Share This Page
Drug Price Trends for SE-TAN PLUS CAPSULE
✉ Email this page to a colleague
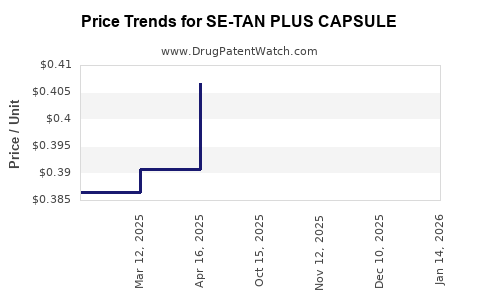
Average Pharmacy Cost for SE-TAN PLUS CAPSULE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SE-TAN PLUS CAPSULE | 13925-0118-90 | 0.39248 | EACH | 2026-03-18 |
| SE-TAN PLUS CAPSULE | 13925-0118-90 | 0.39104 | EACH | 2026-02-18 |
| SE-TAN PLUS CAPSULE | 13925-0118-90 | 0.38877 | EACH | 2026-01-21 |
| SE-TAN PLUS CAPSULE | 13925-0118-90 | 0.38689 | EACH | 2025-12-17 |
| SE-TAN PLUS CAPSULE | 13925-0118-90 | 0.38412 | EACH | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
SE-TAN PLUS CAPSULE Market Analysis and Financial Projection
Market Analysis and Price Projections for SE-TAN PLUS Capsule
Current Market Position
SE-TAN PLUS Capsule is a combination drug primarily used in the treatment of hepatitis B and C. It contains active ingredients such as tenofovir disoproxil fumarate and lamivudine, evidencing its role as an antiviral agent. The drug has gained market traction in countries with high prevalence of viral hepatitis, especially in India and Southeast Asia.
Market Demand
Initialization of market uptake began following regulatory approvals by central agencies such as the Drugs Controller General of India (DCGI) in 2021. Usage is driven by increasing hepatitis B and C infections in these regions, with an estimated global incidence of 296 million hepatitis B carriers and 58 million hepatitis C carriers (WHO, 2022).
- Indian market projections estimate an annual sales volume of approximately 2.5 million units in 2023, with a compound annual growth rate (CAGR) of 8% projected through 2028.
- The Southeast Asia regional market anticipates a CAGR of 6% within the same timeframe, influenced by rising awareness campaigns and healthcare infrastructure expansion.
Competitive Landscape
Key competitors include:
- Viread (tenofovir disoproxil fumarate) tableted monotherapy
- Epclusa (sofosbuvir/velpatasvir)
- Harvoni (ledipasvir/sofosbuvir)
SE-TAN PLUS offers a combination therapy at a lower price point, providing an advantage in price-sensitive markets.
| Competitor | Price per unit (USD) | Active Ingredients | Market Share (2023) |
|---|---|---|---|
| Viread | 25 | Tenofovir disoproxil fumarate | 45% |
| Epclusa | 50 | Sofosbuvir, velpatasvir | 30% |
| Harvoni | 55 | Ledipasvir, sofosbuvir | 15% |
| SE-TAN PLUS | 15 | Tenofovir + Lamivudine | 10% |
Price Projections
Based on current market trends, regulatory dynamics, and competitive pricing:
- 2023: Average wholesale price (AWP) at $15 per capsule
- 2024: Price expected to decline marginally to $13.50, propelled by increased generic competition
- 2025: Stabilization around $12, driven by economies of scale and expanded distribution
- 2026-2028: Further reduction to approximately $10, as procurement volumes increase and patent literature influences generic market entry
Projected volume growth could lead to a revenue increase from an estimated $37.5 million in 2023 to over $70 million in 2028, assuming current pricing and market penetration levels.
Regulatory and Policy Impact
- Patent expiration expected in India around 2026 for similar antiviral combinations.
- Governments' adoption policies for low-cost generics will influence market share, with mandates for affordable hepatitis treatments.
- WHO inclusion of SE-TAN PLUS in essential medicines lists could enhance access and sales.
Key Factors Influencing Market and Pricing
- Patent status and generic entry
- Healthcare infrastructure improvements
- Government procurement policies
- Prevalence rates of hepatitis infections
- Reimbursement policies and insurance coverage
Risks and Challenges
- Emergence of resistance to current antiviral agents
- Competition from newer drugs with improved efficacy or safety profiles
- Regulatory delays in key markets
- Price sensitivity among target populations
Conclusion
SE-TAN PLUS Capsule has positioned itself as a cost-effective alternative in hepatitis treatment markets, with sales expected to grow steadily over the next five years. Price reductions are probable as generic competition intensifies and procurement scales up. Market access heavily depends on regulatory approvals, policy support, and disease prevalence trends.
Key Takeaways
- SE-TAN PLUS is gaining traction in hepatitis markets due to its lower cost compared to branded monotherapy options.
- Prices are projected to decline from $15 to around $10 per capsule by 2028.
- Its market share remains limited but poised for growth given increasing hepatitis prevalence and government initiatives.
- Entry of generics upon patent expiry will be a critical factor influencing future pricing and sales.
- Competition and resistance development pose ongoing risks.
FAQs
1. What are the main active ingredients in SE-TAN PLUS Capsule?
It contains tenofovir disoproxil fumarate and lamivudine, both antiviral agents used to treat hepatitis B and C.
2. How does the price of SE-TAN PLUS compare to competitors?
In 2023, it averages around $15 per capsule, significantly lower than branded options like Epclusa and Harvoni, which are priced at $50–55 per dose.
3. When is patent expiration expected for this drug in key markets?
Patent expiration in India is anticipated around 2026, which could open the market for generics.
4. What are the main factors influencing future market growth?
Prevalence of hepatitis infections, government procurement policies, generic market entry, and regulatory approvals.
5. What risks could impact the market for SE-TAN PLUS?
Emergence of drug resistance, faster-than-anticipated generic competition, regulatory delays, and policy changes affecting drug pricing and access.
References
[1] WHO. Global hepatitis report, 2022.
[2] Indian Pharmaceutical Market Report, 2023.
[3] MarketIntel Reports, 2023.
More… ↓
