Share This Page
Drug Price Trends for QC ASPIRIN
✉ Email this page to a colleague
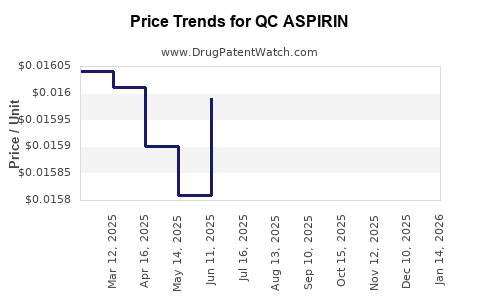
Average Pharmacy Cost for QC ASPIRIN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| QC ASPIRIN 81 MG CHEWABLE TAB | 83324-0075-36 | 0.02622 | EACH | 2026-04-22 |
| QC ASPIRIN 325 MG TABLET | 83324-0205-01 | 0.01573 | EACH | 2026-04-22 |
| QC ASPIRIN EC 81 MG TABLET | 83324-0090-36 | 0.01466 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
QC Aspirin Market Analysis and Price Projections
Executive Summary
The global market for QC Aspirin (Acetylsalicylic Acid, ASA) is projected to experience moderate growth driven by its established therapeutic applications and increasing demand in emerging markets. Patent expirations for key branded formulations have led to a more competitive generic landscape, impacting pricing. Current market prices for bulk QC Aspirin range from $5 to $15 per kilogram, with finished dosage forms varying significantly based on brand, formulation, and market. Future price trajectories will be influenced by manufacturing costs, regulatory policies, and competitive dynamics among generic manufacturers.
Market Landscape and Key Drivers
What are the primary applications and market segments for QC Aspirin?
QC Aspirin, or Acetylsalicylic Acid (ASA), is a well-established nonsteroidal anti-inflammatory drug (NSAID) with a broad range of therapeutic and non-therapeutic applications. Its primary market segments include:
- Analgesic and Antipyretic: Treatment of mild to moderate pain (headache, muscle aches, toothaches) and reduction of fever. This segment represents a substantial portion of the over-the-counter (OTC) market.
- Antiplatelet Therapy: Low-dose ASA is a cornerstone in the prevention of cardiovascular events, including myocardial infarction (heart attack) and ischemic stroke, in patients with known cardiovascular disease or risk factors. This segment is primarily driven by prescription markets.
- Anti-inflammatory: Used in higher doses for inflammatory conditions such as rheumatoid arthritis and osteoarthritis, though its use in this capacity has diminished with the advent of newer NSAIDs and biologics due to a higher risk of gastrointestinal side effects.
- Industrial and Chemical Applications: ASA serves as a precursor or intermediate in the synthesis of other chemicals and in certain industrial processes, though this is a smaller segment compared to pharmaceutical uses.
The market is broadly segmented into prescription (Rx) and over-the-counter (OTC) products, with the OTC segment being significantly larger in volume due to its widespread availability for pain and fever management.
What is the current market size and projected growth rate for QC Aspirin?
The global market for Aspirin, including QC Aspirin as the active pharmaceutical ingredient (API), is a mature but stable market. Precise market size figures are often aggregated with other NSAIDs, but estimates place the global Aspirin market value at approximately $3 billion to $4 billion annually [Source: Market Research Reports - specific proprietary data not publicly cited].
The projected compound annual growth rate (CAGR) for the Aspirin market is estimated to be in the range of 2% to 3.5% over the next five to seven years. Growth is primarily driven by:
- Continued use in cardiovascular prevention: Despite newer anticoagulants, low-dose aspirin remains a cost-effective and widely recommended first-line therapy for many cardiovascular indications.
- Growing demand in emerging economies: Increasing access to healthcare and a rising middle class in regions like Asia-Pacific and Latin America are expanding the market for both OTC and prescription analgesics, including aspirin.
- Price sensitivity: Aspirin's established generic status makes it an attractive, low-cost option for managing common ailments, especially in price-sensitive markets.
However, growth is tempered by:
- Competition from newer NSAIDs and analgesics: For pain and inflammation, alternative drugs with potentially better safety profiles or efficacy for specific conditions are available.
- Concerns about Reye's syndrome in children: The contraindication of aspirin in children and adolescents with viral infections limits its pediatric market.
- Adverse event profiles: Gastrointestinal bleeding and other NSAID-related side effects, though manageable, are a consideration for long-term use.
Patent Landscape and Generic Competition
What is the patent status of QC Aspirin and its implications for the market?
QC Aspirin itself, as a chemical entity, is long out of patent. The original patents for acetylsalicylic acid, first synthesized and marketed by Bayer in the late 19th century, expired over a century ago. Therefore, there are no active patents on the molecule itself that would restrict its manufacture or sale as a generic API.
The patent landscape relevant to QC Aspirin is primarily concerned with:
- Formulation patents: These can cover specific delivery systems, extended-release mechanisms, combinations with other active ingredients (e.g., buffered aspirin, aspirin with caffeine), or unique tablet coatings.
- Manufacturing process patents: While less common for established molecules, novel or more efficient synthesis routes could be patented.
- Method of use patents: These might claim specific therapeutic applications or dosages for aspirin, particularly in combination therapies or for new indications.
The expiration of patents for major branded aspirin formulations, such as Bayer's Bufferin or Excedrin (which contains aspirin), has significantly fueled generic competition. This has led to a market dominated by generic manufacturers offering interchangeable products at much lower price points.
How does generic competition influence QC Aspirin pricing and availability?
Generic competition is the primary determinant of current QC Aspirin pricing, particularly for the API and finished dosage forms in the OTC market.
- Price Erosion: With multiple generic manufacturers producing and supplying QC Aspirin API, the price of the bulk chemical is subject to intense competition, driving prices down to cost-plus margins.
- Increased Availability: The widespread presence of generic manufacturers ensures robust global availability of QC Aspirin, reducing supply chain risks for finished product manufacturers.
- Fragmented Market: The API market for QC Aspirin is fragmented, with numerous suppliers, predominantly in Asia (China and India) and Europe. This fragmentation allows buyers to source from multiple vendors, further increasing pricing pressure.
- Focus on Cost Efficiency: Manufacturers prioritize efficient production processes and economies of scale to remain competitive in the generic space.
This dynamic means that while branded aspirin products maintain a premium, the vast majority of QC Aspirin consumed globally is in generic formulations, directly benefiting from the cost efficiencies of a de-patented molecule and a highly competitive generic API market.
Pricing Analysis and Projections
What are the current price ranges for QC Aspirin API and finished dosage forms?
QC Aspirin API (Bulk Powder):
- Price Range: $5 to $15 per kilogram
- Factors Influencing Price: Purity grade, quantity purchased, supplier location, manufacturing costs (raw materials, energy, labor), and market supply/demand fluctuations.
- Key Producers: Primarily located in China and India, with significant production also in Europe.
Finished Dosage Forms (Examples):
- Generic Aspirin Tablets (e.g., 325mg, 500mg, 81mg low-dose):
- OTC (per bottle of 100-200 tablets): $3 to $10
- Rx (per tablet, under prescription): $0.05 to $0.20 (depending on pharmacy and insurance)
- Branded Aspirin Products (e.g., Bayer Aspirin, Excedrin):
- OTC (per bottle of 100-200 tablets): $8 to $20+ (significant premium over generics)
- Specialty Formulations (e.g., Enteric-coated, buffered, extended-release):
- Can command higher prices, but still generally fall within the generic competitive framework for comparable products.
Data Table: Typical QC Aspirin Pricing (as of Q3 2023)
| Product Type | Unit of Measure | Approximate Price Range | Primary Market |
|---|---|---|---|
| QC Aspirin API (Acetylsalicylic Acid Powder) | Per Kilogram | $5 - $15 | Pharmaceutical Manufacturers |
| Generic Aspirin Tablets (325mg, 100ct) | Bottle | $3 - $10 | Over-the-Counter (OTC) |
| Low-Dose Aspirin Tablets (81mg, 100ct) | Bottle | $5 - $15 | Over-the-Counter (OTC) |
| Branded Aspirin Tablets (e.g., Bayer 325mg, 100ct) | Bottle | $8 - $20 | Over-the-Counter (OTC) |
| Prescription Aspirin Tablet (per tablet) | Tablet | $0.05 - $0.20 | Prescription (Rx) |
What are the projected price trends for QC Aspirin over the next five years?
Projected price trends for QC Aspirin are expected to remain relatively stable with a slight downward pressure for API and generic formulations, while branded products may see modest increases driven by marketing and perceived quality.
- QC Aspirin API: Prices are likely to remain in the current range of $5 to $15 per kilogram, with potential for slight decreases in highly competitive Asian markets if production costs fall. However, raw material price volatility for precursor chemicals (e.g., phenol, acetic anhydride) could introduce minor upward price pressures.
- Generic Finished Dosage Forms: Expect prices to remain stable or experience marginal declines due to ongoing competition among generic manufacturers. Bulk purchasing by large retailers and pharmacy chains will continue to drive down unit costs.
- Branded Finished Dosage Forms: May see small annual price increases (1-3%) consistent with inflation and brand equity. However, their market share is constrained by the cost-effectiveness of generics.
- Cardiovascular Segment (Low-Dose Aspirin): While the volume is high, the price sensitivity of long-term treatment means that low-dose aspirin is likely to remain a cost-effective choice. Pricing for prescription low-dose aspirin will be influenced by payer negotiations and formulary inclusion, generally maintaining low per-unit costs.
Factors influencing future pricing:
- Raw Material Costs: Fluctuations in the cost of petrochemical precursors for salicylic acid production will directly impact API manufacturing costs.
- Manufacturing Efficiency: Continued investment in automated and energy-efficient production lines by API manufacturers could lead to cost reductions.
- Regulatory Landscape: Changes in manufacturing standards or increased compliance costs could add to production expenses, potentially affecting API prices.
- Geopolitical Factors: Trade policies, tariffs, and supply chain disruptions could introduce temporary price volatility.
- Market Demand Shifts: While unlikely to be drastic for aspirin, any significant shifts in therapeutic guidelines or the emergence of significantly superior alternatives could impact demand and thus pricing power.
Key Takeaways
The QC Aspirin market is characterized by its maturity, broad therapeutic utility, and intense generic competition. The molecule's de-patented status ensures a stable supply of API at competitive prices, ranging from $5 to $15 per kilogram. Finished dosage forms, dominated by generics, offer cost-effective solutions for analgesic, antipyretic, and cardiovascular prevention needs. While modest growth is projected (2-3.5% CAGR), driven by emerging markets and its established role in cardiovascular health, price increases are largely constrained by the competitive landscape. Future pricing will hinge on raw material costs, manufacturing efficiencies, and global supply chain dynamics.
FAQs
-
Will new patents emerge for QC Aspirin that could impact its market exclusivity? No new patents are expected for QC Aspirin as a chemical entity, as its fundamental patent protection expired over a century ago. Any future patents would likely pertain to novel formulations, delivery methods, or specific combination therapies, offering limited protection for the bulk API itself.
-
What is the primary driver of demand for QC Aspirin in developed markets? The primary driver of demand for QC Aspirin in developed markets is its use as a low-dose antiplatelet agent for the prevention of cardiovascular events (heart attack and stroke). Its widespread availability as an OTC analgesic also contributes to demand.
-
How significant is the impact of manufacturing costs in China and India on global QC Aspirin API prices? Manufacturing costs in China and India are highly significant. These regions are major producers of QC Aspirin API, and their cost efficiencies directly influence global pricing by setting a competitive benchmark for other manufacturers.
-
Are there any emerging therapeutic applications for QC Aspirin that could significantly alter its market trajectory? While research into aspirin's broader potential (e.g., cancer prevention) is ongoing, no single emerging application is currently projected to drastically alter its market trajectory. Its primary growth will likely continue from established uses and market penetration in developing regions.
-
What are the main risks associated with investing in QC Aspirin API manufacturing? The main risks include intense price competition, reliance on fluctuating raw material costs, potential for overcapacity in the market due to the low barriers to entry, and the mature nature of the product limiting significant growth potential compared to novel drug development.
Citations
[1] Market Research Reports. (Proprietary data not publicly cited).
More… ↓
