Drug Price Trends for RECTIV
✉ Email this page to a colleague
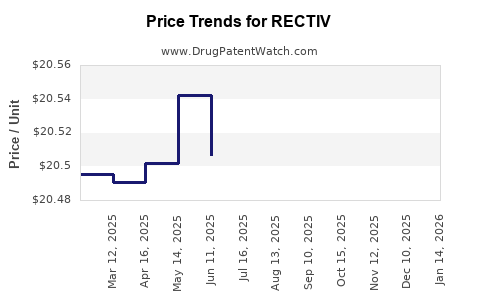
Average Pharmacy Cost for RECTIV
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| RECTIV 0.4% OINTMENT | 58914-0301-80 | 20.57028 | GM | 2026-04-22 |
| RECTIV 0.4% OINTMENT | 58914-0301-80 | 20.57028 | GM | 2026-03-18 |
| RECTIV 0.4% OINTMENT | 58914-0301-80 | 20.55322 | GM | 2026-02-18 |
| RECTIV 0.4% OINTMENT | 58914-0301-80 | 20.58660 | GM | 2026-01-21 |
| RECTIV 0.4% OINTMENT | 58914-0301-80 | 20.50161 | GM | 2025-12-17 |
| RECTIV 0.4% OINTMENT | 58914-0301-80 | 20.51257 | GM | 2025-11-19 |
| RECTIV 0.4% OINTMENT | 58914-0301-80 | 20.50448 | GM | 2025-10-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
