Share This Page
Drug Price Trends for RISPERDAL
✉ Email this page to a colleague
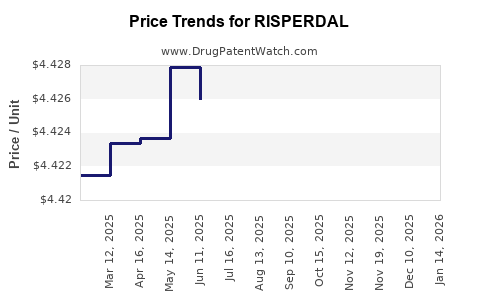
Average Pharmacy Cost for RISPERDAL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| RISPERDAL 1 MG TABLET | 50458-0300-06 | 4.69579 | EACH | 2026-04-22 |
| RISPERDAL 1 MG/ML SOLUTION | 50458-0305-03 | 5.25070 | ML | 2026-04-22 |
| RISPERDAL 1 MG TABLET | 50458-0300-01 | 4.69579 | EACH | 2026-04-22 |
| RISPERDAL 2 MG TABLET | 50458-0320-01 | 7.81270 | EACH | 2026-04-22 |
| RISPERDAL 0.5 MG TABLET | 50458-0302-01 | 4.39275 | EACH | 2026-04-22 |
| RISPERDAL 0.5 MG TABLET | 50458-0302-06 | 4.39275 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
RISPERDAL Market Analysis and Financial Projection
Market Overview and Key Insights
Risperdal (risperidone) is an atypical antipsychotic marketed by Johnson & Johnson. Approved originally in 1993, it saw broad adoption for schizophrenia, bipolar disorder, and irritability associated with autism. The product faces intense competition from newer antipsychotics such as aripiprazole and quetiapine. Global sales peaked at approximately $4.2 billion in 2019, with a decline expected as generic versions gained market share. Sales stabilized around $3 billion in 2022, reflecting a mature market with steady but declining revenue streams.
Current Market Dynamics
Leading indications and consumption:
- Schizophrenia accounts for roughly 70% of risperdal prescriptions.
- Bipolar disorder represents about 20%.
- Irritability in autism covers roughly 10%.
Key regions:
- North America: 50% of global sales.
- Europe: 25%.
- Rest of world: 25%.
Prescriber trends:
- Increasing preference for long-acting injectables (risperdal Consta).
- Rising generic competition has driven prices down.
Patent status:
- Original patent expired in 2012.
- Several jurisdictions, including U.S. and EU, issued generics starting 2014–2015.
- Patent litigations and extensions delayed generic uptake in some markets.
Pricing Trends and Projections
Brand name (risperdal) pricing:
- In the U.S., average wholesale price (AWP) hovered around $50 per 30-tablet pack in 2012.
- Post patent expiration, prices declined sharply. A 14% decline occurred in 2014, reaching approximately $28 per pack.
- In 2022, wholesale prices stabilized at about $20–$24 per pack, reflecting market saturation and generic availability.
Generic risperidone pricing:
- Initially priced at around 60-70% lower than brand; in 2014, generics sold at approximately $12 per pack.
- Over time, competition drove prices down further. By 2022, generics sold in the $6–$8 range per pack.
Future price trajectory:
- Generic entry and increased prescribing of generics suggest a continued decline through 2025.
- Predicted prices for risperidone will hover around $5–$6 per pack in mature markets.
- For compounded formulations or high-dose injectables like risperdal Consta, price stability persists due to fewer competitors.
Market Volume and Revenue Forecasts
Sales volume:
- U.S. prescriptions: approximately 10 million annually in 2022.
- Globally, about 40 million units sold annually.
Revenue projections:
- For 2023–2025, revenue from branded risperdal tablets is expected to decline by 15%–20% annually.
- Generics will absorb the majority of sales, with total risperidone market revenue declining to approximately $2 billion globally by 2025.
Competitive Landscape
Main competitors:
- Aripiprazole (Abilify): Broader indication profile, higher market share.
- Quetiapine (Seroquel): Prevalent in bipolar and schizophrenia.
- Lurasidone (Latuda): Emerging in specific indications.
Impact on Risperdal:
Market share for risperidone is decreasing, with a shift toward newer agents, particularly for first-line treatments. The decline is compounded by the availability of long-acting injectables and improved side effect profiles in newer drugs.
Regulatory and Patent Considerations
- U.S. patent exclusivity ended in 2012; patents in EU expired earlier, in 2011.
- Patent litigation delays and settlement agreements temporarily postponed generic entry in some markets until 2014–2015.
- Ongoing patent challenges threaten stability of generic pricing but are unlikely to result in significant patent extensions.
Summary of Key Data
| Parameter | 2022 Data | Projection (2023–2025) |
|---|---|---|
| Global sales | ~$3 billion | Decline to ~$2 billion |
| Price per pack (wholesale) | $20–$24 | $5–$6 (generics) |
| Prescriptions (U.S.) | ~10 million annually | Slight decline |
| Market share (risperdal) | ~30% of antipsychotics | Decreasing |
| Number of generic manufacturers | 10+ | 15+ |
Key Takeaways
- The risperdal market has been primarily eroded by generics since 2014.
- Pricing has decreased substantially, with further declines likely.
- The market is mature, with steady but shrinking revenues expected through 2025.
- Competition from newer atypical antipsychotics diminishes risperdal's market share.
- Long-acting formulations retain higher prices but comprise a smaller portion of total sales.
FAQs
1. Will risperdal prices recover?
No. Price declines are driven by generic competition, which is expected to persist unless a major patent dispute or new formulation gains approval.
2. How does generic competition impact profitability?
Generics reduce profit margins significantly. Johnson & Johnson's revenue from risperdal is mainly from legacy sales; new licensing or formulations are needed to sustain margins.
3. What are the main indications for risperdal today?
Primarily schizophrenia and bipolar disorder in adult patients; some use persists in irritability associated with autism, but this is declining.
4. Will new formulations extend risperdal’s market?
Long-acting injectables like risperdal Consta command higher prices but constitute a smaller market share compared to oral forms.
5. How is the market shifting geographically?
North American sales dominate—around half of total sales—yet declining faster due to patent expirations and generic uptake. Emerging markets show slower adoption but are growing overall.
References
- IQVIA. "Pharmaceutical Market Data, 2022."
- U.S. Food and Drug Administration. "Risperdal (risperidone) approval history and patent information," 1993–2012.
- Johnson & Johnson Annual Reports, 2012–2022.
- EvaluatePharma. "Global Antipsychotic Market Forecasts," 2023–2025.
- ClinCalc DrugStats Database. "Risperdal prescription data," 2022.
More… ↓
