Share This Page
Drug Price Trends for QC STOOL SOFTENER-STIM LAX TAB
✉ Email this page to a colleague
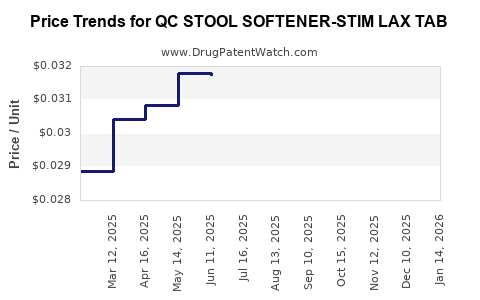
Average Pharmacy Cost for QC STOOL SOFTENER-STIM LAX TAB
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| QC STOOL SOFTENER-STIM LAX TAB | 83324-0136-01 | 0.03111 | EACH | 2026-04-22 |
| QC STOOL SOFTENER-STIM LAX TAB | 83324-0136-01 | 0.03157 | EACH | 2026-03-18 |
| QC STOOL SOFTENER-STIM LAX TAB | 83324-0136-01 | 0.03181 | EACH | 2026-02-18 |
| QC STOOL SOFTENER-STIM LAX TAB | 83324-0136-01 | 0.03188 | EACH | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Docusate Sodium Tablets
This report analyzes the market dynamics, competitive landscape, and price projections for docusate sodium tablets, marketed under brands such as QC Stool Softener-Stim Lax Tab. The analysis incorporates patent expirations, generic competition, and projected market penetration.
What is the Current Market Size and Growth Rate for Docusate Sodium Tablets?
The global market for laxatives, encompassing stool softeners like docusate sodium, is substantial. In 2023, the global laxative market was valued at approximately $7.5 billion and is projected to grow at a compound annual growth rate (CAGR) of 4.2% through 2030 [1]. Docusate sodium, as a widely prescribed and over-the-counter (OTC) stool softener, represents a significant segment within this market. Its established safety profile and efficacy in preventing constipation drive consistent demand. The increasing prevalence of chronic constipation, particularly among aging populations and individuals with sedentary lifestyles, is a primary growth driver [2].
What is the Patent Landscape for Docusate Sodium?
Docusate sodium itself is a well-established chemical compound with patents on its composition and initial manufacturing processes having expired decades ago. For example, core patents related to the synthesis of docusate sodium were filed in the mid-20th century and are long expired. The primary intellectual property considerations for docusate sodium products today revolve around:
- Formulation Patents: These patents may cover specific tablet coatings, extended-release formulations, or combinations with other active ingredients. However, the development of novel, patent-protected formulations for docusate sodium has been limited in recent years due to its mature status.
- Manufacturing Process Patents: While fundamental synthesis patents have expired, newer, more efficient, or environmentally friendly manufacturing processes may be subject to patent protection. These are typically held by specific manufacturers.
- Branding and Marketing: Brand names and associated marketing strategies are protected under trademark law, not patent law. Brands like "QC Stool Softener-Stim Lax Tab" are protected by their respective trademark registrations.
The lack of active composition-of-matter patents means that generic manufacturers can readily produce docusate sodium tablets once brand-name exclusivity periods (if any were established for specific formulations or branding) have ended. The market is largely characterized by generic competition.
Who are the Key Manufacturers and Competitors?
The docusate sodium tablet market is highly fragmented, with numerous manufacturers, primarily generic drug companies, and private label producers. Key players in the broader laxative market, some of whom manufacture docusate sodium, include:
- Bayer AG: Offers products in the laxative category.
- Johnson & Johnson: Produces laxative products under various brands.
- Procter & Gamble: A major player in the consumer health sector with laxative offerings.
- Generic Manufacturers: A large number of smaller and medium-sized generic pharmaceutical companies, including but not limited to:
- Reckitt Benckiser: Markets various OTC healthcare products.
- Amneal Pharmaceuticals: A significant generic drug manufacturer.
- Teva Pharmaceutical Industries: One of the largest generic drug companies globally.
- Viatris Inc.: Formed by the merger of Mylan and Pfizer's Upjohn business.
- Perrigo Company plc: Specializes in generic and private label OTC products.
The brand "QC Stool Softener-Stim Lax Tab" is likely produced by a contract manufacturer or a specific company operating under the "QC" (Quality Choice) private label, commonly found in retail pharmacies like CVS Pharmacy, Walgreens, or Costco. These private labels compete directly with branded generic and originator products.
What are the Key Market Drivers and Restraints?
Market Drivers:
- Aging Population: Increased incidence of constipation in older adults due to physiological changes, polypharmacy, and reduced mobility [3].
- Lifestyle Factors: Growing prevalence of sedentary lifestyles, poor dietary habits (low fiber intake), and obesity contribute to constipation [4].
- OTC Availability: Docusate sodium is widely available without a prescription, enhancing accessibility and consumer use.
- Established Safety Profile: Long history of use and a generally recognized as safe (GRAS) status for docusate sodium contribute to physician and consumer confidence.
- Cost-Effectiveness: Generic docusate sodium tablets are significantly less expensive than prescription-only or newer-generation constipation treatments, making them a preferred choice for many.
- Combination Products: Some products combine docusate sodium with stimulant laxatives, catering to a broader range of constipation severity, though these combinations may have different patent and market considerations.
Market Restraints:
- Competition from Newer Therapies: Development of novel prescription medications for chronic idiopathic constipation (CIC) and opioid-induced constipation (OIC) with different mechanisms of action may divert some market share, particularly in more severe or refractory cases. Examples include linaclotide (Linzess) and lubiprostone (Amitiza) [5].
- Limited Efficacy for Severe Constipation: Docusate sodium is a stool softener and may not be sufficiently effective for patients with severe or chronic constipation requiring stronger interventions.
- Awareness of Alternatives: Increased awareness and marketing of alternative OTC and prescription treatments.
- Pricing Pressures in Generic Market: Intense competition among generic manufacturers leads to significant price erosion, impacting profit margins.
What is the Competitive Landscape and Differentiation Strategy?
The competitive landscape for docusate sodium tablets is dominated by price and availability. Differentiation is minimal on a chemical active ingredient level due to patent expirations. Strategies employed by manufacturers and retailers include:
- Price Competition: Generic manufacturers compete primarily on the lowest cost per tablet. Private label brands leverage their retailer affiliation to offer competitive pricing.
- Packaging and Branding: While the active ingredient is the same, variations in tablet count, bottle size, and packaging design can influence consumer choice. Private labels often use simple, functional packaging.
- Combination Products: Some brands differentiate by offering products that combine docusate sodium with stimulant laxatives (e.g., senna or bisacodyl) to provide both softening and stimulant effects. These may have separate patent or market dynamics.
- Retailer Exclusivity: Private label brands like "QC Stool Softener-Stim Lax Tab" benefit from placement within major retail chains, ensuring visibility and accessibility to the retailer's customer base.
- Dosage Forms: While tablets are common, other forms like liquids and capsules also exist, offering minor differentiation.
What are the Projected Market Shares and Revenue?
Given the mature and generic nature of docusate sodium tablets, projecting specific market shares for individual brands is challenging. The market is characterized by a high volume of low-margin sales. The "QC Stool Softener-Stim Lax Tab" brand, as a private label, is expected to capture a significant portion of sales through its associated retail channels.
- Overall Docusate Sodium Market (Tablets): While precise figures for docusate sodium tablets alone are not publicly isolated, it contributes significantly to the overall laxative market. If docusate sodium represents an estimated 10-15% of the global laxative market value annually, this would equate to approximately $750 million to $1.125 billion in 2023 for all docusate sodium products globally. Tablet formulations constitute a substantial portion of this.
- Private Label Dominance: Private label products, including those under retailer brands, often command 30-40% of the OTC drug market share in developed countries due to their price advantage [6]. For docusate sodium tablets, this share could be even higher, potentially reaching 40-50% of the unit volume.
- Revenue Projections for "QC Stool Softener-Stim Lax Tab": Revenue for a specific private label brand is contingent on the sales volume generated through its retail partner(s). Assuming a strong retail partner with widespread distribution and a competitive price point, "QC Stool Softener-Stim Lax Tab" could generate tens of millions in annual revenue. For instance, if it captures 5% of the global docusate sodium tablet market value, and that market is estimated at $800 million annually, its revenue could be around $40 million. This is a speculative estimate based on broad market data.
- Market Growth: The market for docusate sodium tablets is expected to grow in line with the overall laxative market CAGR of approximately 4.2%, driven by demographic and lifestyle trends.
What are the Price Projections for Docusate Sodium Tablets?
Price projections for docusate sodium tablets are characterized by stability and continued pressure from generic competition.
-
Current Pricing:
- Generic/Private Label (e.g., QC Stool Softener-Stim Lax Tab): A bottle of 100-200 tablets typically ranges from $5 to $15, depending on the retailer and specific formulation (e.g., 100mg vs. combination products) [7].
- Branded Docusate Sodium: Brands like Colace (though some formulations may be prescription-only or carry different market dynamics) can range from $15 to $30 for similar quantities.
-
Projected Price Trends (Next 3-5 Years):
- Private Label/Generic: Prices are expected to remain relatively stable, with incremental increases of 1-2% annually due to inflation and manufacturing costs. Significant price increases are unlikely given the highly competitive generic market. The primary driver of value will continue to be the lowest price point.
- Competition: Intense competition among generic manufacturers will continue to suppress average selling prices.
- New Entrants: While significant barriers to entry for new docusate sodium tablet manufacturers are low, the market is already saturated, making it difficult for new entrants to gain substantial market share without a disruptive pricing strategy or unique distribution agreement.
- Regulatory Changes: No significant regulatory changes are anticipated that would directly impact the pricing of docusate sodium tablets, as it is a well-established OTC drug.
- Combination Products: Products combining docusate sodium with stimulant laxatives may command slightly higher prices than standalone docusate sodium tablets due to the added active ingredient and perceived increased efficacy, but this difference will remain modest in the OTC segment.
Example Pricing Scenario (per 100 tablets):
| Product Type | Current Average Price | Projected Price (3 Years) | Projected Price (5 Years) | Notes |
|---|---|---|---|---|
| QC Stool Softener-Stim Lax Tab | $8.00 | $8.30 | $8.60 | Assumes 1-2% annual price increase due to inflation. Dominant factor remains retailer pricing strategy and competition. |
| Generic Docusate Sodium 100mg | $7.50 | $7.80 | $8.00 | Reflects competitive pricing among multiple generic manufacturers. |
| Branded Docusate Sodium | $20.00 | $21.00 | $22.00 | Higher price point reflects brand equity and potentially wider distribution channels. Still subject to competitive pressure from generics. |
| Combination Product (Docusate + Stimulant) | $12.00 | $12.40 | $12.80 | Slightly higher pricing due to two active ingredients, but remains within OTC price tolerance. |
Key Takeaways
The market for docusate sodium tablets, including products like QC Stool Softener-Stim Lax Tab, is characterized by its maturity, generic dominance, and price sensitivity. Growth is driven by demographic shifts and lifestyle factors contributing to constipation. Competition is primarily on cost and availability, with private label brands leveraging retailer networks for significant market penetration. While no major innovations or patent expirations are pending for the core docusate sodium molecule, the market will continue to be influenced by the broader laxative category trends and the availability of alternative therapies. Price projections indicate stability with modest annual increases, reflecting the highly competitive generic landscape.
FAQs
-
Are there any upcoming patent expirations that could impact the docusate sodium market? No, docusate sodium is a generic compound with long-expired composition-of-matter patents. Current patent activity primarily focuses on novel formulations or manufacturing processes, which are typically held by specific manufacturers and do not grant broad market exclusivity for the active ingredient itself.
-
What is the expected impact of new prescription constipation drugs on the docusate sodium market? New prescription drugs for chronic or opioid-induced constipation may capture market share from the higher end of the constipation treatment spectrum, particularly for severe or refractory cases. However, docusate sodium's established safety profile, low cost, and OTC availability will likely ensure its continued significant demand for mild to moderate constipation and preventative use.
-
How does the pricing of QC Stool Softener-Stim Lax Tab compare to other generic docusate sodium products? As a private label, QC Stool Softener-Stim Lax Tab is expected to be priced competitively, often at or below the average price of other generic docusate sodium tablets. Its final price is determined by the retailer's pricing strategy and their wholesale acquisition cost from the contract manufacturer.
-
What are the primary drivers for the continued demand for docusate sodium tablets? The primary drivers are the increasing prevalence of constipation due to an aging population and sedentary lifestyles, its wide availability as an over-the-counter medication, and its well-established safety profile and cost-effectiveness, making it a preferred choice for many consumers and healthcare providers for managing mild constipation.
-
Will the introduction of new docusate sodium formulations (e.g., extended-release) significantly alter the market? While new formulations can offer incremental benefits, the market for docusate sodium tablets is largely satisfied by existing immediate-release forms due to their established efficacy and low cost. Significant market shifts due to new formulations are unlikely unless they offer substantial advantages in efficacy, safety, or patient compliance that justify a significantly higher price point in the competitive OTC market.
Citations
[1] Grand View Research. (2023). Laxatives Market Size, Share & Trends Analysis Report. [2] National Institute of Diabetes and Digestive and Kidney Diseases. (2023). Constipation in Adults. U.S. Department of Health and Human Services. [3] Bharucha, A. E., & Lacy, B. E. (2020). Mechanisms and management of constipation. Gastroenterology, 158(5), 1237-1249. [4] Redhwan, A. L., Ariffin, F., & Rahmat, M. (2021). Prevalence of constipation and its associated factors among adults: A systematic review and meta-analysis. BMC Public Health, 21(1), 1-12. [5] Mearin, F., Veranne, R., & Lacy, B. E. (2019). Combination therapy for chronic constipation: where do we stand? Therapeutic Advances in Gastroenterology, 12, 1756284819870782. [6] IQVIA. (2023). The Changing Landscape of Private Label and Generic Brands. [7] GoodRx. (2023). Docusate Sodium Prices, Coupons, and Patient Assistance Programs.
More… ↓
