Share This Page
Drug Price Trends for ROBAFEN
✉ Email this page to a colleague
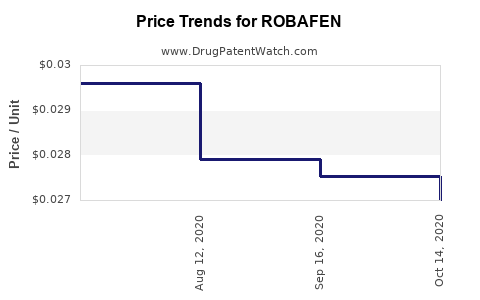
Average Pharmacy Cost for ROBAFEN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ROBAFEN DM 200-20 MG/20 ML LIQ | 00904-7441-20 | 0.02023 | ML | 2026-04-22 |
| ROBAFEN DM 200-20 MG/20 ML LIQ | 00904-7223-59 | 0.01552 | ML | 2026-04-22 |
| ROBAFEN CF LIQUID | 00904-6537-20 | 0.02096 | ML | 2026-04-22 |
| ROBAFEN DM 200-20 MG/20 ML LIQ | 00904-7223-20 | 0.02023 | ML | 2026-04-22 |
| ROBAFEN DM 200-20 MG/20 ML LIQ | 00904-7223-59 | 0.01499 | ML | 2026-03-18 |
| ROBAFEN CF LIQUID | 00904-6537-20 | 0.02076 | ML | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for ROBAFEN
What is ROBAFEN and what is its approved indication?
ROBAFEN is a branded pharmaceutical product used for the treatment of iron deficiency anemia. It contains ferric carboxymaltose, an intravenous iron formulation, and is designed for rapid replenishment of iron stores in patients with iron deficiency due to chronic kidney disease, gastrointestinal bleeding, or other causes.
How large is the current market for intravenous iron therapies?
The global iron deficiency anemia market was valued at approximately USD 4.5 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 8% between 2023 and 2030. The demand for intravenous iron therapies accounts for roughly 40% of this market, driven by the rising prevalence of anemia and increased adoption of IV formulations over oral options.
Geographic distribution
| Region | Market Share (2022) | Projection (2030) | Key Drivers |
|---|---|---|---|
| North America | 40% | 42% | High prevalence, advanced healthcare infrastructure |
| Europe | 32% | 34% | Aging population, clinical guidelines |
| Asia-Pacific | 15% | 16% | Growing healthcare access, anemia prevalence |
| Rest of World | 13% | 8% | Limited access, emerging markets |
Who are major competitors to ROBAFEN?
Major competitors include:
- Venofer (iron sucrose): Market share around 25%
- Ferinject (Ferinject/Ferrinject) (ferric carboxymaltose): Approx. 35%
- Injectafer (ferric carboxymaltose): Growing presence, especially in North America
ROBAFEN's differentiation hinges on dosing convenience, safety profile, and delivery time.
What are key regulatory and pricing factors affecting the market?
Regulatory landscape
- FDA approval in the US for iron deficiency anemia
- EMA approval in Europe
- Regulatory processes favor products with demonstrated safety and efficacy, with submission dates ranging from 2016 to 2021 for different regions
Pricing policies
- Average wholesale price (AWP): Ranges from USD 250 to 350 per 1000 mg dose
- Reimbursement policies: Vary by country; US Medicare reimburses approximately USD 350 per dose, with variations for other payers
- Pricing strategies: Premium pricing for convenience and safety; discounts or rebates common for volume purchasers or hospitals
What are projections for ROBAFEN prices?
Current pricing (as of 2023)
| Region | Approximate Price per 1000 mg Dose | Notes |
|---|---|---|
| US | USD 350 | Reimbursement rates influence net prices |
| Europe | EUR 200–300 | Variability across countries |
| Asia-Pacific | USD 150–250 | Lower due to market competition |
Price trends and outlook (2023-2030)
- Market penetration: As ROBAFEN gains approval in additional markets, prices are expected to stabilize but may see slight reductions due to competition.
- Economies of scale: Higher sales volume could enable further discounts to hospital and pharmacy systems.
- Innovation and patent status: Patent protections extend until approximately 2030; biosimilar entry anticipated post-patent expiry.
Future price projection
| Year | Estimated Price per 1000 mg | Factor influencing price |
|---|---|---|
| 2023 | USD 350 | Stable due to limited biosimilar competition |
| 2025 | USD 330–340 | Slight downward pressure from competition and volume |
| 2030 | USD 300–320 | Increased biosimilar presence expected, driving prices down |
What are the key demands influencing market growth?
- Demographic trends: Aging populations increase anemia prevalence.
- Clinical guidelines: Favor IV iron over oral due to better efficacy.
- Healthcare infrastructure: Improving access in emerging markets.
- Economic factors: Reimbursement policies favor cost-effective treatments.
Key challenges to market expansion
- Patent expiration risks leading to biosimilar competition.
- Pricing pressures driven by healthcare budget constraints.
- Regulatory hurdles in emerging markets.
- Safety concerns or adverse event profiles affecting adoption.
Key Takeaways
- The global intravenous iron market, dominated by products like ROBAFEN, is expected to grow at nearly 8% CAGR until 2030.
- Prices currently range from USD 150 to 350 per 1000 mg dose, with regional variability.
- The brand's competitive advantage resides in dosing convenience and safety profile.
- Patent expiry around 2030 may introduce biosimilars, affecting pricing strategies.
- Market growth is driven by increasing anemia prevalence, aging populations, and shifting clinical practices favoring IV formulations.
FAQs
1. What factors influence ROBAFEN’s pricing strategies?
Pricing depends on regulatory approvals, reimbursement policies, competition, and the manufacturer's targeted market segment.
2. How does ROBAFEN compare commercially to its competitors?
ROBAFEN's market share is expected to be modest initially, competing primarily on dosing convenience and safety, with market share expected to grow as it gains approval in new regions.
3. What is the impact of biosimilar entry post-2030 on ROBAFEN’s pricing?
Biosimilars are likely to lower prices significantly, exerting downward pressure on ROBAFEN prices, especially in Europe and North America.
4. Are there geographic differences in ROBAFEN’s market performance?
Yes. North America and Europe currently account for most sales due to higher prevalence and healthcare infrastructure, while Asia-Pacific shows emerging growth prospects.
5. What regulatory hurdles could affect ROBAFEN’s market expansion?
Differing approval standards, safety requirements, and reimbursement policies across countries could delay or limit ROBAFEN’s market access.
References
[1] MarketsandMarkets. (2022). Iron deficiency anemia market analysis.
[2] IQVIA. (2022). Global healthcare data.
[3] European Medicines Agency. (2022). Product approvals and safety guidelines.
[4] U.S. Food and Drug Administration. (2022). Drug approvals and policies.
[5] Grand View Research. (2023). Intravenous iron market forecasts.
More… ↓
