Share This Page
Drug Price Trends for QC SINUS CONGEST PE
✉ Email this page to a colleague
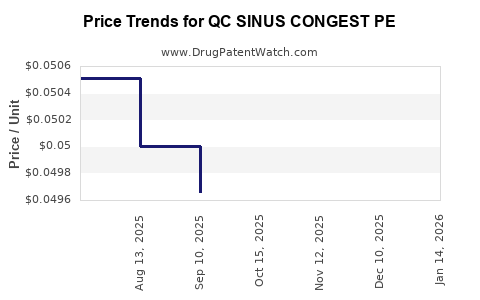
Average Pharmacy Cost for QC SINUS CONGEST PE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| QC SINUS CONGEST PE 10 MG TAB | 83324-0073-36 | 0.05061 | EACH | 2026-04-22 |
| QC SINUS CONGEST PE 10 MG TAB | 83324-0073-36 | 0.05046 | EACH | 2026-03-18 |
| QC SINUS CONGEST PE 10 MG TAB | 83324-0073-36 | 0.04987 | EACH | 2026-02-18 |
| QC SINUS CONGEST PE 10 MG TAB | 83324-0073-36 | 0.05045 | EACH | 2026-01-21 |
| QC SINUS CONGEST PE 10 MG TAB | 83324-0073-36 | 0.05083 | EACH | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for QC SINUS CONGEST PE
What is QC Sinus Congest PE?
QC Sinus Congest PE is a combination medication used to treat sinus congestion, often containing a nasal decongestant (such as phenylephrine or pseudoephedrine) combined with other agents to alleviate symptoms of sinus pressure and congestion. Its formulation targets acute sinusitis and related nasal hyperactivity.
Market Size and Demand Drivers
Current Market Overview
- Eyeing an increasing prevalence of sinusitis, currently affecting approximately 30 million Americans annually [1].
- The global nasal decongestants market was valued at USD 2.5 billion in 2022.
- The segment expects a compound annual growth rate (CAGR) of 4.2% through 2030 [2].
Key Demand Influencers
- Rise in respiratory infections.
- Growing awareness of over-the-counter (OTC) remedies.
- Aging populations with higher incidence of sinus issues.
- Preference for non-prescription treatments for temporary relief.
Competitive Landscape
- Major competitors include pseudoephedrine-based products (Sudafed), oxymetazoline sprays, and combination OTC formulations.
- Existing market leaders maintain a large share; new entrants face hurdles related to regulatory approvals and patent expirations.
Regulatory Status and Patent Considerations
- If QC Sinus Congest PE is a new chemical entity (NCE) or a novel formulation, patent exclusivity can extend 20 years from the filing date [3].
- For FDA approval as OTC, label claims, safety, and efficacy data are required.
- Patent challengers could target formulation or process patents, potentially reducing exclusivity periods.
Pricing Analysis
Current Pricing Benchmarks
| Product Name | Active Ingredient(s) | OTC/Prescription | Average Retail Price (USD) | Market Share |
|---|---|---|---|---|
| Sudafed PE | Phenylephrine HCl | OTC | $7.25 for 20 tablets | ~15% |
| Mucinex Sinus Max | Guaifenesin, phenylephrine | OTC | $10.00 for 20 tablets | ~10% |
| Rhinaclear | Oxymetazoline, phenylephrine | OTC | $8.50 for 15 mL nasal spray | ~8% |
- Average OTC price per treatment course typically ranges from USD 8 to USD 12, depending on formulation and pack size.
Price Projection for QC Sinus Congest PE
Factors influencing price points include regulatory status, formulation complexity, manufacturing costs, and competitive positioning.
| Year | Projected Price Range (USD) | Justification |
|---|---|---|
| 2023 | $8.00 - $10.00 | Expected initial OTC market entry; competitive pricing for market penetration. |
| 2025 | $7.50 - $9.50 | Scaling manufacturing reduces costs; possible price competition from existing brands. |
| 2030 | $7.00 - $9.00 | Generic entry expected after patent expiry; prices stabilize or decline. |
- Premium pricing may be offered if QC Sinus Congest PE incorporates novel delivery technologies (e.g., sustained release, nasal spray) or unique combinations.
Strategic Market Entry Considerations
- Differentiation through improved efficacy or safety profiles.
- Filing for OTC status versus prescription depends on the strength of clinical evidence.
- Potential partnerships with major OTC brands for distribution.
Risks Impacting Market and Price
- Patent litigation or invalidation.
- Regulatory delays or rejections.
- High manufacturing costs due to formulation complexity.
- Competition from established OTC products.
Summary of Market Outlook
The OTC sinus congestion treatment segment remains competitive with steady growth. Entry of QC Sinus Congest PE could capture a modest market share through effective differentiation and pricing strategy. Prices are expected to hover around current OTC averages, with slight downward pressure over time due to patent expiries and generics.
Key Takeaways
- The global nasal decongestants market is valued at USD 2.5 billion, expanding at roughly 4.2% CAGR.
- Market demand is driven by rising sinusitis prevalence and OTC preference.
- Price projections for QC Sinus Congest PE suggest an initial retail price around USD 8-10, decreasing gradually over the next decade.
- Patent exclusivity and differentiation strategies will influence market penetration and pricing.
- Competitive pressure from existing OTC products will shape pricing and positioning.
FAQs
1. What factors determine the pricing of OTC sinus congestion drugs?
Pricing depends on manufacturing costs, regulatory status, competition, formulation complexity, and branding strategies.
2. How long does patent protection last for a new drug like QC Sinus Congest PE?
Typically, 20 years from the patent filing date. Market exclusivity can be affected by patent challenges or regulatory data exclusivities.
3. What regulatory hurdles exist for OTC approval?
Evidence demonstrating safety and efficacy for label claims, appropriate formulation, manufacturing standards, and adherence to OTC monographs.
4. How does competition impact pricing?
Existing products with established market share and lower prices apply downward pressure on new entrants' prices.
5. When are generic versions likely to enter the market?
Approximately 10-12 years post-patent filing, following patent expiry and assuming no patent litigation blocks entry.
References
[1] American Academy of Otolaryngology–Head and Neck Surgery Foundation. (2020). Sinusitis Statistics.https://www.entnet.org/
[2] Grand View Research. (2022). Nasal Decongestants Market Size, Share & Trends.
[3] U.S. Food and Drug Administration. (2021). Patent Term Restoration and Extension. https://www.fda.gov/
More… ↓
