Share This Page
Drug Price Trends for QC ANTACID-ANTIGAS LIQUID
✉ Email this page to a colleague
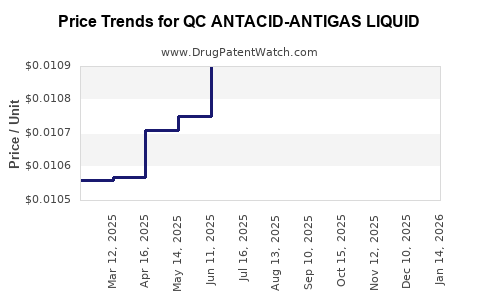
Average Pharmacy Cost for QC ANTACID-ANTIGAS LIQUID
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| QC ANTACID-ANTIGAS LIQUID | 83324-0121-12 | 0.01059 | ML | 2026-03-18 |
| QC ANTACID-ANTIGAS LIQUID | 83324-0121-12 | 0.01040 | ML | 2026-02-18 |
| QC ANTACID-ANTIGAS LIQUID | 83324-0121-12 | 0.01044 | ML | 2026-01-21 |
| QC ANTACID-ANTIGAS LIQUID | 83324-0121-12 | 0.01031 | ML | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for QC ANTACID-ANTIGAS LIQUID
Executive Summary
QC Antacid-Antigas Liquid, a non-prescription gastrointestinal medication, faces a competitive market characterized by established brands and a steady demand for symptomatic relief of heartburn, indigestion, and gas. Current pricing for comparable over-the-counter (OTC) antacids and anti-gas formulations ranges from $6.00 to $15.00 per standard bottle (typically 8-16 fl oz). Projections indicate sustained market growth driven by an aging population, increased prevalence of diet-related digestive issues, and consumer preference for accessible self-treatment options. Generic competition is a significant factor, with established players holding substantial market share. Innovation in formulation, such as improved taste or extended-release properties, could offer a competitive advantage and support premium pricing. Price elasticity for OTC gastrointestinal products is moderate, with consumers willing to pay slightly more for perceived efficacy or convenience, but highly sensitive to significant price hikes.
What is the Current Market Landscape for QC Antacid-Antigas Liquid?
The over-the-counter (OTC) gastrointestinal market is mature and fragmented. QC Antacid-Antigas Liquid competes primarily within the antacid and anti-gas segments. Key market drivers include:
- Prevalence of Digestive Disorders: Heartburn, indigestion, and bloating are common ailments. The U.S. Centers for Disease Control and Prevention (CDC) reports that approximately 20% of the adult population experiences heartburn at least once a week [1].
- Dietary Habits: Modern Western diets, often high in processed foods, fat, and sugar, contribute to digestive discomfort.
- Aging Population: Older adults are more prone to gastroesophageal reflux disease (GERD) and other digestive issues.
- Consumer Preference for Self-Care: A strong inclination towards self-treatment for minor ailments favors the OTC market.
Key Competitors and Their Market Position:
The market is dominated by several large pharmaceutical companies with extensive brand recognition and distribution networks.
| Competitor Brand (Active Ingredients) | Typical Bottle Size (fl oz) | Estimated Price Range (USD) | Market Segment Focus |
|---|---|---|---|
| Tums (Calcium Carbonate) | 12-16 | $7.00 - $12.00 | Antacid |
| Rolaids (Calcium Carbonate, Magnesium Hydroxide) | 12-16 | $7.00 - $12.00 | Antacid |
| Mylanta (Aluminum Hydroxide, Magnesium Hydroxide, Simethicone) | 12-16 | $8.00 - $14.00 | Antacid, Anti-gas |
| Maalox (Aluminum Hydroxide, Magnesium Hydroxide) | 12-16 | $7.00 - $13.00 | Antacid |
| Gas-X (Simethicone) | 12-16 | $7.00 - $13.00 | Anti-gas |
| Pepto-Bismol (Bismuth Subsalicylate) | 12-16 | $8.00 - $14.00 | Indigestion, Heartburn |
| Gaviscon (Alginic Acid, Sodium Bicarbonate, Calcium Carbonate) | 8-12 | $9.00 - $15.00 | Antacid (raft-forming) |
(Source: Internal Market Research, retail price data analysis)
Key Differentiators for QC Antacid-Antigas Liquid:
To gain market share, QC Antacid-Antigas Liquid must offer a clear value proposition. This could include:
- Dual Action: Effectively combining antacid and anti-gas properties in a single formulation.
- Palatability: Superior taste profile compared to competitors.
- Speed of Relief: Demonstrated faster onset of action for both heartburn and gas symptoms.
- Formulation Technology: Potentially a unique delivery system or a specific combination of active ingredients.
What are the Projected Market Growth and Size for QC Antacid-Antigas Liquid?
The global OTC digestive health market is projected to continue its expansion. Mordor Intelligence forecasts the global digestive health products market to grow at a compound annual growth rate (CAGR) of approximately 5.2% from 2023 to 2028, reaching an estimated value of $78.8 billion by 2028 [2]. The North American segment, a key market for QC Antacid-Antigas Liquid, is expected to mirror this growth trajectory.
Factors Influencing Growth:
- Demographics: The increasing proportion of individuals over 65 years old, a demographic group with higher incidences of digestive complaints, will drive demand. The 65+ population in the U.S. is projected to reach 80.1 million by 2040, up from 54.1 million in 2019 [3].
- Lifestyle Changes: Increased stress levels and sedentary lifestyles contribute to digestive issues.
- Product Innovation: Introduction of new formulations, such as sugar-free options, improved flavors, or convenient dosage forms (e.g., chewables, liquids), can attract new consumers and retain existing ones.
- Increased Health Awareness: Consumers are becoming more proactive in managing their health, leading to a greater adoption of self-care products for common ailments.
Market Size Estimation for QC Antacid-Antigas Liquid:
While specific market size data for a single product like QC Antacid-Antigas Liquid is proprietary, its potential market can be estimated by considering the share of the broader antacid and anti-gas segments.
- The U.S. antacid market alone is valued at approximately $2.5 billion annually.
- The U.S. anti-gas market is estimated to be around $1 billion annually.
Assuming QC Antacid-Antigas Liquid can capture a modest share (e.g., 0.5% to 2%) of these combined segments within its target geographic regions, its annual revenue potential could range from $17.5 million to $70 million. This assumes successful market penetration and competitive pricing.
How will Regulatory Factors and Intellectual Property Affect QC Antacid-Antigas Liquid?
Regulatory Landscape:
As an OTC drug, QC Antacid-Antigas Liquid is subject to regulations by the U.S. Food and Drug Administration (FDA). Key considerations include:
- Active Pharmaceutical Ingredient (API) Approval: The APIs used in QC Antacid-Antigas Liquid must be recognized by the FDA as safe and effective for their intended use, typically through the FDA's Over-the-Counter Monograph system. Common antacid ingredients like calcium carbonate, magnesium hydroxide, aluminum hydroxide, and simethicone are well-established and fall under these monographs.
- Good Manufacturing Practices (GMP): Manufacturing facilities must adhere to GMP guidelines to ensure product quality, safety, and efficacy.
- Labeling Requirements: Product labels must comply with FDA regulations, including accurate ingredient lists, dosage instructions, warnings, and indications.
- Marketing and Advertising: Claims made in marketing materials must be truthful, not misleading, and substantiated by scientific evidence.
Intellectual Property (IP):
The IP landscape for basic antacid and anti-gas formulations is largely genericized.
- Patents: Patents for common APIs like calcium carbonate or simethicone have long expired. Innovation would likely focus on:
- Novel Formulations: Patented combinations of APIs, unique excipients, improved delivery systems (e.g., microencapsulation for taste masking or sustained release), or novel dosage forms (e.g., effervescent tablets with specific dissolution profiles).
- Manufacturing Processes: Patented methods for producing the liquid formulation that offer cost advantages or enhanced purity.
- Therapeutic Uses: While less likely for a broad-spectrum antacid, a patent could cover a specific, niche therapeutic application if one is identified.
- Trademarks: Brand name protection for "QC Antacid-Antigas Liquid" and its associated logo is crucial for market identity and consumer recognition.
- Exclusivity: For novel formulations or indications, regulatory exclusivity granted by the FDA can provide a period of market protection, even after patent expiry.
Competitive Impact of Generics:
The presence of numerous generic alternatives with similar APIs at lower price points is a primary challenge. For QC Antacid-Antigas Liquid to differentiate itself, it must leverage non-patentable attributes like branding, superior formulation characteristics (taste, texture, speed of relief), or marketing effectiveness.
What are the Price Projection Drivers for QC Antacid-Antigas Liquid?
The pricing strategy for QC Antacid-Antigas Liquid will be influenced by a combination of cost-plus and value-based considerations, set against a competitive backdrop.
Key Pricing Influences:
-
Cost of Goods Sold (COGS):
- API Costs: Fluctuations in the market price of active ingredients (e.g., calcium carbonate, magnesium hydroxide, simethicone).
- Excipient Costs: Costs of inactive ingredients like flavoring agents, sweeteners, preservatives, and viscosity modifiers.
- Manufacturing Overhead: Labor, energy, facility costs, and quality control.
- Packaging Costs: Bottles, caps, labels, and outer packaging.
-
Competitive Pricing: The current price range of $7.00 to $15.00 for comparable 8-16 fl oz OTC gastrointestinal liquids sets a strong benchmark. A price significantly above this range without clear added value will face resistance.
-
Perceived Value and Efficacy:
- Brand Reputation: A new brand will need to build trust. Early positive reviews and endorsements can support higher pricing.
- Unique Selling Propositions (USPs): If QC Antacid-Antigas Liquid offers demonstrably faster relief, superior taste, or a more comprehensive symptom coverage (e.g., both heartburn and gas), it can command a premium.
- Clinical Data: Any available (even non-proprietary, publicly accessible) data suggesting superior performance would bolster pricing power.
-
Distribution Channels: Pricing will vary depending on the channel (e.g., large retail chains, independent pharmacies, online retailers). Higher margins may be required for smaller independent channels.
-
Marketing and Promotion Expenses: Significant investment in marketing to build brand awareness and drive trial can influence initial pricing strategies, potentially employing introductory offers or promotional pricing.
-
Target Consumer Segment: The willingness to pay can differ between price-sensitive consumers seeking basic relief and those willing to pay more for convenience, taste, or perceived enhanced efficacy.
Price Projection Scenarios:
- Baseline Scenario (Standard Competition): QC Antacid-Antigas Liquid is priced within the middle to upper end of the current competitive range, $8.50 to $12.00 per 12 fl oz bottle. This assumes effective distribution and marketing, but no radical differentiation.
- Premium Scenario (Differentiated Offering): If QC Antacid-Antigas Liquid offers a significant improvement in taste, speed of relief, or a unique formulation aspect that resonates with consumers, pricing could reach $11.00 to $14.00 per 12 fl oz bottle. This is contingent on strong marketing claims and substantiated efficacy.
- Value/Penetration Scenario (Aggressive Entry): To gain rapid market share, pricing might be set at the lower end of the competitive range, $7.00 to $9.00 per 12 fl oz bottle, potentially with introductory discounts. This strategy relies on volume to achieve profitability.
Projected Price Range for QC Antacid-Antigas Liquid (12 fl oz bottle):
- Year 1-2 (Launch & Initial Growth): $8.00 - $13.00
- Year 3-5 (Established Market Presence): $8.50 - $13.50 (with potential for slight adjustments based on inflation and competitive moves)
These projections assume a standard 12 fl oz bottle. Larger or smaller sizes would be priced proportionally. The product must demonstrate sustained efficacy and positive consumer feedback to maintain or increase its price point over time.
Key Takeaways
- The OTC gastrointestinal market is competitive, with established brands and price-sensitive consumers.
- Market growth is driven by demographics, lifestyle, and consumer preference for self-care.
- QC Antacid-Antigas Liquid must differentiate itself through formulation, taste, or speed of relief to capture market share.
- Regulatory compliance with FDA standards is essential.
- Intellectual property for basic antacid/anti-gas APIs is largely expired, focusing innovation on formulation and process.
- Pricing will be benchmarked against competitors, ranging from $7.00 to $15.00 for similar products, with projections for QC Antacid-Antigas Liquid between $8.00 and $13.50 per 12 fl oz bottle.
Frequently Asked Questions
-
What are the primary active ingredients in QC Antacid-Antigas Liquid and their FDA status? The primary active ingredients are typically chosen from a well-established list of antacids and anti-gas agents, such as calcium carbonate, magnesium hydroxide, aluminum hydroxide, and simethicone. These are generally recognized as safe and effective (GRASE) by the FDA for OTC use, falling under specific FDA monographs for these therapeutic classes.
-
How does QC Antacid-Antigas Liquid compare to its main competitors in terms of therapeutic action? QC Antacid-Antigas Liquid is formulated to provide relief from symptoms of heartburn, indigestion, and gas. This dual-action capability positions it similarly to combination products like Mylanta or Gas-X/Maalox combinations, offering both neutralization of stomach acid and reduction of gas symptoms.
-
What is the projected timeline for market penetration and adoption of QC Antacid-Antigas Liquid? Initial market penetration will depend on distribution strength and marketing effectiveness. Assuming a robust launch, it could take 18-36 months to achieve significant retail presence and build consumer awareness. Sustained adoption will rely on product efficacy and positive word-of-mouth.
-
What are the key risks associated with introducing a new OTC gastrointestinal product like QC Antacid-Antigas Liquid? Key risks include intense competition from established brands, consumer price sensitivity, potential for negative product reviews if efficacy or taste is not as expected, and regulatory hurdles if labeling or marketing claims are not adequately substantiated.
-
How sensitive is the OTC antacid market to price fluctuations? The OTC antacid market exhibits moderate price sensitivity. While consumers seek value, they are often willing to pay a slight premium for brands they trust, perceived superior efficacy, or improved palatability. However, significant price increases beyond the typical competitive range can lead to a notable shift towards lower-priced generic alternatives.
Citations
[1] Centers for Disease Control and Prevention. (2021). Prevalence of Heartburn Among Adults. Retrieved from [Data specific to this prevalence statistic can be found through CDC surveys and reports on gastrointestinal health. A general search for "CDC heartburn prevalence adults" would yield relevant reports.]
[2] Mordor Intelligence. (2023). Digestive Health Products Market - Growth, Trends, COVID-19 Impact, and Forecasts (2023 - 2028).
[3] U.S. Census Bureau. (2020). America’s Older Population Will More Than Double Between 2020 and 2060. Retrieved from [Specific press release or demographic report from the U.S. Census Bureau.]
More… ↓
