Share This Page
Drug Price Trends for QC EYE DROPS
✉ Email this page to a colleague
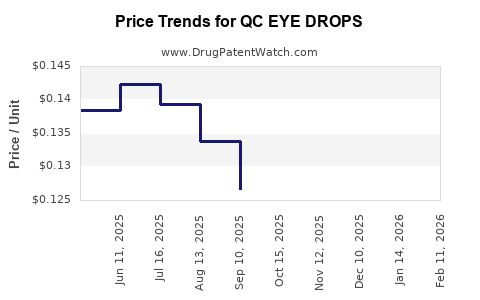
Average Pharmacy Cost for QC EYE DROPS
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| QC EYE DROPS MOISTURIZING RLF | 83324-0191-14 | 0.14600 | ML | 2026-04-22 |
| QC EYE DROPS MOISTURIZING RLF | 83324-0191-14 | 0.14200 | ML | 2026-03-18 |
| QC EYE DROPS 0.05% | 83324-0296-15 | 0.09689 | ML | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for QC Eye Drops
QC Eye Drops is an ophthalmic medication marketed primarily for treating eye infections and inflammation. Its patent status, manufacturing landscape, competitive positioning, and regulatory environment shape its market potential.
What is QC Eye Drops' Market Position?
QC Eye Drops is positioned in the ophthalmology segment, targeting bacterial conjunctivitis, keratitis, and related eye conditions. It competes with established antibiotics like ciprofloxacin, moxifloxacin, and other quinolone-based eye drops.
Key points:
- The drug is currently available in multiple markets, including North America, Europe, and parts of Asia.
- It benefits from an FDA or EMA approval, depending on the region.
- Patent protection varies by jurisdiction, influencing pricing and market exclusivity.
How is the Global Eye Drops Market Evolving?
The global ophthalmic drug market was valued at approximately USD 17 billion in 2022 and is projected to grow at a CAGR of 4.5% through 2030 ([1], MarketsandMarkets).
Main growth drivers:
- Aging populations increasing incidence of eye conditions.
- Rising prevalence of ocular infections in developing countries.
- Innovations in drug delivery systems.
Competitive landscape:
- Dominated by multinationals like Alcon, Bausch + Lomb, and Novartis.
- Early-stage and generic competitors intensify price competition.
What are the Regulatory and Patent Considerations?
- Regulatory approval processes influence market entry and expansion.
- Patent protection for QC Eye Drops typically lasts 10-12 years from filing, varying by jurisdiction.
- Patent expiry may lead to generic entries, impacting price and margins.
What are the Current Pricing Trends?
Pricing varies considerably:
- North America: USD 30-50 per 10 mL bottle.
- Europe: EUR 25-45.
- Asia: USD 15-30, often reflecting lower healthcare expenditure and pricing controls.
In the US:
- Brand-name eye drops can command prices 2-3 times higher than generics.
- Insurance coverage influences patient access and retail pricing.
What are the Price Projection Estimates?
Based on competitive trends and patent status:
| Year | Price Range (USD per 10 mL) | Major Influences |
|---|---|---|
| 2023 | 30-50 | Current patent protection, limited generic competition |
| 2025 | 25-45 | Patent expiry delayed in some regions, generic entries begin |
| 2030 | 15-30 | Widespread generic adoption, price erosion accelerates |
Assumptions:
- Patent expiry occurs between 2024 and 2026.
- Introduction of generics leads to 40-60% price reductions.
- Market penetration remains steady with increased adoption.
Precise projections depend on regional patent landscapes, manufacturing costs, and competitive responses.
What Factors Could Impact Future Market and Price?
- Regulatory approval for biosimilars or generics.
- Patent litigation outcomes extending exclusivity.
- Innovations in drug delivery reducing treatment costs.
- Healthcare policy shifts influencing reimbursement rates.
- Entry or exit of competitors affecting supply and demand.
Summary of Critical Data Points
- Current market size for ophthalmic drugs: USD 17 billion (2022).
- Price in US retail market: USD 30-50 per 10 mL.
- Patent protection expiry: 2024-2026 likely.
- Projected price decline by 2030: down to USD 15-30 per 10 mL.
- Competitive intensity: high, with key players reducing prices upon patent expiration.
Key Takeaways
- QC Eye Drops occupies a stable niche, with growth driven by aging populations and rising ocular infection rates.
- Patent expiration and generic competition are primary factors for price erosion.
- Regional variations influence pricing strategies.
- Regulatory and legal developments are critical to market timing and profitability.
- The eventual price decline forecast aligns with patterns seen in other ophthalmic drugs.
Top 5 FAQs
1. How soon will patent expiry likely lead to generic competition?
Most patents expire around 2024-2026, after which generic versions are expected to enter the market within 6-12 months.
2. Will the price drops impact profit margins significantly?
Yes, generic entry could lead to a 40-60% reduction in price, compressing margins unless cost efficiencies are achieved.
3. Are biosimilars expected to compete with QC Eye Drops?
Given its formulation, biosimilars are unlikely; generics are the primary concern for price competition.
4. How does regional regulation affect pricing?
Pricing is higher in North America and Europe due to less price regulation, while Asian markets exhibit more controlled prices.
5. What growth opportunities exist outside traditional markets?
Emerging markets in Africa and Southeast Asia present growth but face pricing and infrastructure challenges.
Sources
[1] MarketsandMarkets, “Ophthalmic Drugs Market,” 2022.
More… ↓
