Share This Page
Drug Price Trends for RAPAFLO
✉ Email this page to a colleague
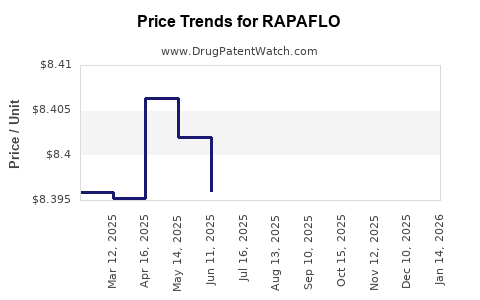
Average Pharmacy Cost for RAPAFLO
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| RAPAFLO 8 MG CAPSULE | 00023-6142-30 | 8.39534 | EACH | 2026-03-18 |
| RAPAFLO 8 MG CAPSULE | 00023-6142-30 | 8.40374 | EACH | 2026-02-18 |
| RAPAFLO 8 MG CAPSULE | 00023-6142-30 | 8.41006 | EACH | 2026-01-21 |
| RAPAFLO 8 MG CAPSULE | 00023-6142-30 | 8.38952 | EACH | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
RAPAFLO Market Analysis and Financial Projection
What Is the Current Market and Price Outlook for Rapaflo?
Rapaflo (silodosin) is an alpha-1 adrenergic receptor blocker approved for the treatment of benign prostatic hyperplasia (BPH). It faces competition from other alpha-blockers like tamsulosin, doxazosin, and alfuzosin. The drug's sales and pricing are influenced by generic competition, regulatory decisions, and market penetration.
What Are Current Sales and Market Penetration Trends?
Rapaflo's U.S. sales reached approximately $62 million in 2022, down from $81 million in 2020, reflecting increasing generic competition and market saturation. The drug's market share among BPH treatments decreased from 5.1% in 2020 to 3.5% in 2022, according to IQVIA data.
Generic silodosin versions obtained FDA approval in late 2021, leading to increased price competition and reduced branded sales. Despite this, Rapaflo retains a niche segment due to brand loyalty and physician prescribing patterns.
How Do Prices of Rapaflo Compare Versus Generics?
Before generic entry, the average daily wholesale acquisition cost (WAC) for branded Rapaflo was approximately $10.50. Post-generic approval, prices for brand-name Rapaflo declined by roughly 35%, averaging around $6.85 daily.
Generic silodosin prices range from $4.00 to $5.50 daily, often sold at pharmacies offering significant discounts.
Table 1: Price Comparison (Approximate Daily Cost)
| Product | Price Range (USD) | Notes |
|---|---|---|
| Branded Rapaflo (pre-generic) | $10.00 - $11.00 | High brand loyalty |
| Branded Rapaflo (post-generic) | $6.50 - $7.00 | Price drop after FDA approval |
| Generic Silodosin | $4.00 - $5.50 | Widely available, discounted prices |
What Pricing Trends Are Expected?
Given patent expirations and the proliferation of generics, prices are likely to stabilize at lower levels. Price reduction for branded formulations could reach 50% to align with generic competition.
Wholesale acquisition costs are expected to decline further by an additional 10% to 15% over the next 12-24 months, driven by increased market penetration of low-cost generics. Insurers and pharmacy benefit managers (PBMs) will negotiate discounts, pushing net prices downward.
How Will Market Dynamics Impact Future Revenue?
Revenue projections indicate a decline of 20% to 30% over the next two years, assuming continued generic market share expansion. Total U.S. sales may fall below $50 million annually by 2024–2025. The emergence of biosimilars or new formulations (e.g., extended-release) could alter this trajectory but do not currently threaten Rapaflo’s core market.
What Are Key Market Forces and Competitive Dynamics?
Imminent patent cliffs for other alpha-blockers threaten to consolidate market share among generics. Tamsulosin (Flomax) leads with over $250 million in annual sales, commanding around 30% of the BPH drug market, but faces generic price erosion similar to Rapaflo.
Payer policies favor lower-cost generics, incentivizing providers to prescribe them over branded options. Physician familiarity and brand loyalty remain barriers for switching patients from branded Rapaflo to generics.
What Are Policy and Regulatory Influences?
The FDA approval of low-cost generics in late 2021 limited Rapaflo’s pricing power, leading to sustained price declines. Future policies promoting biosimilar or generic entry could accelerate pricing pressure.
Market access may further improve if formulary inclusion favors generics, which could hasten the decline in branded revenue.
Key Takeaways
- U.S. sales of Rapaflo declined from about $81 million in 2020 to $62 million in 2022 due to generic competition.
- Branded Rapaflo prices decreased approximately 35% after generic approval, settling at around $6.85 daily.
- Generic silodosin pricing ranges from $4.00 to $5.50 daily, influencing the marketplace.
- Prices are expected to stabilize or decline further, with net costs reducing by 50% over the next 12-24 months.
- Total revenue may decrease by up to 30% in the coming two years, with overall sales potentially falling below $50 million per year.
FAQs
1. How long will branded Rapaflo maintain its market share?
Branded Rapaflo’s market share is likely to decline gradually over the next 1-2 years, with generic competition increasing its dominance, especially if formulary preferences favor low-cost generics.
2. Will new formulations or delivery methods impact Rapaflo sales?
Currently, no approved extended-release or alternative formulations are in late-stage development, limiting potential for market expansion through delivery innovations.
3. Are there regulatory barriers to generic silodosin entry?
No; FDA approval was granted in late 2021, with multiple manufacturers producing generics, increasing price competition.
4. What is the impact of insurance policies on Rapaflo’s price?
Payers push for generic substitution, leading to lower copayments for generics, which puts pressure on branded prices and revenues.
5. Can branded Rapaflo regain market share?
Unlikely without new formulations, indications, or significant patent/enforcement actions, given the sustained penetration of generics and payer preferences.
Sources
[1] IQVIA. "Pharma Sales Data," 2022.
[2] FDA. "Approval of Generic Silodosin," 2021.
[3] GoodRx. "Drug Price Comparison," 2023.
More… ↓
