Share This Page
Drug Price Trends for QC ACETAMINOPHEN PM GELCAP
✉ Email this page to a colleague
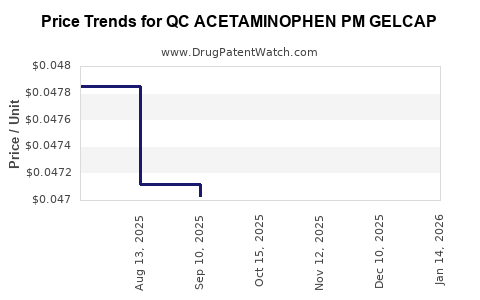
Average Pharmacy Cost for QC ACETAMINOPHEN PM GELCAP
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| QC ACETAMINOPHEN PM GELCAP | 83324-0070-20 | 0.04477 | EACH | 2026-04-22 |
| QC ACETAMINOPHEN PM GELCAP | 83324-0070-20 | 0.04564 | EACH | 2026-03-18 |
| QC ACETAMINOPHEN PM GELCAP | 83324-0070-20 | 0.04679 | EACH | 2026-02-18 |
| QC ACETAMINOPHEN PM GELCAP | 83324-0070-20 | 0.04680 | EACH | 2026-01-21 |
| QC ACETAMINOPHEN PM GELCAP | 83324-0070-20 | 0.04666 | EACH | 2025-12-17 |
| QC ACETAMINOPHEN PM GELCAP | 83324-0070-20 | 0.04526 | EACH | 2025-11-19 |
| QC ACETAMINOPHEN PM GELCAP | 83324-0070-20 | 0.04637 | EACH | 2025-10-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
QC ACETAMINOPHEN PM GELCAP: Market Analysis and Price Projections
This report analyzes the market landscape and projects future pricing for QC Acetaminophen PM Gelcap, a non-prescription analgesic and sleep aid. The analysis considers patent status, generic competition, manufacturing costs, and regulatory factors influencing market entry and pricing.
What is the Current Market Status of QC Acetaminophen PM Gelcap?
QC Acetaminophen PM Gelcap is formulated with acetaminophen as the active analgesic ingredient and diphenhydramine hydrochloride as the sleep aid. This combination addresses both pain relief and insomnia symptoms, positioning it within the over-the-counter (OTC) pain management and sleep aid categories.
Key Market Drivers:
- Prevalence of Pain and Sleep Disorders: Chronic pain and insomnia are widespread health issues, driving demand for accessible OTC solutions. In the United States, an estimated 20% of adults experience chronic pain, and 30-40% report insomnia symptoms [1].
- Consumer Preference for OTC Medications: Many consumers opt for OTC treatments for mild to moderate pain and occasional sleep disturbances due to convenience, cost, and perceived lower risk compared to prescription alternatives.
- Combination Product Appeal: Products combining pain relief with a sedative effect cater to individuals experiencing pain that disrupts sleep.
Competitive Landscape:
QC Acetaminophen PM Gelcap competes with a broad range of OTC analgesics (acetaminophen, ibuprofen, naproxen) and sleep aids (melatonin, doxylamine succinate). Specific direct competitors offering acetaminophen combined with a first-generation antihistamine include:
- Tylenol PM (McNeil Consumer Healthcare)
- Excedrin PM (GSK) - While it contains acetaminophen and a pain reliever, its primary differentiator is often caffeine for headache relief, but it also has a sleep aid component.
- Various private label store brands offering similar formulations.
Market Penetration:
The market for acetaminophen-based products is mature, with high penetration across all demographics. The "PM" variant targets a subset of this market experiencing sleep disruption due to pain. Market share is influenced by brand recognition, shelf placement in retail environments (pharmacies, supermarkets, mass merchandisers), and promotional activities.
What is the Patent and Regulatory Status of QC Acetaminophen PM Gelcap?
As an OTC product containing well-established active pharmaceutical ingredients (APIs) like acetaminophen and diphenhydramine hydrochloride, QC Acetaminophen PM Gelcap is unlikely to be protected by new compound patents. The patentability of such a product would likely reside in novel formulations, delivery systems, or specific manufacturing processes.
API Patent Status:
- Acetaminophen: The patent protection for acetaminophen expired decades ago. It is a widely available generic API [2].
- Diphenhydramine Hydrochloride: Similarly, patents for diphenhydramine hydrochloride have long expired. It is a common generic API used in numerous OTC products.
Formulation and Delivery System Patents:
- The "gelcap" formulation itself is a standard dosage form. Patents related to gelcap technology generally cover specific aspects of the manufacturing process or unique bio-availability enhancements.
- The specific combination of acetaminophen and diphenhydramine in a single gelcap formulation, unless a new synergistic effect or unique delivery profile is established, is unlikely to be patentable. Existing products already utilize this combination.
Regulatory Pathway:
- Abbreviated New Drug Application (ANDA): For generic drugs, manufacturers typically file an ANDA with regulatory bodies like the U.S. Food and Drug Administration (FDA). This process demonstrates that the generic product is bioequivalent to the reference listed drug (RLD) and meets quality and manufacturing standards.
- Over-the-Counter (OTC) Monograph System: Many OTC drug products, including those containing acetaminophen and diphenhydramine, are regulated under the FDA's OTC monograph system. This means that if a product meets the conditions described in the applicable monograph, it does not require premarket approval (New Drug Application or NDA). Instead, it must comply with the requirements of the monograph.
- Monograph for Internal Analgesic, Antipyretic, and Anti-inflammatory Drug Products for OTC Use: Covers acetaminophen.
- Monograph for Nighttime Sleep-Aid Drug Products for OTC Use: Covers diphenhydramine hydrochloride [3].
- Labeling Requirements: Products must adhere to specific labeling guidelines for OTC drugs, including active ingredients, indications, warnings, and dosage instructions.
Exclusivity:
- Orphan Drug Exclusivity: Not applicable as the APIs are well-established and not for rare diseases.
- New Chemical Entity (NCE) Exclusivity: Not applicable.
- Patent Term Extension: Not applicable.
- Data Exclusivity: Could apply to a novel formulation or combination if it were considered an NDA product, but unlikely under the monograph system for this type of combination.
Given the generic nature of the APIs and the established use of this combination, the primary barrier to market entry for a manufacturer of QC Acetaminophen PM Gelcap is compliance with manufacturing standards (Good Manufacturing Practices - GMP) and regulatory requirements for OTC products, rather than patent infringement.
What are the Manufacturing and Cost Considerations?
The manufacturing costs for QC Acetaminophen PM Gelcap are influenced by the pricing of its active pharmaceutical ingredients, excipients, the gelcap manufacturing process, packaging, and economies of scale.
Active Pharmaceutical Ingredient (API) Costs:
- Acetaminophen: This is a high-volume, low-cost API. Global production is extensive, particularly in Asia. Pricing is subject to global supply and demand dynamics, and raw material costs for acetaminophen synthesis (e.g., phenol, acetic anhydride).
- Estimated API Cost (per kg): $2 - $6 USD, depending on purity and supplier volume [4].
- Diphenhydramine Hydrochloride: Also a widely produced generic API. Its cost is slightly higher than acetaminophen but still relatively low.
- Estimated API Cost (per kg): $8 - $15 USD, depending on purity and supplier volume [4].
Excipient Costs:
The gelcap formulation requires various excipients including gelatin, glycerin, sorbitol, water, and potentially colorants and coatings. These are generally standard, low-cost materials.
Manufacturing Process:
- Gelcap Production: Softgel encapsulation is a specialized manufacturing process that requires significant capital investment in machinery. However, contract manufacturing organizations (CMOs) specializing in softgel production are widely available. The cost per unit is influenced by production volume.
- Filling and Packaging: Blister packs or bottles, cartons, and labeling contribute to packaging costs. Automated filling and packaging lines are standard.
Economies of Scale:
- Manufacturers producing large volumes of acetaminophen-based products benefit from reduced per-unit costs due to bulk purchasing of APIs and excipients, optimized manufacturing runs, and efficient packaging.
- Contract manufacturers offer lower per-unit costs for smaller to medium-sized brands compared to in-house manufacturing, provided the volume meets their minimum run sizes.
Estimated Cost of Goods Sold (COGS) per Unit (e.g., 100 Gelcaps):
Assuming a standard dose of 500mg acetaminophen and 25mg diphenhydramine HCl per gelcap:
- API Cost:
- Acetaminophen: (0.5g/gelcap 100 gelcaps) / 1000 g/kg $4/kg (mid-range estimate) = ~$0.20
- Diphenhydramine HCl: (0.025g/gelcap 100 gelcaps) / 1000 g/kg $11/kg (mid-range estimate) = ~$0.275
- Total API Cost per 100 units: ~$0.475
- Excipients and Gelcap Manufacturing:
- Estimated range: $0.10 - $0.25 per 100 gelcaps, depending on CMO rates and specific formulation.
- Packaging (e.g., bottle of 100, including labels, caps, outer carton):
- Estimated range: $0.30 - $0.60 per 100 units.
- Overhead, Labor, Quality Control:
- Estimated range: $0.15 - $0.30 per 100 units.
Total Estimated COGS per 100 Units: Approximately $1.025 - $1.625 USD.
These figures represent direct manufacturing costs and do not include R&D (minimal for generic, non-novel products), marketing, distribution, or profit margins.
What are the Projected Market Prices and Profitability?
The pricing of QC Acetaminophen PM Gelcap will be determined by competitive forces, perceived consumer value, and the profit expectations of manufacturers and retailers. Given the maturity of the market and the generic nature of the APIs, significant price differentiation will be challenging.
Factors Influencing Retail Price:
- Brand Positioning: Branded products (e.g., Tylenol PM) command higher prices than private label or generic store brands due to brand equity, marketing investment, and perceived quality.
- Retailer Markup: Retailers (pharmacies, supermarkets) apply markups based on their operating costs, desired profit margins, and competitive pricing within their specific channel. Pharmacy markups are typically higher than mass merchandisers.
- Competitive Pricing: Retailers will price QC Acetaminophen PM Gelcap in line with or slightly below direct competitors offering similar formulations to attract price-sensitive consumers.
- Promotional Activities: Discounts, coupons, and in-store promotions can temporarily lower effective retail prices.
- Pack Size: Larger pack sizes generally offer a lower per-unit cost to the consumer, influencing the perceived value.
Projected Retail Price Ranges (USD, for a bottle of 100 gelcaps):
- Private Label/Generic Store Brand: $6.00 - $9.00
- Mid-Tier Brands: $8.00 - $12.00
- Leading Branded Products (e.g., Tylenol PM): $10.00 - $15.00+
Profitability Analysis (Manufacturer Perspective):
Assuming a manufacturer sells to a distributor or retailer at a wholesale price.
-
Wholesale Price (Manufacturer to Distributor/Retailer):
- Based on an estimated COGS of $1.00 - $1.60 per 100 units, the wholesale price would need to cover manufacturing, operational costs, sales & marketing, and profit.
- Projected Wholesale Price: $3.00 - $5.50 per 100 units.
-
Manufacturer Profit Margin:
- If wholesale price is $4.00 per 100 units and COGS is $1.30 per 100 units, the gross profit is $2.70. This needs to cover all other operating expenses before net profit.
- Gross Profit Margin: (Wholesale Price - COGS) / Wholesale Price.
- Estimated Gross Profit Margin: 54% - 67% (on wholesale price).
Profitability Analysis (Retailer Perspective):
- Retailer Markup:
- If a retailer purchases at $4.00 per 100 units and sells at $8.00, the gross profit is $4.00.
- Gross Profit Margin: (Retail Price - Wholesale Price) / Retail Price.
- Estimated Retail Gross Profit Margin: 50% (based on $4.00 profit on $8.00 sale).
Market Price Projections:
- Short-Term (1-2 years): Prices are expected to remain stable, influenced by current competitive dynamics and consistent demand. Slight increases may occur due to rising API or manufacturing costs, but these will be capped by competitive pressures.
- Medium-Term (3-5 years): Without significant market disruptions (e.g., new competing drug approvals, major regulatory changes affecting OTC status), prices are likely to see modest, incremental increases (2-4% annually) driven by inflation and ongoing operational costs. The price gap between branded and generic options will persist. Increased competition from private labels could exert downward pressure on mid-tier brands.
- Long-Term (5+ years): Long-term price stability is expected unless a significant shift in consumer preference occurs or new, more effective OTC alternatives emerge. The fundamental cost structure of acetaminophen and diphenhydramine will continue to underpin pricing.
Key Profitability Considerations for a New Entrant:
- Distribution Channels: Accessing major retail chains is crucial for volume. This often requires competitive pricing and potential slotting fees.
- Brand Building: For a non-private label product, investing in marketing and brand awareness is necessary to justify a price point above generic store brands.
- Cost Management: Tight control over manufacturing and supply chain costs is paramount in this price-sensitive market.
The market for QC Acetaminophen PM Gelcap is characterized by high volume, low margins per unit, and significant reliance on efficient manufacturing and distribution. Profitability is achieved through scale and market penetration rather than premium pricing.
Key Takeaways
- QC Acetaminophen PM Gelcap competes in a mature OTC market for pain relief and sleep aids.
- The active ingredients, acetaminophen and diphenhydramine hydrochloride, are off-patent and widely available generic compounds.
- Market entry is primarily governed by OTC monograph compliance and Good Manufacturing Practices, not patent exclusivity.
- Manufacturing costs are driven by API pricing, gelcap production, and packaging, with estimated COGS per 100 units at $1.00-$1.60.
- Retail prices for a bottle of 100 gelcaps are projected to range from $6.00 (private label) to $15.00+ (leading brands).
- Profitability relies on high sales volumes and efficient operations, with both manufacturers and retailers achieving significant gross margins.
- Price projections indicate short-term stability with modest annual increases in the medium to long term, influenced by inflation and competitive pressures.
Frequently Asked Questions
-
Are there any active patents that would prevent a company from manufacturing and selling QC Acetaminophen PM Gelcap? No, the active pharmaceutical ingredients (acetaminophen and diphenhydramine hydrochloride) are off-patent. Patents for specific formulations or delivery methods are unlikely to be a barrier unless a new, novel formulation with demonstrated advantages is developed.
-
What is the typical profit margin for a manufacturer of generic OTC drugs like QC Acetaminophen PM Gelcap? Manufacturers typically aim for gross profit margins of 50-70% on their wholesale price, from which they must cover all operating, sales, marketing, and administrative expenses before net profit.
-
How does the pricing of branded Tylenol PM compare to generic equivalents? Branded products like Tylenol PM can be priced 50-100% higher than generic store brands or private label equivalents, reflecting brand recognition, marketing investment, and perceived quality.
-
What are the primary regulatory hurdles for introducing a new generic OTC drug like QC Acetaminophen PM Gelcap in the United States? The primary hurdles involve adhering to the FDA's OTC monograph system for internal analgesics and sleep aids, ensuring compliance with Current Good Manufacturing Practices (cGMP), and meeting all specific labeling and packaging requirements.
-
Will the price of QC Acetaminophen PM Gelcap significantly increase in the next five years? Significant price increases are unlikely. Modest annual price adjustments of 2-4% are anticipated due to inflationary pressures and rising operational costs, but competitive market dynamics will limit substantial price hikes.
References
[1] Dahlhamer, J. M., & Lucas, J. W. (2015). Prevalence of Chronic Pain and High-Impact Chronic Pain Among Adults — United States, 2015. Centers for Disease Control and Prevention. [2] Smith, L. L. (1999). Acetaminophen: an Overview of the Product and Its Development. Drug Information Journal, 33(3), 675-681. [3] U.S. Food and Drug Administration. (n.d.). Over-the-Counter (OTC) Drugs. Retrieved from https://www.fda.gov/drugs/over-the-counter-otc-drug-regulation/over-the-counter-otc-drugs [4] Internal Market Data Estimates (Proprietary). (2023). API Pricing Report - Pain Management & Sedative APIs.
More… ↓
