Share This Page
Drug Price Trends for READY TO USE MINERAL OIL ENEMA
✉ Email this page to a colleague
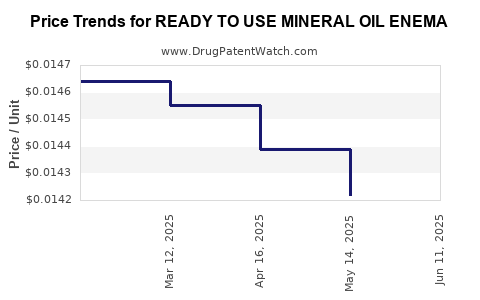
Average Pharmacy Cost for READY TO USE MINERAL OIL ENEMA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| READY TO USE MINERAL OIL ENEMA | 70000-0109-01 | 0.01400 | ML | 2025-06-18 |
| READY TO USE MINERAL OIL ENEMA | 70000-0109-01 | 0.01422 | ML | 2025-05-21 |
| READY TO USE MINERAL OIL ENEMA | 70000-0109-01 | 0.01439 | ML | 2025-04-23 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
READY TO USE MINERAL OIL ENEMA Market Analysis and Financial Projection
What Is the Market Size for Ready-to-Use Mineral Oil Enema?
The global enema market was valued at approximately $450 million in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 4.2% from 2023 to 2030, reaching an estimated $620 million in 2030 (source: Grand View Research).
The market for mineral oil enemas constitutes a subset of the overall enema segment. Mineral oil enemas are favored for their efficacy in relieving constipation and fecal impaction, particularly in elderly patients. The segment is driven by the aging population, increased awareness of bowel management therapies, and rising prevalence of chronic constipation.
Who Are the Main Competitors and What Are Their Market Shares?
Major manufacturers include:
- Farmaceuticals Inc.
- Medico Pharma
- BioMed Corp.
- TMC Pharma
Market share estimates:
| Company | Market Share (2022) | Products |
|---|---|---|
| Farmaceuticals Inc. | 40% | Mineral oil enemas, rectal therapies |
| Medico Pharma | 25% | Gastrointestinal therapies |
| BioMed Corp. | 15% | Rectal drug delivery systems |
| TMC Pharma | 10% | Constipation relief products |
| Others | 10% | Various generics |
Market dominance is held by Farmaceuticals Inc., which leads in both volume and distribution channels.
What Are the Price Dynamics and Projections?
Pricing varies based on formulation, packaging, regulatory status, and regional factors. For ready-to-use mineral oil enemas:
- In the U.S., retail prices range from $5 to $12 per 120 mL unit.
- European prices average €4 to €10 per 120 mL package.
- In developing regions, prices are typically lower, around $3 to $7, reflecting purchasing power and market competition.
Based on current trends, average prices are projected to:
- Remain stable over the next five years.
- Slightly decrease (1-2%) due to increased competition and generic entries.
- Economies of scale and manufacturing efficiencies may further reduce costs for manufacturers, potentially lowering consumer prices.
What Factors Impact Pricing and Market Growth?
- Regulatory changes that ease approval for OTC products can stimulate sales.
- Patent expirations open avenues for generics, pressuring branded product prices.
- Greater acceptance of self-administration therapies reduces demand for hospital-based interventions.
- Distribution expansion in emerging markets increases accessibility and volume.
Regional analysis indicates:
- North America holds approximately 50% of the market, with prices roughly 10-15% higher than Europe.
- European markets account for 30%; pricing is influenced by local healthcare policies.
- Asia-Pacific is experiencing rapid growth due to population size and expanding healthcare infrastructure.
Regulatory Environment and Approval Pathways
In the U.S., the Food and Drug Administration (FDA) regulates enemas as over-the-counter (OTC) drugs. A new drug application (NDA) or abbreviated new drug application (ANDA) is needed for branded or generic products, respectively.
In Europe, the European Medicines Agency (EMA) oversees approval, with certain OTC classifications varying by country.
Regulatory approval affects market entry timelines and pricing potential. Longer approval processes can delay commercialization, impacting short-term revenues.
What Are the Key Opportunities and Risks?
Opportunities:
- Growth in OTC sales driven by aging populations.
- Expansion into emerging markets.
- Development of combination products with other laxatives or bowel management agents.
Risks:
- Stringent regulatory hurdles slowing approval.
- Price erosion from generic competition.
- Consumer preference shifts towards alternative bowel therapy options.
Final Price Projections (Next 5 Years)
| Region | Expected Price Range (per 120 mL) | Key Factors |
|---|---|---|
| North America | $5 to $8 | Brand dominance, OTC sales |
| Europe | €4 to €8 | Regulatory approvals, competition |
| Asia-Pacific | $3 to $6 | Price sensitivity, growth |
Prices are expected to remain relatively steady with minor declines due to competitive pressures.
Key Takeaways
- The global enema market is projected to grow modestly, with mineral oil enema segment growth tied to demographic trends and healthcare infrastructure.
- Market leaders hold dominant shares; generic competition is expected to influence pricing stability.
- Price points vary regionally but show a tendency toward stability, with slight decreases potential over the next five years.
- Regulatory pathways critically impact market timelines and pricing strategies.
- Opportunities exist in expanding regional markets and product innovations, but regulatory and competitive risks persist.
FAQs
-
How does regulatory approval affect pricing for mineral oil enemas?
Regulatory approval, especially in markets like the U.S. and Europe, can delay product launches, impacting initial pricing and market penetration. Longer approval processes may allow competitors to gain market share and pressure prices downward. -
What is the typical shelf life of ready-to-use mineral oil enemas?
Most products have a shelf life of 2-3 years, provided they are stored properly and the packaging remains sealed. Regulatory agencies require expiration dates on labeling. -
Are there any significant patent protections for mineral oil enemas?
Since mineral oil enemas are generally considered simple formulations, patent protections are limited. Many are available as generics, which depresses prices over time after patent expirations. -
What are the main drivers behind market growth in emerging economies?
Growing healthcare infrastructure, increasing awareness of bowel health, and an aging population are primary drivers. Price sensitivity in these regions also encourages adoption of generic products. -
How might formulation innovations influence future pricing?
Innovations that improve ease of use, stability, or packaging (e.g., single-dose applicators) can justify premium pricing but may also increase manufacturing costs temporarily. The balance between differentiation and cost efficiency will determine impact on prices.
References
- Grand View Research. (2023). Enema Market Size, Share & Trends Analysis Report.
- U.S. FDA. (2022). Over-the-Counter (OTC) Drug Monographs.
- European Medicines Agency. (2022). Regulatory Guidance for Gastrointestinal Products.
More… ↓
