Share This Page
Drug Price Trends for QC NAPROXEN SOD
✉ Email this page to a colleague
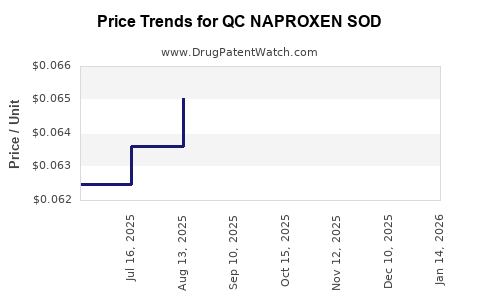
Average Pharmacy Cost for QC NAPROXEN SOD
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| QC NAPROXEN SOD 220 MG CAPLET | 83324-0108-50 | 0.06423 | EACH | 2026-03-18 |
| QC NAPROXEN SOD 220 MG TABLET | 83324-0109-50 | 0.06423 | EACH | 2026-03-18 |
| QC NAPROXEN SOD 220 MG CAPLET | 83324-0108-50 | 0.06520 | EACH | 2026-02-18 |
| QC NAPROXEN SOD 220 MG TABLET | 83324-0109-50 | 0.06520 | EACH | 2026-02-18 |
| QC NAPROXEN SOD 220 MG CAPLET | 83324-0108-50 | 0.06674 | EACH | 2026-01-21 |
| QC NAPROXEN SOD 220 MG TABLET | 83324-0109-50 | 0.06674 | EACH | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for QC Naproxen Sodium
Market Overview
Naproxen sodium is a nonsteroidal anti-inflammatory drug (NSAID) used to reduce pain, swelling, and fever. It is commonly prescribed for conditions such as arthritis, gout, and musculoskeletal injuries. The global NSAID market exceeds $5 billion annually, with naproxen sodium accounting for a significant share due to its efficacy and over-the-counter (OTC) status in many regions.
Market Dynamics
- Key Regions: North America and Europe dominate the NSAID market, driven by high healthcare expenditure and OTC sales. Asia-Pacific is an expanding market due to rising healthcare access and self-medication trends.
- Market Drivers: Increasing incidence of chronic inflammatory diseases, aging population, and demand for OTC pain relief products.
- Market Constraints: Safety concerns related to NSAID side effects, including gastrointestinal issues and cardiovascular risks, influence prescribing and OTC sale policies. Regulatory scrutiny impacts product formulation, labeling, and pricing.
- Competitive Landscape:
- Generic formulations of naproxen sodium hold a dominant position.
- Major brand competitors include Aleve (Bayer), Anaprox (Pfizer), and generic manufacturers.
- Patent expiries in 2010-2011 facilitated generic proliferation, leading to price erosion.
Legal and Regulatory Factors
- Regulations: US FDA classifies naproxen sodium as OTC for doses up to 220 mg per tablet, with prescriptions available for higher dosages or pediatric formulations.
- Patent Status: Key patents expired in 2010, contributing to market commoditization and price competition.
- Pricing Regulations: Importation and pricing policies vary across jurisdictions, influencing retail and wholesale pricing strategies.
Current Pricing Landscape
- In the US:
- OTC brands like Bayer’s Aleve are priced between $8-$12 for a 50-count bottle (220 mg strength).
- Generic versions are priced approximately 20-30% lower, typically $6-$10.
- In Europe:
- Retail prices vary but are comparable, with OTC naproxen sodium generally priced between €8-€15 per package.
- In Asia-Pacific:
- Prices are lower, commonly $3-$8, reflecting local manufacturing and regulatory environments.
Price Projections
Short-term (next 1-2 years)
- Generic Competition: Continued price stability or slight decreases due to ongoing generic competition.
- OTC Market Stability: No significant change expected in OTC prices unless new formulations or regulatory changes occur.
Medium-term (3-5 years)
- Market Consolidation: Entry of new OTC formulations or combination products could influence pricing.
- Regulatory Impact: Changes restricting OTC sales or prescribing could cause price increases for prescriptive formulations.
- Emerging Markets: Price declines as manufacturing costs decrease and regional competition intensifies.
Long-term (over 5 years)
- Innovation and Branding: Development of new formulations or delivery methods could command premium pricing.
- Regulatory Shifts: Tightening safety regulations might influence labeling requirements and consequently prices.
- Market Penetration: Expansion in emerging markets is likely to pressure prices downward, unless differentiated products capture market share.
Pricing Strategy Recommendations
- For manufacturers, maintaining competitive pricing in OTC channels requires cost efficiencies and brand differentiation.
- Entry into emerging markets necessitates localized pricing strategies aligned with regional income levels and regulatory constraints.
- Patent expiries highlight opportunities for cost leadership via generic manufacturing, requiring focus on supply chain optimization.
Summary
Naproxen sodium’s market is mature, with stable but competitive pricing structures primarily influenced by generic entries and regulatory regimes. Price stability is likely in the near term, with potential modest declines driven by regional competition. In the longer term, product differentiation and regulatory shifts may influence pricing dynamics.
Key Takeaways
- The global NSAID market surpasses $5 billion annually, with naproxen sodium a core product due to OTC availability.
- US OTC prices for naproxen sodium are generally between $8-$12 for a 50-count bottle, with generics priced lower.
- Patent expiries in 2010-2011 led to increased generic competition, exerting downward pressure on prices.
- Medium-term projections suggest prices will stabilize or decline slightly, with regional market factors influencing variations.
- Innovation and emerging market growth may support premium pricing opportunities in the long term.
FAQs
1. How does patent expiry affect naproxen sodium pricing?
Patent expiry facilitates generic entry, increasing competition and generally reducing prices.
2. What regulatory factors influence naproxen sodium pricing?
Regulations impose limits on OTC dosing, shape formulations, and influence distribution channels, all impacting retail and wholesale prices.
3. Are there notable regional differences in naproxen sodium prices?
Yes. North America has higher prices due to OTC sales and brand premiums. Emerging markets tend to have lower prices because of manufacturing costs and regulatory differences.
4. What market trends could impact price projections?
Growth in OTC channel sales, new product formulations, regulatory changes, and market penetration in Asia-Pacific impact future pricing.
5. How do generic competition and innovation influence long-term pricing?
Generic competition tends to decrease prices, while product innovation can command higher prices if it offers superior efficacy or convenience.
Citations
[1] MarketsandMarkets. NSAID Market Report 2022.
[2] US FDA. Over-the-Counter Pain Relief Drug Regulations.
[3] IBISWorld. Analgesic Drug Manufacturing Industry Report, 2022.
[4] Statista. NSAID Market Revenue and Price Trends.
[5] EvaluatePharma. Prescription and OTC Market Forecasts, 2022–2030.
More… ↓
