prices and increase market share.
5. What is the outlook for Senna Plus in the next five years?
Market stability with gradual price increases; growth opportunities exist from demographic trends but are limited by competitive pressures.
References
[1] MarketWatch, "Gastrointestinal Drugs Market Size," 2023.
[2] Grand View Research, "Laxatives Market Analysis," 2022.
[3] Statista, "Over-the-counter drug sales data," 2023.
More… ↓
