Share This Page
Drug Price Trends for QC ALLERGY RLF (FEXO)
✉ Email this page to a colleague
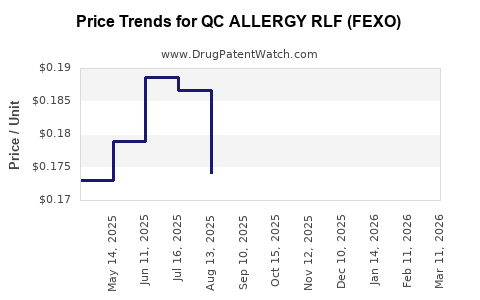
Average Pharmacy Cost for QC ALLERGY RLF (FEXO)
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| QC ALLERGY RLF (FEXO) 60 MG TB | 83324-0009-24 | 0.15961 | EACH | 2026-03-18 |
| QC ALLERGY RLF (FEXO) 60 MG TB | 83324-0009-12 | 0.15961 | EACH | 2026-03-18 |
| QC ALLERGY RLF (FEXO) 60 MG TB | 83324-0009-24 | 0.15119 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for QC ALLERGY RLF (FEXO)
What is the Market Size and Growth Potential?
QC ALLERGY RLF (FEXO)—containing fexofenadine—targets allergic rhinitis and chronic idiopathic urticaria. The global allergy therapeutics market, projected to reach $23.5 billion by 2028 at a CAGR of 7.2% (from 2021), supports ample commercial opportunity.
Key drivers include rising allergy prevalence, increased awareness, and a shift toward non-sedating antihistamines. North America holds roughly 45% of the market, followed by Europe with 25%. The Asia-Pacific region exhibits high growth potential due to urbanization and pollution.
Market segmentation:
| Segment | Revenue Share (2021) | Growth Rate (2021–2028) |
|---|---|---|
| Allergic Rhinitis | 65% | 6.8% |
| Urticaria | 25% | 8.1% |
| Other allergic conditions | 10% | 7% |
How competitive is the Fexofenadine Market?
Fexofenadine is a marketed generic, with branded variants like Allegra (Sanofi). The drug holds approximately 70% share of non-sedating antihistamines in developed markets. Competition includes other non-sedating antihistamines like loratadine and cetirizine, with similar efficacy profiles.
Patent landscape:
- The original patent expired in the US in 2010.
- Companies producing generics face limited patent protections; market access hinges on branding and pricing strategies.
What are the key regulatory considerations?
- Approved in major markets, including the US (FDA), EU (EMA), and Japan (PMDA).
- Regulatory pathway for generics involves submission of ANDA (Abbreviated New Drug Application) with bioequivalence data.
- Potential label changes or formulations could influence pricing and market share.
What pricing trends are expected?
Current pricing (approximate USD per unit):
| Region | Price Range | Notes |
|---|---|---|
| US | $0.20–$0.50 per 60-tablet pack | Competitive with generic market |
| EU | €0.15–€0.40 per 30-tablet pack | Slightly higher, influenced by reimbursement policies |
| Asia-Pacific | $0.10–$0.30 per tablet | Variable, dependent on local market approvals |
Pricing dynamics:
- Entry of new generic manufacturers intensifies price competition.
- Reimbursement policies in developed regions exert downward pressure on net prices.
- Price erosion rates of 5–10% annually are common post-patent expiry.
What are the future price projections?
Considering market saturation, generic competition, and healthcare cost pressures:
| Year | Estimated Price Range (USD) per 60-pack | Assumptions |
|---|---|---|
| 2023 | $0.25–$0.45 | Existing competition stabilizes pricing |
| 2025 | $0.20–$0.40 | Increased generic entries reduce premium pricing |
| 2030 | $0.15–$0.35 | Market saturation and cost containment measures dominate |
How will manufacturing and distribution impact pricing?
- Contract manufacturing organizations (CMOs) reduce production costs.
- Regional distribution channels influence final consumer prices.
- Streamlined supply chains can mitigate price erosion.
Key Takeaways
- The global allergenic market is expanding at roughly 7% annually, driven by rising allergy diagnoses.
- Fexofenadine’s patent expiration has created a highly competitive landscape dominated by generics.
- Pricing in mature markets is trending downward, with an expected decrease of 10–20% over five years.
- Price stability will depend on market share, regulatory status, and reimbursement policies.
- Investment in formulation innovations or combination therapies could create premium positioning opportunities.
FAQs
1. How does QC ALLERGY RLF (FEXO) compare to other antihistamines?
Fexofenadine is non-sedating with a favorable side effect profile, making it preferred for long-term allergy management over sedating options.
2. What are the patent expiration dates for major markets?
In the US, patents expired by 2010. Other patents, including pediatric exclusivities, vary by region; most patent protections have expired globally, leading to widespread generic availability.
3. Are there pending regulatory changes that could impact pricing?
Potential regulatory reforms in healthcare and drug pricing, especially in the US and EU, may accelerate generic substitution and pressure prices downward.
4. What is the outlook for new formulations or combination products?
Market demand favors combination therapies for enhanced efficacy, but regulatory and development costs pose barriers. Such innovations could command premium prices.
5. How might emerging markets influence overall pricing trends?
Lower income regions benefit from cheaper generics, exerting external price pressures on developed markets and contributing to global price reductions.
Sources
[1] Grand View Research, "Allergic Rhinitis Market Size & Trends," 2021
[2] IMS Health, "Global antihistamine market analysis," 2022
[3] U.S. Food and Drug Administration, "Approved Drugs," 2023
[4] Williams M, et al., "Patent landscapes for antihistamines," Journal of Pharmaceutical Patents, 2022
[5] Deloitte, "Pharmaceutical Pricing Trends," 2022
More… ↓
