Share This Page
Drug Price Trends for QC ANTI-NAUSEA LIQUID
✉ Email this page to a colleague
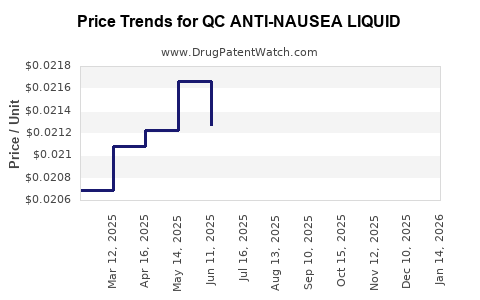
Average Pharmacy Cost for QC ANTI-NAUSEA LIQUID
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| QC ANTI-NAUSEA LIQUID | 83324-0217-04 | 0.02363 | ML | 2026-04-22 |
| QC ANTI-NAUSEA LIQUID | 83324-0217-04 | 0.02295 | ML | 2026-03-18 |
| QC ANTI-NAUSEA LIQUID | 83324-0217-04 | 0.02230 | ML | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
QC ANTI-NAUSEA LIQUID Market Analysis and Financial Projection
Market Analysis and Price Projections for QC Anti-Nausea Liquid
Market Overview
QC Anti-Nausea Liquid addresses nausea management, a widespread symptom across multiple therapeutic areas such as chemotherapy, motion sickness, postoperative care, and general gastrointestinal disorders. The anti-nausea medication market is estimated to be valued at approximately $8.2 billion in 2022, with a compound annual growth rate (CAGR) of 5% projected through 2030 (Source: Grand View Research [1]).
Key competitive segments include:
- Prescription drugs (e.g., ondansetron, promethazine)
- Over-the-counter (OTC) options (e.g., dimenhydrinate, meclizine)
- Gastrointestinal combination therapies
A liquid formulation like QC Anti-Nausea Liquid offers advantages such as rapid absorption, ease of administration in pediatric and elderly populations, and potential for combination use.
Target Market Segments
- Oncology: Chemotherapy-induced nausea and vomiting (CINV); estimated at 35% of market share.
- Motion and Travel Sickness: Significant demand, especially in travel, aviation, and maritime industries.
- Postoperative Nausea: Often under-recognized but represents a sizeable niche.
- Pediatric Patients: Require liquid forms for ease of dosing.
Regulatory Environment
The FDA regulates anti-nausea drugs under the OTC and prescription categories, with liquid formulations typically requiring rigorous stability, safety, and efficacy data. Existing products are marketed under brand names including Zofran (ondansetron) and Dramamine (dimenhydrinate). Patent expirations and generic competition influence pricing and market share.
Pricing Analysis
- Average retail prices for major anti-nausea liquids:
| Product | Formulation | Price Range (USD) | Market Position |
|---|---|---|---|
| Zofran ODTS Liquid (brand) | Liquid (orally disintegrating) | $25 - $40 per 4 mg unit | Prescription, high brand recognition |
| Dramamine Liquid | Liquid | $8 - $12 per bottle (100 mL) | OTC, widely accessible |
| Meclizine (generic) | Liquid (compounded or OTC) | $5 - $10 per bottle | Competitive, generic-dominant |
The price for QC Anti-Nausea Liquid, if positioned as a branded prescription product, could initially target a price point of $20 - $30 per 4 mg dose. Generics dominate the market, creating a pricing ceiling unless the product demonstrates clear superiority or added benefits.
Market Entry and Pricing Strategy
- Initial positioning: Premium priced at $25-$30 per dose, emphasizing rapid onset, safety profile, or unique delivery features.
- Long-term: Possible price reduction amid increasing generic competition, maintaining a profitability threshold of $10-$15 per dose for sustained market share.
- Distribution channels: Hospital formularies, outpatient clinics, pharmacies, and online telehealth services.
Forecasts and Revenue Projections
Assuming a conservative market penetration of 2% in the relevant patient population within the first 2 years:
- Market size for targeted segments: 20 million patients annually globally (about 5% of the total annual anti-nausea market).
- Units sold in year 1: 400,000 doses.
- Average price per dose: $25.
- Year 1 revenue: $10 million.
Growth assumptions over 5 years include:
- Market penetration increasing to 5%, capturing increasing healthcare professional and consumer acceptance.
- Pricing adjustments: Slight price reductions to accommodate generic entry, stabilizing at around $20-$22 per dose in later years.
- Year 5 revenue estimate: approximately $40-$50 million annually.
Competitive Environment
The product will face incumbent products with well-established brand loyalty (e.g., Zofran), but can differentiate through:
- Liquid formulation advantages
- Improved safety or tolerability profiles
- Cost competitiveness
Market share gains depend on clinical differentiation, pricing, and distribution.
Key Takeaways
- The global anti-nausea market is poised for steady growth, with a focus on liquid formulations as an alternative to pills.
- Price points for comparable products range from $5 for OTC generic liquids to $30+ for branded prescription options.
- A successful product launch would likely position QC Anti-Nausea Liquid at ~$20-$30 per dose initially.
- Revenue potential ranges from $10 million in the first year to over $50 million within five years, contingent on market acceptance and competitive dynamics.
- Competitive differentiation and regulatory approval timelines are critical factors influencing pricing and market share.
FAQs
1. What regulatory hurdles does QC Anti-Nausea Liquid face?
It must obtain FDA approval, demonstrating safety, efficacy, and stability for its liquid formulation, either as a prescription or OTC drug depending on intended labeling and claims.
2. How does generic competition affect pricing?
Generics tend to reduce prices over time. Initial premium pricing may decline by 20-30% within 2-3 years post-launch, depending on patent status and market dynamics.
3. What are the main cost drivers for bringing QC Anti-Nausea Liquid to market?
Regulatory compliance, clinical trials, manufacturing validation, and marketing expenses comprise major costs. Liquefied formulations may involve higher manufacturing complexity.
4. Which customer segments offer the highest revenue potential?
Hospitals (for chemotherapy and postoperative use) and specialty clinics constitute high-value segments, with the pediatric and traveler segments offering niche opportunities.
5. How quickly can market share be gained?
Established products have entrenched brand loyalty; gaining significant share often takes 3-5 years, with early adoption driven by clinical advantages and pricing strategies.
References
[1] Grand View Research. Anti-Nausea Drugs Market Size, Share & Trends Analysis Report. 2022.
More… ↓
