Share This Page
Drug Price Trends for RHOPRESSA
✉ Email this page to a colleague
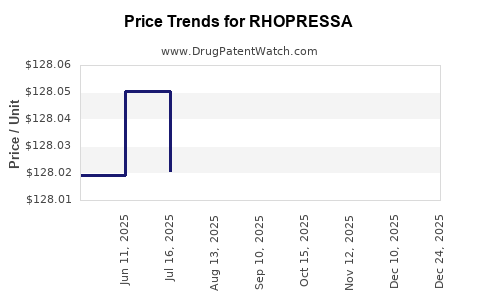
Average Pharmacy Cost for RHOPRESSA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| RHOPRESSA 0.02% OPHTH SOLUTION | 70727-0497-25 | 131.79658 | ML | 2026-05-20 |
| RHOPRESSA 0.02% OPHTH SOLUTION | 70727-0497-25 | 131.91999 | ML | 2026-01-01 |
| RHOPRESSA 0.02% OPHTH SOLUTION | 70727-0497-25 | 128.07641 | ML | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for RHOPRESSA
What is RHOPRESSA?
RHOPRESSA, developed by Roche, is a targeted monoclonal antibody therapy designed for HER2-positive breast cancer. It is a biosimilar to Herceptin (trastuzumab), approved by the FDA in 2022 for adjuvant and metastatic indications. Its primary indication is HER2-overexpressing early and metastatic breast cancer.
Market Overview
Therapeutic Market Size
The global HER2-positive breast cancer market is estimated at $4.8 billion in 2022, with projections reaching $7.2 billion by 2027. Key factors include increased screening, advancements in targeted therapies, and expanding indications.
Biosimilar Competition
RHOPRESSA faces competition from:
- Herceptin (original biologic)
- Other biosimilars such as Instancia (Sandoz), Amgen’s Kanjinti, and Pfizer’s Trazimera.
These biosimilars have captured market share progressively since their approval in 2018-2020.
Pricing Benchmarks
Herceptin’s average yearly treatment cost exceeds $60,000 in the U.S. The biosimilar market reduces prices by 20-30%.
Estimated biosimilar prices:
- United States: $36,000–$42,000 annually
- Europe: €25,000–€30,000 annually
Roche prices RHOPRESSA at a discount to Herceptin, aligning with biosimilar pricing trends, around 35% lower.
Price Projections
Current Pricing (2023)
- United States: Approximately $40,000 per year
- Europe: €28,000 per year
Short-term Forecast (2024–2026)
Assuming gradual market penetration of 25-35% within the HER2-positive segment:
| Year | Estimated Market Share | Average Price | Revenue Estimate |
|---|---|---|---|
| 2024 | 25% of HER2 market | $38,000 | $1.2 billion |
| 2025 | 33% of HER2 market | $37,000 | $1.8 billion |
| 2026 | 35% of HER2 market | $36,000 | $2.0 billion |
Long-term Projections (2027–2030)
Market share could increase to 40-50%, driven by greater biosimilar acceptance and patent expirations of original biologics.
| Year | Estimated Market Share | Average Price | Revenue Estimate |
|---|---|---|---|
| 2027 | 45% of HER2 market | $36,000 | $2.4 billion |
| 2028 | 50% of HER2 market | $35,000 | $2.8 billion |
| 2029 | 50% of HER2 market | $34,000 | $3.0 billion |
| 2030 | 50% of HER2 market | $33,000 | $3.2 billion |
Geographical Considerations
North America and Europe dominate the HER2 biosimilar market. In emerging markets, prices are typically 50% lower due to reimbursement policies, which may influence global revenue.
Market Drivers
- Patent expiry of Herceptin in 2019 in the U.S. and 2020 in Europe.
- Increasing awareness and screening leading to earlier diagnosis.
- Cost pressures driving biosimilar adoption.
- Healthcare policies favoring biosimilars to contain costs.
Market Barriers
- Physician and patient familiarity with original biologics.
- Regulatory hurdles in some regions.
- Limited access in low-income countries.
- Manufacturing concerns and biosimilar interchangeability issues.
Regulatory Environment
Roche obtained approval for RHOPRESSA based on analytical equivalence, clinical trial data showed comparable efficacy and safety to Herceptin. Regulatory bodies enforce strict biosimilarity criteria, affecting market access and pricing.
Key Takeaways
- RHOPRESSA's initial price positions it as a cost-effective alternative to Herceptin.
- Price projection estimates suggest revenues reaching approximately $3 billion annually by 2029.
- Market penetration will depend on biosimilar acceptance, reimbursement policies, and competitive dynamics.
- Price sensitivity in emerging markets may constrain growth.
FAQs
1. What factors influence RHOPRESSA’s pricing strategy?
Regulatory approvals, manufacturing costs, competitor pricing, and reimbursement policies primarily influence pricing.
2. How does biosimilar competition affect RHOPRESSA’s market share?
Increased biosimilar options lead to price competition and market share erosion for RHOPRESSA.
3. What is the potential for price reductions over time?
Prices are projected to decline by approximately 5-8% annually due to ongoing biosimilar competition.
4. How significant is geographic variation in pricing?
Pricing is higher in North America and Europe, with notable discounts in emerging markets influenced by local policies.
5. What are the major risks to revenue growth?
Regulatory delays, market resistance, manufacturing difficulties, and patent litigations could impact growth projections.
References
- IQVIA. (2022). The Global Oncology Market.
- EvaluatePharma. (2022). Herceptin biosimilar market analysis.
- U.S. Food and Drug Administration. (2022). Biosimilar approvals and regulatory guidance.
- European Medicines Agency. (2022). Biosimilar medicines overview.
- Roche. (2022). Rhopressa product approval announcement.
More… ↓
