Share This Page
Drug Price Trends for QC LAXATIVE PEG
✉ Email this page to a colleague
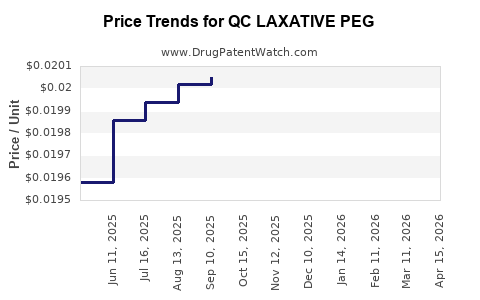
Average Pharmacy Cost for QC LAXATIVE PEG
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| QC LAXATIVE PEG 3350 POWDER | 83324-0002-16 | 0.02078 | GM | 2026-04-22 |
| QC LAXATIVE PEG 3350 POWDER | 83324-0002-08 | 0.02424 | GM | 2026-04-22 |
| QC LAXATIVE PEG 3350 POWDER | 83324-0002-16 | 0.02039 | GM | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
QC LAXATIVE PEG Market Analysis and Financial Projection
What Is the Market Size and Demand for QC Laxative PEG?
QC LAXATIVE PEG (Polyethylene Glycol) is a laxative used primarily for chronic constipation and bowel cleansing before procedures. The global polyethylene glycol market reached approximately USD 300 million in 2022, with a CAGR of about 6% projected through 2027. The market demand for PEG-based laxatives stems from increasing awareness of gastrointestinal health, aging populations, and rising prevalence of chronic constipation across developed and emerging markets.
Key regional drivers include:
- North America: Regulations favor over-the-counter (OTC) laxatives; high adoption rates.
- Europe: Aging demographics and healthcare systems support market growth.
- Asia-Pacific: Rising urbanization and income levels expand OTC product consumption.
How Does the Competitive Landscape Look?
Major players include:
- Johnson & Johnson (MiraLAX/Colace formulations)
- Braintree Laboratories (PEG3350)
- Rekabay (generic PEG products)
- Teva Pharmaceuticals (generic PEG formulations)
Market share concentrates among several top firms, with generics accounting for 65-75% of sales. Proprietary formulations or brand-specific versions like Miralax hold premium pricing in North America.
What Are the Pricing Trends for PEG Laxatives?
Average retail prices vary by region. In the U.S., a 238g bottle of OTC PEG laxative (like MiraLAX) retails for USD 15-20, with generics priced around USD 10-15. Cost per unit decreases when purchased in bulk or wholesale.
Factors influencing price:
- Brand versus generic status
- Packaging size
- Distribution channel (pharmacy, online, hospital supply)
- Patent status and exclusivity periods
In markets with less regulation or differing health insurance reimbursements, prices tend to be lower.
How Will Regulatory Policies Affect Pricing and Market Entry?
FDA approval and European Medicines Agency (EMA) regulations govern safety and efficacy standards. Pacification of governments toward OTC status reduces barriers to entry, facilitating competitive pricing.
Patent expirations—most PEG formulations are off-patent since 2010—allow generic manufacturers to enter the market, compress prices over time. New formulations optimized for faster action or better tolerability may command higher premiums temporarily but face eventual generic competition.
What Are Future Price and Market Growth Projections?
Market volume for PEG laxatives is expected to grow at 6% CAGR, reaching approximately USD 430 million by 2027, driven by:
- Expanded demand in emerging markets
- Aging demographics
- Increased acceptance of OTC bowel management products
Price stabilization is projected in mature markets, with slight declines anticipated due to generic competition. Premium formulations with added benefits (e.g., flavoring, quicker onset) may sustain higher prices temporarily.
What Are the Key Factors to Monitor?
- Patent expirations and generic entry timelines
- Regulatory changes impacting OTC classification
- Consumer health trends influencing demand
- Price sensitivity in different regions
- New product innovations that could redefine standards
Key Takeaways
- The PEG laxative market is approximately USD 300 million globally (2022), growing at 6% annually.
- Major brands like MiraLAX dominate in North America, with generics controlling significant share.
- Prices in North America range USD 10-20 per bottle; similar trends exist elsewhere, adjusted per market specifics.
- Patent expirations have led to increased generic competition, depressing prices over the past decade.
- Future growth hinges on emerging markets’ adoption and product innovations, while prices stabilize in mature markets.
FAQs
-
What factors influence PEG laxative pricing globally?
Pricing depends on brand status, packaging size, distribution, patent expiration, and regional regulatory environments. -
How do patent expirations impact market prices?
Patent expirations enable generic manufacturers to enter the market, increasing competition and driving prices downward. -
What regions present the most growth opportunities?
Asia-Pacific, Latin America, and the Middle East show rising demand due to demographic shifts and improving healthcare access. -
Are there regulatory barriers to new PEG laxative formulations?
Yes. Approval requires demonstration of safety and efficacy, with ongoing scrutiny over claims and manufacturing standards. -
What innovations might influence future prices?
Formulations offering faster relief, improved tolerability, or added features like flavoring could command premium pricing temporarily.
Sources
- MarketsandMarkets. "Polyethylene Glycol Market," 2023.
- IQVIA. "OTC gastrointestinal products market analysis," 2022.
- U.S. Food and Drug Administration. "Regulation of Laxatives," 2022.
- Grand View Research. "Gastrointestinal Therapeutics Market," 2022.
- Statista. "Market revenue for laxatives in North America," 2022.
More… ↓
