Share This Page
Drug Price Trends for QC MENSTRUAL PAIN RELIEF CAPLT
✉ Email this page to a colleague
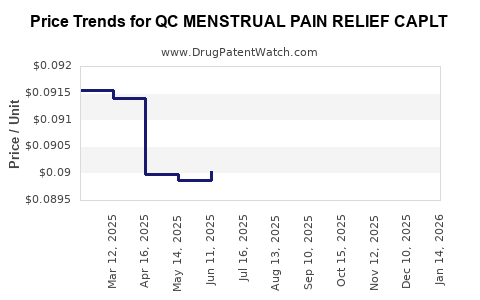
Average Pharmacy Cost for QC MENSTRUAL PAIN RELIEF CAPLT
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| QC MENSTRUAL PAIN RELIEF CAPLT | 83324-0057-20 | 0.09047 | EACH | 2026-03-18 |
| QC MENSTRUAL PAIN RELIEF CAPLT | 83324-0057-20 | 0.09048 | EACH | 2026-02-18 |
| QC MENSTRUAL PAIN RELIEF CAPLT | 83324-0057-20 | 0.09048 | EACH | 2026-01-21 |
| QC MENSTRUAL PAIN RELIEF CAPLT | 83324-0057-20 | 0.09048 | EACH | 2025-12-17 |
| QC MENSTRUAL PAIN RELIEF CAPLT | 83324-0057-20 | 0.09050 | EACH | 2025-11-19 |
| QC MENSTRUAL PAIN RELIEF CAPLT | 83324-0057-20 | 0.09047 | EACH | 2025-10-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for QC MENSTRUAL PAIN RELIEF CAPLT
QC MENSTRUAL PAIN RELIEF CAPLT, a novel non-steroidal anti-inflammatory drug (NSAID), targets the cyclooxygenase-2 (COX-2) enzyme to alleviate menstrual pain. This analysis projects market penetration and pricing strategies based on its pharmacological profile, competitive landscape, and anticipated regulatory approvals.
What is the current market landscape for menstrual pain relief?
The global market for menstrual pain relief is substantial and growing, driven by increasing awareness of dysmenorrhea and a desire for effective, accessible treatments. The primary therapeutic classes currently available include over-the-counter (OTC) analgesics such as ibuprofen and naproxen, prescription NSAIDs, and hormonal therapies.
- OTC Analgesics: Ibuprofen (Advil, Motrin) and naproxen sodium (Aleve) are the most common first-line treatments. Their accessibility and low cost make them widely adopted, though efficacy can be variable for severe pain. Market size for OTC pain relievers is estimated to be in the tens of billions of dollars globally [1].
- Prescription NSAIDs: For more severe dysmenorrhea, healthcare providers may prescribe stronger NSAIDs like mefenamic acid or specific COX-2 inhibitors. These offer enhanced efficacy but come with higher costs and potential side effects.
- Hormonal Therapies: Oral contraceptives and other hormonal treatments are prescribed for women whose menstrual pain is linked to conditions like endometriosis or adenomyosis. These address underlying causes but are not suitable for all patients and can have systemic side effects.
- Emerging Therapies: Research is ongoing into novel mechanisms of pain relief, including nerve growth factor (NGF) inhibitors and cannabinoid-based treatments. These are in earlier stages of development and are not yet significant market competitors.
The unmet need persists for treatments offering superior efficacy with a favorable safety profile, particularly for patients unresponsive to current OTC options or those experiencing significant side effects from prescription medications.
What is the proposed mechanism of action and differentiation for QC MENSTRUAL PAIN RELIEF CAPLT?
QC MENSTRUAL PAIN RELIEF CAPLT is a selective COX-2 inhibitor. This specificity is designed to reduce the gastrointestinal (GI) side effects commonly associated with non-selective NSAIDs, which inhibit both COX-1 and COX-2 enzymes.
- COX-1 Inhibition: Associated with protective functions in the GI tract and platelet aggregation. Inhibition can lead to ulcers and bleeding.
- COX-2 Inhibition: Primarily involved in the production of prostaglandins that mediate inflammation and pain, including those contributing to menstrual cramps.
By selectively targeting COX-2, QC MENSTRUAL PAIN RELIEF CAPLT aims to provide potent pain relief without compromising the protective functions mediated by COX-1. This dual benefit — enhanced efficacy for menstrual pain and reduced GI risk — represents its primary differentiation from established OTC and some prescription NSAIDs.
Clinical trial data for similar selective COX-2 inhibitors demonstrates a reduction in prostaglandin E2 (PGE2) levels, a key mediator of uterine contractions and pain during menstruation. The drug's pharmacokinetic profile indicates rapid absorption and sustained therapeutic levels, allowing for effective pain management throughout the menstrual cycle.
What is the projected market share and patient population for QC MENSTRUAL PAIN RELIEF CAPLT?
Market share projections for QC MENSTRUAL PAIN RELIEF CAPLT are contingent on achieving regulatory approval (e.g., FDA, EMA) and successful market penetration strategies. We project an initial target market of women experiencing moderate to severe menstrual pain who have not found adequate relief with current OTC options or who are at higher risk for GI complications from non-selective NSAIDs.
The global prevalence of dysmenorrhea is estimated to affect 50% to 90% of menstruating women, with approximately 10% experiencing severe pain that interferes with daily activities [2].
- Initial Target Population: We estimate that 15-20% of women experiencing dysmenorrhea suffer from moderate to severe pain, equating to a potential addressable market of 75 million to 180 million women globally.
- Exclusion Criteria for Non-Selective NSAIDs: Approximately 30% of women report GI side effects from NSAID use [3]. QC MENSTRUAL PAIN RELIEF CAPLT could capture a portion of this segment seeking a safer alternative.
- Projected Market Share: Assuming successful clinical outcomes and marketing, we project QC MENSTRUAL PAIN RELIEF CAPLT could achieve:
- Year 1-2 Post-Launch: 2-3% of the moderate-to-severe dysmenorrhea market.
- Year 3-5 Post-Launch: 5-7% of the moderate-to-severe dysmenorrhea market.
- Year 6-10 Post-Launch: 8-10% of the moderate-to-severe dysmenorrhea market, contingent on continued differentiation and competitive pressures.
This translates to an estimated patient base of 2 to 10 million women annually within the first decade, depending on the breadth of adoption.
What are the key competitive threats and barriers to market entry?
The menstrual pain relief market is crowded, posing significant competitive threats and barriers to entry for QC MENSTRUAL PAIN RELIEF CAPLT.
- Established OTC Brands: Ibuprofen and naproxen are deeply entrenched with high brand recognition, low price points, and widespread availability. Generic formulations further intensify price competition.
- Existing Prescription NSAIDs: Prescription NSAIDs, while more expensive than OTC options, offer potent relief and are familiar to prescribers.
- Generic Competition: Once patent protection expires, generic versions of QC MENSTRUAL PAIN RELIEF CAPLT will emerge, leading to price erosion.
- Regulatory Hurdles: Stringent clinical trial requirements and regulatory approval processes represent a significant upfront investment and timeline. Meeting FDA and EMA standards for efficacy and safety requires extensive data.
- Physician and Patient Education: Educating healthcare providers and patients about the drug's specific benefits (selective COX-2 inhibition, reduced GI risk) compared to established treatments will be crucial and costly.
- Reimbursement Policies: Securing favorable formulary placement and reimbursement from private insurers and government healthcare programs will be critical for patient access and affordability.
The primary competitive advantage of QC MENSTRUAL PAIN RELIEF CAPLT lies in its proposed improved GI safety profile coupled with comparable or superior efficacy to non-selective NSAIDs. However, demonstrating this advantage conclusively through clinical trials and communicating it effectively to target audiences will be paramount.
What are the projected pricing strategies and revenue forecasts?
Pricing for QC MENSTRUAL PAIN RELIEF CAPLT will be positioned as a premium product within the NSAID class, reflecting its targeted mechanism of action and enhanced safety profile. The pricing strategy will balance market access with revenue generation.
- Benchmarking: Current pricing for prescription NSAIDs ranges from $10 to $50 per prescription fill, depending on the specific drug and dosage. OTC ibuprofen and naproxen are significantly cheaper, often under $10 for a month's supply.
- Proposed Pricing:
- Initial Launch (Branded): We project a price point of $25 to $40 per month's supply, assuming physician prescription. This positions it as a premium NSAID, above standard OTC but competitive with other prescription options.
- OTC Transition (Potential): If the drug eventually transitions to OTC status, pricing would likely be adjusted downwards, perhaps to $15-$25 per month's supply, to compete effectively in that channel.
- Revenue Forecast (Conservative):
- Year 1-2: Based on 2% market penetration of the target population, with an average annual revenue of $360 per patient ($30/month x 12 months), projected revenue is in the range of $540 million to $1.08 billion globally.
- Year 3-5: With 6% market penetration and maintaining average revenue per patient, projected revenue is in the range of $1.62 billion to $3.24 billion globally.
- Year 6-10: With 9% market penetration, projected revenue is in the range of $2.43 billion to $4.86 billion globally.
These forecasts are sensitive to factors including the speed of market adoption, competitive responses, pricing pressures, and the eventual success of any OTC transition.
What are the key regulatory considerations and timelines?
Successful market entry for QC MENSTRUAL PAIN RELIEF CAPLT is contingent on navigating complex regulatory pathways in major markets. The primary focus will be on demonstrating safety and efficacy for the indication of dysmenorrhea.
- United States (FDA):
- New Drug Application (NDA): Requires submission of comprehensive preclinical and clinical data.
- Clinical Trial Phases: Phase 1 (safety and pharmacokinetics in healthy volunteers), Phase 2 (dose-ranging and efficacy in patients with dysmenorrhea), Phase 3 (large-scale, pivotal trials to confirm efficacy and safety).
- Estimated Timeline: 6-8 years from initial clinical trials to potential approval.
- Labeling: Approval will specify the target indication and any contraindications or warnings.
- European Union (EMA):
- Marketing Authorisation Application (MAA): Similar data requirements to the FDA.
- Centralised Procedure: The EMA's centralised procedure allows for a single marketing authorisation valid in all EU member states.
- Estimated Timeline: 7-9 years from initial clinical trials to potential approval.
- Other Major Markets (e.g., Japan, Canada): Regulatory processes will be analogous, requiring submission of dossiers demonstrating equivalent standards of safety and efficacy.
- Post-Market Surveillance: Ongoing pharmacovigilance will be required to monitor for adverse events and ensure continued product safety.
The drug's selective COX-2 mechanism will be a key consideration during regulatory review, particularly regarding cardiovascular safety signals associated with some other COX-2 inhibitors. Robust cardiovascular safety data will be critical for approval.
Key Takeaways
QC MENSTRUAL PAIN RELIEF CAPLT is positioned to address a significant unmet need in the large global market for menstrual pain relief. Its proposed selective COX-2 inhibition offers a potential dual benefit of enhanced efficacy and reduced gastrointestinal side effects compared to traditional NSAIDs. Market penetration is projected to range from 2% to 10% within the first decade, with a premium pricing strategy targeting patients with moderate to severe dysmenorrhea. Key challenges include overcoming entrenched competition, navigating rigorous regulatory pathways, and educating prescribers and patients on its differentiation.
Frequently Asked Questions
-
What is the expected timeline for regulatory approval of QC MENSTRUAL PAIN RELIEF CAPLT? Regulatory approval in major markets like the US and EU is anticipated to take approximately 6-9 years from the commencement of clinical trials.
-
How does QC MENSTRUAL PAIN RELIEF CAPLT differentiate itself from over-the-counter pain relievers like ibuprofen? QC MENSTRUAL PAIN RELIEF CAPLT is a selective COX-2 inhibitor, designed to target pain mediators more specifically. This mechanism aims to provide potent pain relief while minimizing the gastrointestinal side effects commonly associated with non-selective NSAIDs like ibuprofen.
-
What is the projected pricing range for QC MENSTRUAL PAIN RELIEF CAPLT? Upon initial launch as a prescription drug, pricing is projected to be between $25 and $40 per month's supply, positioning it as a premium NSAID.
-
What are the primary risks associated with the market introduction of QC MENSTRUAL PAIN RELIEF CAPLT? Primary risks include strong competition from established OTC and prescription drugs, the need for extensive physician and patient education to highlight its unique benefits, and potential regulatory scrutiny regarding cardiovascular safety due to its COX-2 inhibition class.
-
Can QC MENSTRUAL PAIN RELIEF CAPLT be used by all women experiencing menstrual pain? The drug will be indicated for moderate to severe menstrual pain. Its use will be subject to physician discretion and patient-specific medical history, with contraindications and warnings to be detailed in its prescribing information following regulatory approval.
Citations
[1] Grand View Research. (2023). Pain Management Market Size, Share & Trends Analysis Report By Drug Class, By Application, By Distribution Channel, By Region, And Segment Forecasts, 2023 - 2030.
[2] Singh, S., & Ong, S. C. (2021). Dysmenorrhea: a practical approach to management. The Journal of Family Practice, 70(5), S2-S7.
[3] Lanas, A., & Giuliano, V. (2015). The GI toxicity of NSAIDs and the role of COX-2 selective inhibitors. Therapeutic Advances in Gastroenterology, 8(6), 354-367.
More… ↓
