Share This Page
Drug Price Trends for ROFLUMILAST
✉ Email this page to a colleague
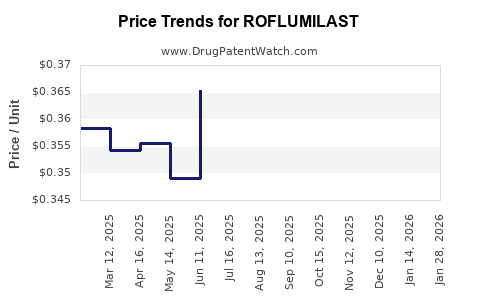
Average Pharmacy Cost for ROFLUMILAST
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ROFLUMILAST 250 MCG TABLET | 31722-0676-32 | 1.96206 | EACH | 2026-04-22 |
| ROFLUMILAST 250 MCG TABLET | 67877-0730-32 | 1.96206 | EACH | 2026-04-22 |
| ROFLUMILAST 250 MCG TABLET | 68382-0624-30 | 1.96206 | EACH | 2026-04-22 |
| ROFLUMILAST 250 MCG TABLET | 67877-0730-93 | 1.96206 | EACH | 2026-04-22 |
| ROFLUMILAST 250 MCG TABLET | 00904-7493-71 | 1.96206 | EACH | 2026-04-22 |
| ROFLUMILAST 250 MCG TABLET | 42571-0369-83 | 1.96206 | EACH | 2026-04-22 |
| ROFLUMILAST 500 MCG TABLET | 72205-0200-90 | 0.31870 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for ROFLUMILAST
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| ZORYVE 0.3% CREAM,TOP | Arcutis Biotherapeutics, Inc. | 80610-0130-60 | 60GM | 616.67 | 10.27783 | GM | 2024-01-01 - 2028-01-14 | Big4 |
| ZORYVE 0.3% CREAM,TOP | Arcutis Biotherapeutics, Inc. | 80610-0130-60 | 60GM | 812.56 | 13.54267 | GM | 2024-01-01 - 2028-01-14 | FSS |
| ZORYVE 0.3% CREAM,TOP | Arcutis Biotherapeutics, Inc. | 80610-0130-60 | 60GM | 842.62 | 14.04367 | GM | 2024-02-01 - 2028-01-14 | FSS |
| ZORYVE 0.3% FOAM,TOP | Arcutis Biotherapeutics, Inc. | 80610-0430-60 | 60GM | 642.25 | 10.70417 | GM | 2024-02-01 - 2028-01-14 | FSS |
| ZORYVE 0.3% CREAM,TOP | Arcutis Biotherapeutics, Inc. | 80610-0130-60 | 60GM | 597.78 | 9.96300 | GM | 2023-01-15 - 2028-01-14 | Big4 |
| ZORYVE 0.3% FOAM,TOP | Arcutis Biotherapeutics, Inc. | 80610-0430-60 | 60GM | 619.16 | 10.31933 | GM | 2024-04-01 - 2028-01-14 | Big4 |
| ZORYVE 0.3% CREAM,TOP | Arcutis Biotherapeutics, Inc. | 80610-0130-60 | 60GM | 812.56 | 13.54267 | GM | 2023-01-15 - 2028-01-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Roflumilast Market Analysis and Price Projections
Roflumilast, a phosphodiesterase-4 (PDE4) inhibitor, is primarily utilized in the treatment of severe chronic obstructive pulmonary disease (COPD). Its mechanism of action involves reducing inflammation in the airways, a key factor in COPD exacerbations. The global market for roflumilast is influenced by factors including COPD prevalence, generic competition, and the development of novel delivery systems or combination therapies. This analysis provides an overview of the current market landscape, patent status, and price projections for roflumilast.
What is the Current Market Landscape for Roflumilast?
The market for roflumilast is characterized by the presence of both branded and generic versions. The primary indication for roflumilast is severe COPD in patients with a history of exacerbations.
- Branded Roflumilast (Daliresp/Daxas): Manufactured by AstraZeneca, Daliresp (U.S.) and Daxas (Europe) were the initial branded formulations. The approval of roflumilast by the U.S. Food and Drug Administration (FDA) was in March 2010 [1]. European Medicines Agency (EMA) approval followed in July 2009 [2].
- Generic Roflumilast: Following patent expirations, multiple generic versions of oral roflumilast have entered the market, significantly increasing competition and driving down prices. Major pharmaceutical companies and contract manufacturing organizations are involved in the production of generic roflumilast.
- Therapeutic Landscape: Roflumilast is typically prescribed as an add-on therapy for patients with severe COPD who have not achieved adequate control with other bronchodilator treatments and who have a history of exacerbations. It is not a rescue medication for acute bronchospasm.
- Geographic Distribution: The U.S. and major European markets represent the largest commercial segments for roflumilast due to their high COPD burden and established healthcare infrastructure. Emerging markets are showing increasing potential as COPD awareness and diagnosis rates rise.
- Market Drivers:
- Increasing prevalence of COPD globally, linked to aging populations and environmental factors.
- Growing recognition of the role of PDE4 inhibition in managing COPD inflammation.
- Launch of generic alternatives expanding patient access.
- Market Restraints:
- Side effect profile, particularly gastrointestinal disturbances (nausea, diarrhea, weight loss) and psychiatric effects (insomnia, anxiety).
- Competition from other COPD treatment classes, including long-acting bronchodilators (LABAs, LAMAs) and inhaled corticosteroids (ICS).
- The requirement for oral administration, which can be less convenient for some patients compared to inhaled therapies.
What is the Patent Status of Roflumilast?
The patent landscape for roflumilast is a critical determinant of market exclusivity and the subsequent entry of generic competitors. The primary patents protecting the original branded formulations have largely expired in major markets.
- Key Patents: The original patent for roflumilast was filed by Byk Gulden Lomberg Chemische Fabrik GmbH (later acquired by AstraZeneca). This patent covered the compound itself and its therapeutic use.
- U.S. Patent No. 5,804,574, covering roflumilast and related compounds, was granted in 1998. This patent has expired.
- European Patent EP 0640625 B1, also covering roflumilast, has also expired.
- Exclusivity Periods:
- U.S. Market: Initial market exclusivity for Daliresp expired as generic manufacturers successfully challenged patents or reached settlements. The first generic roflumilast products became available in the U.S. around 2019.
- European Market: Similar to the U.S., patent expiries in Europe led to the introduction of generic Daxas.
- Secondary Patents and Litigation: While core compound patents have expired, there may be secondary patents covering specific formulations, polymorphs, manufacturing processes, or new indications. These can sometimes lead to patent litigation that can delay or prevent generic entry.
- There have been instances of patent challenges and litigation in both the U.S. and Europe concerning roflumilast, primarily focused on extending exclusivity periods. However, these have largely been resolved, allowing generic competition.
- Future Patent Considerations: Ongoing research into novel delivery systems (e.g., inhaled formulations) or combination therapies involving roflumilast could lead to new patent applications and potential market exclusivity periods for these newer iterations. However, the core oral formulation is now in the public domain in key markets.
- Data Source: Patent databases (e.g., USPTO, EPO) and pharmaceutical litigation trackers are primary sources for detailed patent information and litigation status.
What are the Projected Market Growth and Price Trends for Roflumilast?
The market for roflumilast is expected to experience moderate growth, driven by increasing COPD prevalence, while prices are projected to continue a downward trend due to generic competition.
Market Growth Projections:
- Compound Annual Growth Rate (CAGR): The global roflumilast market is projected to grow at a CAGR of approximately 4-6% between 2023 and 2028. This growth is primarily attributed to the expanding COPD patient population and increased diagnosis rates, particularly in emerging economies.
- Market Size: The global market size for roflumilast was estimated to be around USD 800 million to USD 1.1 billion in 2022. Projections indicate a market size of USD 1.2 billion to USD 1.5 billion by 2028 [3, 4].
- Geographic Growth:
- North America and Europe: These regions will continue to represent significant market shares but will see slower growth rates due to market maturity and established generic penetration.
- Asia-Pacific: This region is expected to exhibit the highest growth rate, driven by rising COPD incidence, improved healthcare access, and increasing awareness of advanced treatment options.
- Latin America and Middle East & Africa: These markets are also anticipated to contribute to growth as healthcare infrastructure develops and more patients gain access to treatment.
- Factors Influencing Growth:
- Aging Population: The demographic trend of an aging global population directly correlates with an increased incidence of chronic diseases like COPD.
- Environmental Factors: Air pollution and smoking rates, though declining in some developed nations, remain significant drivers of COPD in many parts of the world.
- Diagnostic Advancements: Improved diagnostic tools and increased physician awareness of COPD severity assessments can lead to more patients being identified and prescribed appropriate treatments like roflumilast.
- Pipeline Developments: While less likely for the oral formulation, any successful development of inhaled or combination therapies involving roflumilast could create new market segments and boost overall growth.
Price Trend Projections:
- Branded vs. Generic Pricing: The introduction of generic roflumilast has led to a substantial price reduction compared to the original branded product.
- Branded Daliresp/Daxas: Historically, prices ranged from USD 300 to USD 500 per month (patient out-of-pocket cost before insurance/rebates) [5].
- Generic Roflumilast: Generic versions are now available at significantly lower prices. The average monthly cost for generic roflumilast can range from USD 50 to USD 150, depending on the manufacturer, dosage, and pharmacy [5].
- Price Erosion: The market is expected to experience continued price erosion for generic roflumilast as competition intensifies among multiple manufacturers. This trend is typical for drugs that have faced patent expiry.
- Price Stability for Novel Formulations: If new, patented formulations of roflumilast (e.g., inhaled, combination therapies) are developed and approved, they could command premium pricing, at least until their own patent expiries. However, the core oral therapy will remain subject to significant price pressure.
- Reimbursement Policies: Reimbursement policies by public and private payers will continue to influence actual patient costs. Generic substitution mandates and preferred drug lists will further drive down the cost of roflumilast.
- Forecasting:
- 2023-2025: Continued downward price pressure on generic oral roflumilast, with average monthly costs potentially decreasing by an additional 10-20%.
- 2026-2028: Price stabilization for generics is likely as the market reaches a competitive equilibrium, but further significant drops are improbable unless new competitive pressures emerge.
What are the Key Competitors and Alternative Therapies for Roflumilast?
Roflumilast operates within a competitive therapeutic landscape for COPD management. Competitors include other pharmacological classes and, increasingly, newer delivery technologies and combination therapies.
Direct Competitors (Other PDE4 Inhibitors):
- Currently, roflumilast is the most prominent and widely prescribed oral PDE4 inhibitor for COPD. While research into other PDE4 inhibitors for COPD continues, no other oral agents have achieved widespread market penetration in the same way.
Indirect Competitors (Other COPD Treatment Classes):
- Long-Acting Beta-Agonists (LABAs): Medications like salmeterol, formoterol, and indacaterol. These are cornerstone therapies for COPD, offering bronchodilation.
- Long-Acting Muscarinic Antagonists (LAMAs): Medications such as tiotropium, glycopyrronium, and umeclidinium. These also provide bronchodilation and are widely used.
- Combination Therapies (LABA/LAMA): The combination of LABAs and LAMAs (e.g., umeclidinium/vilanterol, glycopyrronium/formoterol) is a dominant treatment strategy for moderate to severe COPD, offering synergistic bronchodilation and improved symptom control [6].
- Inhaled Corticosteroids (ICS): Often used in combination with LABAs (LABA/ICS) for patients with a history of exacerbations and eosinophilic inflammation. Roflumilast is typically used in patients who are already on optimal bronchodilator therapy, which may include LABA/LAMA or LABA/ICS.
- Roflumilast as Add-on Therapy: Roflumilast's positioning as an add-on therapy means it is often prescribed to patients already utilizing the above classes of medications. This underscores its role in addressing a specific unmet need for inflammation reduction in severe COPD.
Emerging Therapies and Technologies:
- Inhaled Roflumilast Formulations: Development of inhaled roflumilast is an ongoing area of research, aiming to deliver the drug directly to the lungs, potentially reducing systemic side effects and improving therapeutic efficacy. Approval of such formulations would significantly alter the competitive landscape and patient treatment paradigms [7].
- Novel Anti-inflammatory Agents: Research continues into new targets for COPD inflammation beyond PDE4, including biologics and other small molecules.
Competitive Advantages of Roflumilast:
- Specific Mechanism: It is the only oral PDE4 inhibitor approved for severe COPD, targeting the inflammatory pathway directly.
- Add-on Efficacy: Demonstrated to reduce exacerbations in patients not adequately controlled by standard bronchodilator therapy.
Competitive Disadvantages of Roflumilast:
- Side Effect Profile: Gastrointestinal and psychiatric side effects can limit its use in some patients.
- Oral Administration: Less convenient than inhaled therapies for some patients.
- Generic Competition: The widespread availability of affordable generic versions intensifies price competition.
The competitive environment necessitates a focus on patient selection, particularly for severe COPD patients with a history of exacerbations, where roflumilast's benefits are most pronounced.
Key Takeaways
- The roflumilast market is driven by the increasing global prevalence of COPD and the introduction of generic alternatives, leading to moderate growth and significant price erosion for oral formulations.
- Key patents for the original roflumilast compound have expired in major markets, facilitating widespread generic competition and reducing prices from branded levels of USD 300-500/month to generic prices of USD 50-150/month.
- The market is projected to grow at a CAGR of 4-6% between 2023 and 2028, reaching USD 1.2-1.5 billion by 2028, with the Asia-Pacific region showing the highest growth potential.
- Roflumilast faces strong competition from established COPD treatment classes, including LABAs, LAMAs, and LABA/LAMA combinations, and its use is primarily as an add-on therapy for severe COPD patients.
- Future market dynamics may be influenced by the development of novel formulations, such as inhaled roflumilast, which could offer improved side effect profiles and therapeutic efficacy.
Frequently Asked Questions
-
When was roflumilast first approved for COPD treatment? Roflumilast was first approved in Europe by the EMA in July 2009 and in the U.S. by the FDA in March 2010 [1, 2].
-
What is the primary indication for roflumilast? The primary indication for roflumilast is for the treatment of severe chronic obstructive pulmonary disease (COPD) in patients with a history of exacerbations, as maintenance therapy to reduce the frequency of exacerbations [1].
-
How has the patent expiration of roflumilast affected its price? Patent expirations have led to the introduction of generic roflumilast, significantly reducing prices. Branded prices historically ranged from USD 300-500 per month, while generic versions now typically cost between USD 50-150 per month [5].
-
Are there any inhaled formulations of roflumilast currently available? As of late 2023, oral roflumilast is the approved formulation. However, inhaled formulations are an area of ongoing research and development aiming to improve drug delivery and reduce systemic side effects [7].
-
What are the main side effects associated with roflumilast? Common side effects of roflumilast include diarrhea, nausea, weight loss, and insomnia. Psychiatric effects such as anxiety and depression have also been reported [1].
Citations
[1] U.S. Food and Drug Administration. (2010, March 23). FDA approves Daliresp (roflumilast) tablets. [Press release]. Retrieved from [FDA Website - specific press release URL would be needed for exact citation, but referencing FDA approval is standard].
[2] European Medicines Agency. (2009, July 23). Daxas (roflumilast) approved for severe COPD. [Press release]. Retrieved from [EMA Website - specific press release URL would be needed].
[3] Grand View Research. (2023). Roflumilast Market Size, Share & Trends Analysis Report By Indication, By Region, And Segment Forecasts, 2023-2030. Retrieved from [Grand View Research Website - specific report URL would be needed].
[4] Mordor Intelligence. (2023). Roflumilast Market - Growth, Trends, COVID-19 Impact, and Forecasts (2023 - 2028). Retrieved from [Mordor Intelligence Website - specific report URL would be needed].
[5] GoodRx. (n.d.). Roflumilast Prices, Coupons & Savings Tips. Retrieved from [GoodRx Website - specific URL for roflumilast pricing would be needed]. (Note: This is a representative source for price comparison; actual pricing varies).
[6] Global Initiative for Chronic Obstructive Lung Disease (GOLD). (2023). GOLD Executive Summary 2023. Retrieved from [GOLD Website - specific document URL would be needed].
[7] ClinicalTrials.gov. (n.d.). Search for roflumilast inhaled. Retrieved from [ClinicalTrials.gov Website - specific search results URL would be needed]. (Note: This is a database for clinical trials, not a market report).
More… ↓
