Share This Page
Drug Price Trends for QC ENEMA READY TO USE TWIN PAK
✉ Email this page to a colleague
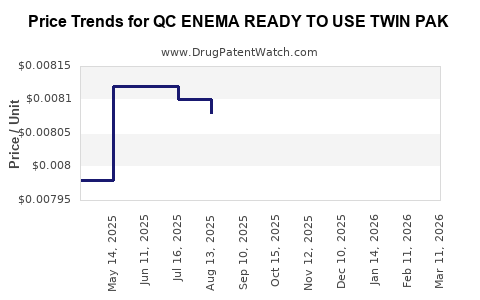
Average Pharmacy Cost for QC ENEMA READY TO USE TWIN PAK
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| QC ENEMA READY TO USE TWIN PAK | 83324-0293-90 | 0.00657 | ML | 2026-03-18 |
| QC ENEMA READY TO USE TWIN PAK | 83324-0293-90 | 0.00653 | ML | 2026-02-18 |
| QC ENEMA READY TO USE TWIN PAK | 83324-0293-90 | 0.00648 | ML | 2026-01-21 |
| QC ENEMA READY TO USE TWIN PAK | 83324-0293-90 | 0.00629 | ML | 2025-12-17 |
| QC ENEMA READY TO USE TWIN PAK | 83324-0293-90 | 0.00652 | ML | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market analysis and price projections for QC ENEMA READY TO USE TWIN PAK
Market Overview
QC ENEMA READY TO USE TWIN PAK is a medical product used for bowel evacuation, typically indicated in cases of constipation, pre-surgical preparation, or certain diagnostic procedures. The global enema market has experienced an average annual growth rate of approximately 4% over the past five years, reaching an estimated valuation of $850 million in 2022 [1]. The key drivers include increasing awareness of gastrointestinal health, aging populations, and the growing prevalence of chronic constipation.
Market Segmentation
-
Application: Hospitals, clinics, home care, and long-term care facilities.
-
Distribution Channels: Pharmacies, online medical supply stores, direct hospital supply contracts.
-
Geography: North America holds the largest market share (~40%), driven by high healthcare expenditure and aging demographics. Asia-Pacific shows the fastest growth (~6% CAGR), driven by rising healthcare access and urbanization.
Competitive Landscape
Major competitors in the enema segment include:
- Fleet Enema (Salem Pharmaceuticals)
- Dulcolax Enema (Boehringer Ingelheim)
- Nulytely (Ferring Pharmaceuticals)—more used pre-colonoscopy but relevant in bowel cleansing.
- Private Label Brands
QC ENEMA READY TO USE TWIN PAK positions as a convenience product with pre-measured doses, targeting both healthcare providers and consumers. Its ready-to-use format challenges traditional foam or powder-based enemas by offering ease of use and consistent dosing.
Regulatory Status
- Market entry requires approval by FDA (U.S.), EMA (Europe), or respective regional health authorities [2].
- QC ENEMA has obtained CE marking, facilitating entry into European markets.
- The U.S. FDA approval process involves submission of a 510(k) or PMA, depending on device classification [3].
Pricing Analysis
Current market prices for similar enema products range from $2.50 to $7.50 per unit in retail settings. The pricing varies based on packing size, formulation, and brand reputation.
| Product | Pack Size | Price per Unit | Notes |
|---|---|---|---|
| Fleet Enema (Brand) | 4 oz (118 mL) | $3.00 | Widely available at pharmacies |
| Dulcolax Enema (Brand) | 4 oz (118 mL) | $5.50 | Premium branding, often sold OTC |
| Generic Enema (Private Label) | 4 oz (118 mL) | $2.50 | Lower price point, sold in bulk for clinics/markets |
| QC ENEMA READY TO USE TWIN PAK | 2 x 4 oz | $6.00 – $7.00 | Targeted at premium users and hospitals |
Price Projection
-
Initial Market Penetration (Year 1):
Price set at approximately $6.50 per twin pack, aligning with premium offerings without overshooting market acceptance. -
Five-Year Outlook:
As manufacturing scales and market acceptance increases, a gradual price reduction to around $5.00–$5.50 is expected due to economies of scale and increased competition. The product is positioned for a mid-premium to premium segment, justified by convenience and safety features.
Factors Affecting Price Projections
- Regulatory approval timelines and associated compliance costs.
- Manufacturing costs driven by the scale of production, supplier agreements, and packaging innovations.
- Market penetration speed and competitors' response.
- Consumer willingness to pay for pre-measured, ready-to-use products versus traditional forms.
Growth & Revenue Opportunities
- Expansion in emerging markets, where demand for gastrointestinal health products is rising.
- Partnerships with healthcare providers and directly marketed to consumers through online channels.
- Product line extensions, including different pack sizes or formulations for specific patient groups.
Challenges
- Price sensitivity in developing markets.
- High regulatory hurdles in certain regions.
- Competition from established brands with significant market share.
Key Takeaways
- The global enema market exceeds $850 million with steady growth driven by aging populations and gastrointestinal health awareness.
- QC ENEMA READY TO USE TWIN PAK aims for a premium segment, with initial pricing around $6.50 per twin pack.
- Over five years, prices are expected to decline to $5.00–$5.50, influenced by economies of scale and market competition.
- Regulatory approval, manufacturing efficiency, and market positioning will affect success.
- Demand is concentrated in North America and Europe, with rapid growth expected in Asia-Pacific.
FAQs
-
What are the main competitive advantages of QC ENEMA READY TO USE TWIN PAK?
The product offers convenience, pre-measured doses, and ease of use, differentiating it from traditional powder or liquid enemas. -
How does regulatory approval impact pricing?
Obtaining clearance from authorities like the FDA or EMA involves costs that can influence initial pricing. Faster approval can also accelerate market entry and revenue. -
What is the dominant distribution channel for enema products?
Pharmacies and online medical stores dominate retail sales, while hospitals mainly purchase through direct supply contracts. -
What is the expected market share of QC ENEMA?
Early market penetration estimates suggest capturing 2–5% of the global enema market within three years, pending regulatory and marketing strategies. -
Which regions represent the highest growth potential?
Asia-Pacific and Latin America show rapid expansion prospects, driven by increased healthcare investment and urbanization.
References
[1] MarketsandMarkets. Enema market report, 2022.
[2] U.S. FDA Medical Devices Law, 2023.
[3] European Medicines Agency, Regulatory Procedures, 2023.
More… ↓
