Share This Page
Drug Price Trends for QC IBUPROFEN
✉ Email this page to a colleague
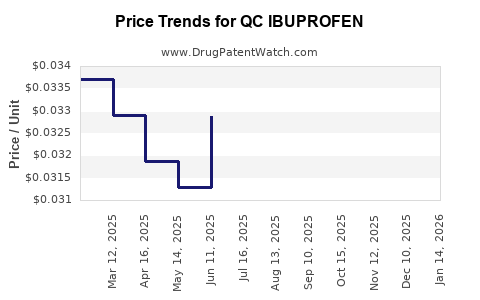
Average Pharmacy Cost for QC IBUPROFEN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| QC IBUPROFEN 200 MG SOFTGEL | 83324-0206-48 | 0.07958 | EACH | 2026-03-18 |
| QC IBUPROFEN 200 MG TABLET | 83324-0059-24 | 0.03282 | EACH | 2026-03-18 |
| QC IBUPROFEN 200 MG TABLET | 83324-0056-24 | 0.03282 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for QC Ibuprofen
Market Overview
QC Ibuprofen, a generic version of the widely used nonsteroidal anti-inflammatory drug (NSAID), belongs to a competitive segment dominated by established companies such as Johnson & Johnson, Teva, and Walgreens Boots Alliance. The global analgesics market, valued at approximately $9.2 billion in 2022, is expected to grow at a compound annual growth rate (CAGR) of 4.2% through 2030. A significant portion of this market is driven by OTC sales, where ibuprofen remains a core product due to its effectiveness, safety profile, and consumer familiarity.
Key Market Drivers:
- Increased prevalence of pain-related conditions
- Rising aging population
- Growing self-medication trends
- Expanding availability in emerging markets
Regulatory Environment
In the U.S., the Food and Drug Administration (FDA) classifies OTC ibuprofen as generally recognized as safe (GRAS). Manufacturers must comply with Good Manufacturing Practices (GMP), labeling requirements, and post-market surveillance. Patent expirations in major markets have facilitated market entry for generics, intensifying competition.
In other regions, regulatory systems vary: the European Medicines Agency (EMA) oversees approval in Europe, requiring bioequivalence data for generics. In countries with less regulated markets, pricing and distribution are less controlled but depending heavily on local health policies and import regulations.
Competitive Landscape
Pre patent expiry, the primary brands controlled the market. Post patent expiry, generics have entered rapidly, reaching market shares exceeding 90% in many regions. Key competitors include:
- Teva Pharmaceuticals
- Mylan (now part of Viatris)
- Walgreens Boots Alliance
- Local generic manufacturers in emerging markets
Pricing Trends
Pricing varies significantly based on geography, formulation, packaging, and brand status. In the U.S., typical retail prices for OTC ibuprofen (200 mg tablets) range from $0.02 to $0.08 per tablet, averaging around $0.04. Generic versions tend to be 15-30% cheaper than branded equivalents.
In European countries, prices are often regulated, with retail prices around €0.02–€0.05 per tablet. Bulk purchasing and healthcare system negotiations further impact pricing, especially in public health contexts.
Price Projections
Assumptions:
- QC Ibuprofen launches at a competitive price point, similar or marginally below current generic averages.
- Regulatory approvals and supply chain logistics are streamlined.
- Demand remains consistent with historical OTC consumption data.
- Competitive pressures persist, constraining price increases.
| Year | Price per Tablet (USD) | Notes |
|---|---|---|
| 2023 | $0.04 | Launch year, aligned with existing generics |
| 2024 | $0.04–$0.05 | Slight upward pressure due to inflationary costs |
| 2025 | $0.045–$0.055 | Market stabilization, potential volume growth |
| 2026 | $0.045–$0.06 | Market saturation, price competition persists |
Revenue Projections
Assuming initial annual sales volume of 2 billion tablets globally in the first year, with annual growth of 3-5% due to market penetration and brand recognition:
| Year | Units Sold (billions) | Average Price per Tablet (USD) | Estimated Revenue (USD billions) |
|---|---|---|---|
| 2023 | 2.0 | $0.04 | $80 million |
| 2024 | 2.06 | $0.045 | $92.7 million |
| 2025 | 2.16 | $0.05 | $108 million |
| 2026 | 2.27 | $0.055 | $125 million |
Note: Market share gains are critical to surpassing $100 million in annual revenue within 3 years, depending on geographic expansion and competition.
Market Entry Strategies
- Competitive pricing to secure initial market share.
- Establishing supply agreements with distributors and pharmacy chains.
- Emphasizing quality assurance, regulatory compliance, and OTC marketing.
Regulatory and Patent Impact
The absence of patent barriers and low regulatory hurdles in key markets favor rapid entry. However, patent litigation and regulatory delays can alter projected timelines and prices.
Summary
QC Ibuprofen is positioned within a saturated yet expanding OTC analgesics market. Pricing will likely mirror existing generic brands, with slight variability driven by regional factors. Revenue growth depends heavily on volume expansion, geographic reach, and competitive dynamics.
Key Takeaways
- Market is mature with high generic penetration.
- Pricing varies regionally; typical costs are $0.02–$0.08 per tablet.
- Launching at $0.04 per tablet aligns with existing generics.
- Revenue projections suggest potential for exceeding $100 million annually within three years, contingent on market penetration.
- Regulatory environment favors rapid entry due to generic status post-patent expiry in major markets.
FAQs
1. What are the main factors influencing ibuprofen prices globally?
Pricing is driven by manufacturing costs, regulatory approval requirements, market competition, and regional health policies. In developed markets, regulation keeps prices relatively stable, while in emerging markets, prices are more volatile.
2. How does patent status affect market entry?
The patent expiry of innovator brands allows generic manufacturers to enter the market freely. This results in immediate price reductions and increased competition, suppressing prices for several years.
3. What sales channels are most effective for OTC ibuprofen?
Pharmacy chains, supermarkets, convenience stores, and online retail platforms represent significant channels, with pharmacies often commanding higher margins and visibility.
4. How does regional regulation impact pricing and market share?
Stringent regulation can increase costs due to compliance and delays, whereas less regulated markets permit lower prices but present quality assurance risks.
5. What are the growth prospects for QC Ibuprofen over the next five years?
Growth hinges on effective market penetration strategies, expansion into emerging markets, and establishing brand recognition. Price stability and supply chain reliability will support incremental volume increases.
Sources
[1] Grand View Research, "Analgesics Market Size, Share & Trends," 2022.
[2] FDA, "OTC Drugs," 2022.
[3] European Medicines Agency, "Pharmacovigilance and Regulation," 2022.
[4] IBISWorld, "Over-the-Counter Drugs in the US," 2022.
[5] Statista, "Global Analgesics Market Forecast," 2022.
More… ↓
