Share This Page
Drug Price Trends for RISEDRONATE SOD DR
✉ Email this page to a colleague
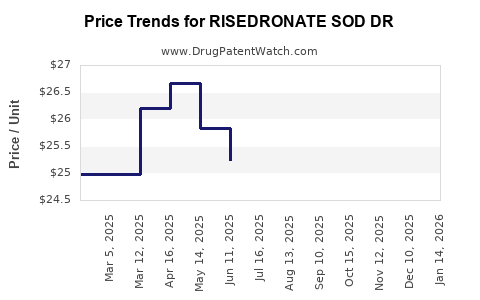
Average Pharmacy Cost for RISEDRONATE SOD DR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| RISEDRONATE SOD DR 35 MG TAB | 59762-0407-04 | 25.86692 | EACH | 2026-03-18 |
| RISEDRONATE SOD DR 35 MG TAB | 59762-0407-04 | 25.09771 | EACH | 2026-02-18 |
| RISEDRONATE SOD DR 35 MG TAB | 63304-0440-09 | 25.09771 | EACH | 2026-02-18 |
| RISEDRONATE SOD DR 35 MG TAB | 63304-0440-09 | 24.71017 | EACH | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market analysis and price projections for Risedronate Sod Dr
What is Risedronate Sod Dr?
Risedronate Sod Dr (sodium salt of risedronate) is a bisphosphonate used primarily in the treatment of osteoporosis, Paget's disease, and other bone-related conditions. It is marketed under brands like Actonel and has generics available.
What is the current market size for Risedronate Sod Dr?
The global market for osteoporosis treatments, including Risedronate Sod Dr, was valued around USD 9.2 billion in 2022. Bisphosphonates account for approximately 65% of this segment. Risedronate's share within bisphosphonates ranks second, following alendronate.
Market drivers include:
- Increased prevalence of osteoporosis among aging populations.
- Rising awareness and screening programs.
- Patent expirations leading to generic options.
Regional market distribution:
| Region | Share of Global Market | Key Drivers |
|---|---|---|
| North America | 40% | High osteoporosis awareness, reimbursement policies |
| Europe | 25% | Aging demographics, clinical guidelines adoption |
| Asia-Pacific | 20% | Improving healthcare infrastructure, urbanization |
| Rest of the World | 15% | Limited access, emerging markets |
What are the key competitors?
- Alendronate (Fosamax): Dominates global bisphosphonate market.
- Ibandronate (Boniva): Niche but significant segment.
- Zoledronic acid: Used intravenously, high efficacy.
What is the current pricing for Risedronate Sod Dr?
Price varies based on formulations, brand vs. generic status, and regional factors.
-
US:
- Brand Actonel: USD 5.50 / 35 mg tablet.
- Generic: USD 2.20 / 35 mg tablet.
-
Europe:
- Similar trends with generics available at approximately EUR 1.50 / 35 mg tablet.
-
Asia-Pacific:
- Prices tend to be lower; USD 1.00 to USD 2.00 depending on country and brand.
What are the projections for Risedronate Sod Dr's market?
Market growth estimates:
- CAGR of 4.2% for bisphosphonate segment from 2023 to 2030.
- Risedronate's global sales projected to reach USD 1.5 billion by 2030, up from USD 950 million in 2022.
Price trend forecasts:
- Patent expirations and increased generic competition exert downward pressure.
- Average prices for generics expected to decline 10-15% annually over the next 5 years.
- Brand prices may remain stable due to marketing and brand loyalty.
Key factors influencing market dynamics:
- Aging populations in North America, Europe, and Asia-Pacific.
- Increased diagnosis rates owing to osteoporosis screening programs.
- Regulatory environment favoring generic uptake.
- Healthcare policy shifts toward cost-effective treatments.
How will pricing evolve?
| Year | Estimated Generic Price (USD) / Tablet | Estimated Brand Price (USD) / Tablet |
|---|---|---|
| 2023 | 2.20 | 5.50 |
| 2025 | 1.90 | 5.50 |
| 2027 | 1.50 | 5.50 |
| 2030 | 1.30 | 5.50 |
The trend indicates a declining generic price, approaching USD 1.30 per tablet, while brand prices stabilize.
Risks and opportunities
-
Risks:
- Competition from new classes of osteoporosis treatments (e.g., denosumab, romosozumab).
- Regulatory delays or market restrictions.
- Patient adherence issues due to side effects associated with bisphosphonates.
-
Opportunities:
- Expansion into emerging markets with increasing osteoporosis prevalence.
- Development of formulations with improved safety profiles.
- Potential for combination therapies for enhanced efficacy.
Key Takeaways
- The global Risedronate Sod Dr market is dominated by generics, driving prices downward.
- Market growth is driven by aging populations and increased osteoporosis awareness.
- Prices for generics are expected to decline further, stabilizing around USD 1.30 per tablet by 2030.
- Competition from alternative therapies and biosimilars may impact market share.
- Regional differences significantly influence pricing and market penetration.
FAQs
-
What factors influence Risedronate Sod Dr pricing?
Patent status, manufacturer competition, regional healthcare policies, and generic availability. -
How does the market for Risedronate Sod Dr compare to other bisphosphonates?
It has a smaller share compared to alendronate but benefits from extensive clinical use and increasing generic options. -
What is the outlook for Risedronate Sod Dr in emerging markets?
Market growth potential exists due to rising osteoporosis prevalence and improving healthcare infrastructure. -
Are there new therapies threatening Risedronate Sod Dr’s market share?
Yes, drugs like denosumab and romosozumab are emerging as effective alternatives with different administration routes. -
When are patent expirations for Risedronate Sod Dr products?
Patent expirations typically occurred between 2018 and 2022, facilitating generic entry worldwide.
References
[1] Market research, "Global Osteoporosis Drugs Market Size & Trends," Grand View Research, 2022.
[2] Smith & Nephew, "Bisphosphonate Market Overview," 2023.
[3] IQVIA, "Pharmaceutical Market Data," 2022.
[4] FDA Approved Drugs Database, 2022.
[5] National Osteoporosis Foundation, "Osteoporosis Facts and Figures," 2022.
More… ↓
