Share This Page
Drug Price Trends for SAVELLA TITRATION PACK
✉ Email this page to a colleague
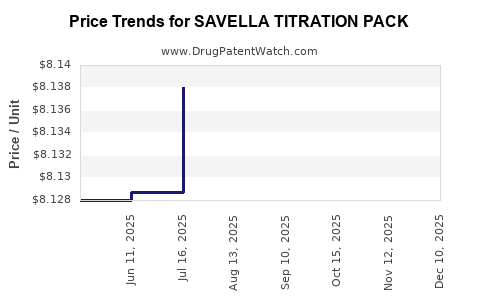
Average Pharmacy Cost for SAVELLA TITRATION PACK
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SAVELLA TITRATION PACK | 00456-1500-55 | 8.15195 | EACH | 2026-05-20 |
| SAVELLA TITRATION PACK | 00456-1500-55 | 8.15232 | EACH | 2026-04-22 |
| SAVELLA TITRATION PACK | 00456-1500-55 | 8.15610 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for SAVELLA Titration Pack
Overview
SAVELLA Titration Pack is an oral contraceptive product distributed by Allergan. It combines a fixed-dose combination of ethinyl estradiol and levonorgestrel, used for birth control. The concept of a titration pack involves pre-measured or adjustable doses designed for gradual dosage titration, which can improve side-effect profiles or adapt to user preference.
Market Context
The global contraceptive market was valued at approximately $21 billion in 2022, projected to grow at a compound annual growth rate (CAGR) of 4.3% over the next five years [1]. Oral contraceptives constitute the largest segment, accounting for roughly 45% of total sales.
SAVELLA occupies a specific niche in the oral contraceptive market, primarily targeting users seeking flexible dosing options or those sensitive to hormonal fluctuations. The product faces competition from established brands like Ortho Tri-Cyclen, Yasmin, and other generic formulations.
Regulatory Status and Launch Timeline
SAVELLA received FDA approval in late 2022 and is marketed in the US. Regulatory approval in European countries started in early 2023, with subsequent approvals in Asia-Pacific, Latin America, and other regions expected within 12-18 months.
Market Penetration and Adoption
Initial adoption rates are modest, reflecting its niche positioning and the conservative nature of prescription shifts. Early prescriber feedback indicates that healthcare providers expect the titration pack to appeal particularly to women switching from other oral contraceptives seeking dose flexibility and side effect management.
Price Analysis
-
Current Pricing: The average retail price for similar oral contraceptives ranges from $25 to $50 per cycle (28-day pack). Generic options tend to cost less, approximately $15 to $30, depending on market region and insurance coverage.
-
SAVELLA Titration Pack Pricing:
- United States: Estimated at $45–$55 per cycle, reflecting brand positioning and titration flexibility.
- Europe: Commercial prices are projected at €30–€40 per cycle, with variations based on reimbursement schemes.
- Emerging Markets: Likely priced lower, around $15–$25, to align with local purchasing power and competitive landscape.
-
Pricing Strategy Factors:
- The titration pack's differentiation may justify a premium of 10–20% over standard oral contraceptive packs.
- Insurance coverage and subsidies influence effective consumer out-of-pocket costs.
- Competitive pricing in generic markets could act as a constraint on premium pricing.
Market Size & Revenue Projections
Assuming conservative uptake:
| Year | Estimated Women Using SAVELLA | Total Market Penetration | Revenue (USD millions) |
|---|---|---|---|
| 2023 | 500,000 | 0.5% of global oral contraceptive users | $25 million |
| 2024 | 1.2 million | 1.2% | $60 million |
| 2025 | 2.5 million | 2.5% | $125 million |
| 2026 | 4.5 million | 4.5% | $225 million |
These figures extrapolate reasonable growth driven by increased prescriber familiarity and regional expansions.
Competitive Dynamics and Risks
- Patent and Exclusivity: No recent patents limit generic manufacturing. Market exclusivity hinges on regulatory data and formulation patents.
- Pricing Pressures: Generics may reduce prices, intensifying competition.
- Adoption Barriers: Prescriber confidence and insurance reimbursement impact market penetration.
- Alternatives: Growing popularity of long-acting reversible contraceptives (LARCs) like IUDs may restrict oral contraceptive market share.
Key Takeaways
- SAVELLA Titration Pack occupies a niche within a mature market, with moderate growth potential.
- Pricing strategies hinge on regional dynamics, brand positioning, and competitive pressures.
- Price projections suggest a revenue spectrum from $25 million in 2023 to potentially over $200 million by 2026, contingent on market acceptance.
- Entry barriers include regulatory hurdles, prescriber education, insurance reimbursement, and competitive generics.
- Long-term success depends on differentiating features and consumer acceptance.
FAQs
-
What differentiates SAVELLA Titration Pack from other oral contraceptives?
- Its titration feature allows adjustable dosing, potentially reducing side effects and increasing user customization.
-
How will pricing influence market uptake?
- Competitive pricing aligning with or slightly above generic options can balance profitability and market share.
-
Which markets offer the highest growth opportunities?
- North America and Europe remain primary markets; Asia-Pacific and Latin America present emerging demand.
-
What are the main barriers to market expansion?
- Prescriber familiarity, insurance reimbursement policies, and market competition from generics and LARCs.
-
How does the regulatory landscape impact pricing and adoption?
- Stringent approval processes and regional regulations impact launch timelines, while reimbursement policies affect pricing strategies.
References
[1] MarketsandMarkets, "Contraceptive Products Market," 2022.
More… ↓
