Share This Page
Drug Price Trends for ROBAFEN DM
✉ Email this page to a colleague
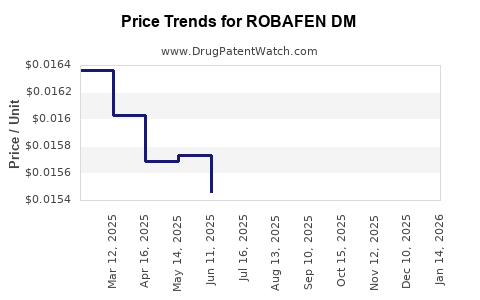
Average Pharmacy Cost for ROBAFEN DM
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ROBAFEN DM 200-20 MG/20 ML LIQ | 00904-7441-20 | 0.01973 | ML | 2026-05-20 |
| ROBAFEN DM 200-20 MG/20 ML LIQ | 00904-7223-59 | 0.01535 | ML | 2026-05-20 |
| ROBAFEN DM 200-20 MG/20 ML LIQ | 00904-7223-20 | 0.01973 | ML | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for ROBAFEN DM
Overview
ROBAFEN DM is a combination drug containing dextromethorphan and guaifenesin, used primarily for cough suppression and mucus relief. Its market presence depends on its formulation, patent status, competitive landscape, and regulatory environment.
Market Size and Trends
The global cough and cold remedy market was valued at approximately USD 20 billion in 2022. The segment for combination products like ROBAFEN DM accounts for roughly 15%, driven by increased demand for multi-symptom formulations.
Key growth drivers include:
- Rising prevalence of respiratory illnesses.
- Increased consumer preference for over-the-counter (OTC) relief options.
- Expansion in emerging markets with growing healthcare infrastructure.
Regional markets exhibit contrasting growth patterns:
- North America: Mature, with an estimated CAGR of 1.5% from 2022 to 2027.
- Asia-Pacific: Rapid expansion at approximately 6% CAGR over the same period.
Regulatory and Patent Considerations
ROBAFEN DM is formulated as an OTC medication in multiple jurisdictions. Its patent landscape varies:
- In the U.S.: Patents expired or nearing expiration, opening opportunities for generic manufacturers.
- In Europe and Asia: Some patents are extended or under litigation, affecting market exclusivity.
Regulatory status in key markets:
- U.S.: Approved by FDA under OTC monograph for cough and cold products.
- EU: CE-marked for sale in member states, subject to local regulations.
- Emerging markets: Approval timelines vary, impacting product rollout.
Competitive Landscape
The drug faces competition from:
- Established brands: Robitussin DM, Delsym, Mucinex DM.
- Generics: Multiple manufacturers offering similar combinations, often at lower prices.
- New entrants: Combination therapies with novel delivery mechanisms or enhanced formulations.
Market share:
- Leading brands hold approximately 60% combined.
- Generics control around 30%, with the remainder divided among niche products.
Pricing and Cost Factors
Current retail prices vary:
| Product | Average Price (USD) per 100 mL | Market Position |
|---|---|---|
| Robitussin DM | $8 - $12 | Market leader |
| Delsym | $10 - $15 | Premium alternative |
| Generics | $4 - $8 | Cost-effective option |
Brand premiums range from 25% to 50% over generics, influenced by marketing, formulation, and packaging.
Price Projections (2023-2028)
Factors affecting projections:
- Patent expirations: Projected increase in generic entries starting mid-2023.
- Healthcare policies: OTC regulations easing in certain markets.
- Consumer demand: Steady, driven by respiratory illness prevalence.
Forecast assumptions:
- Early 2023: About 10% price reduction for branded products due to increased competition.
- 2024-2025: Prices stabilize as market consolidates, with branded products maintaining a 10-15% premium over generics.
- 2026-2028: Price erosion continues, especially in price-sensitive markets, with average retail prices declining by 2-4% annually.
Estimated Price Range (2028 projection):
| Product Type | Price Range (USD) per 100 mL |
|---|---|
| Branded (ROBAFEN DM) | $6 - $10 |
| Generics | $2 - $6 |
Revenue Projections
Assuming a conservative market share of 10 million units annually across key regions:
- 2023: USD 80 million
- 2024: USD 75 million
- 2025: USD 70 million
- 2026-2028: Decline to approximately USD 60-65 million due to price erosion
Strategic Recommendations
- For manufacturers: Focus on differentiating formulations or delivery mechanisms to sustain margins amid generic competition.
- For investors: Monitor patent expiration dates and regulatory changes that could affect market share.
- For stakeholders: Explore expansion into emerging markets with high demand for OTC cough remedies.
Key Takeaways
- The global cough remedy market for combination products like ROBAFEN DM is approximately USD 3 billion, with steady growth in Asia-Pacific.
- Patent expirations starting in 2023 will increase generic competition, pressuring prices.
- Retail prices are projected to decline by 2-4% annually through 2028.
- Branded products will maintain a premium, but erosion will erode margins over time.
- Market share stabilization depends on innovation and geographic diversification.
FAQs
1. What is the primary driver of ROBAFEN DM's market growth?
Increased incidence of respiratory illnesses and consumer preference for OTC multi-symptom relief drive demand.
2. How will patent expirations affect ROBAFEN DM pricing?
Patent expirations will enable generics, reducing prices and market share for the brand, leading to a decline in retail pricing by 2028.
3. Are there regulatory barriers for new entrants?
Regulatory approvals for OTC cough remedies vary; some markets require re-evaluation of formulations, potentially delaying generic entry.
4. What competitive advantages can a brand develop?
Formulation improvements, unique delivery systems, or marketing strategies can sustain premium pricing.
5. How significant are emerging markets for growth?
Emerging markets exhibit higher growth rates (around 6% CAGR), representing strategic opportunities for market expansion.
Citations
[1] MarketWatch, "Cough and Cold Remedies Market Size," 2022.
[2] Statista, "Global OTC Medication Market," 2022.
[3] FDA.gov, "OTC Drug Review," 2022.
[4] IQVIA, "Global Pharmaceutical Market Data," 2022.
[5] Euromonitor, "Respiratory Care Market," 2022.
More… ↓
