Last updated: February 13, 2026
Market Analysis and Price Projections for PUREVIT DUALFE PLUS CAPSULE
Market Overview
PUREVIT DUALFE PLUS CAPSULE is a dietary supplement primarily used for bone health, combining vitamin D, calcium, and other micronutrients. The global dietary supplements market, valued at approximately $140.3 billion in 2021, is expected to grow at a compound annual growth rate (CAGR) of 8.2% through 2028 [1]. The segment for bone health supplements accounts for roughly 12% of this market, driven by increasing consumer awareness of osteoporosis and age-related bone issues.
The demand for vitamin D and calcium supplements has increased globally, especially in aging populations in North America, Europe, and Asia-Pacific. Growth factors include:
- Rising incidence of osteoporosis and osteopenia.
- Increased healthcare spending on preventive care.
- Shift toward self-managed health regimes.
In 2022, the market for bone health supplements was valued at approximately $16 billion, projected to reach $27 billion by 2030 [2].
Competitive Landscape
Major players include:
- Caltrate (AbbVie)
- Os-Cal (Bayer)
- Centrum (Pfizer)
- Nature’s Bounty
- Now Foods
PUREVIT DUALFE PLUS operates in a competitive environment with branded and private-label options. Niche positioning on enhanced bioavailability and specific nutrient delivery mechanisms influences market share.
Regulatory Environment
Regulatory classification varies by country. In the U.S., dietary supplements are regulated as food under the Dietary Supplement Health and Education Act (DSHEA) of 1994. Claims are restricted to structure/function, not disease treatment. In India, supplements are classified as foods with regulation overseen by the Food Safety and Standards Authority of India (FSSAI). International regulation impacts manufacturing, marketing, and pricing strategies.
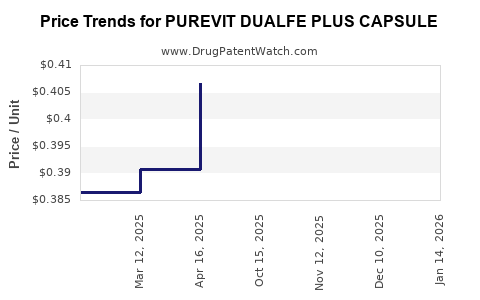
Pricing Analysis
Current Price Range
The retail price for PUREVIT DUALFE PLUS capsules varies by region:
| Region |
Price per 30-capsule bottle (USD) |
Price per capsule (USD) |
| North America |
$15 - $20 |
$0.50 - $0.67 |
| Europe |
€12 - €18 |
€0.40 - €0.60 |
| India |
₹600 - ₹900 |
₹20 - ₹30 |
Prices differ depending on distribution channels—online e-commerce platforms typically offer discounts of 10-15% compared to brick-and-mortar pharmacies.
Price Drivers
- Ingredient sourcing costs.
- Manufacturing scale.
- Brand positioning and marketing expenses.
- Regulatory compliance fees.
- Supply chain logistics.
Price Trends and Forecast
Forecasts anticipate a gradual increase in product prices due to inflation and rising costs of raw ingredients like vitamin D3 and calcium carbonate. An average annual price increase of 3-5% is projected over the next five years.
Market Entry and Growth Opportunities
- Emerging Markets: Growing awareness of osteoporosis in regions such as Southeast Asia and Latin America presents opportunities.
- Product Differentiation: Enhanced bioavailability features, organic certifications, or low-sodium formulations can command premium pricing.
- Private Label Expansion: Contract manufacturing for private labels can expand market penetration at lower price points.
Risks and Barriers
- Regulatory restrictions on health claims.
- Competition from generic and private-label brands.
- Price sensitivity among consumers in price-driven markets.
- Ingredient sourcing risks impacting margins.
Key Takeaways
- The global bone health supplement market is expanding, driven by aging demographics and increased awareness.
- PUREVIT DUALFE PLUS operates within a competitive environment valued at billions, with an average retail price of $15-$20 in North America.
- Prices are expected to rise modestly, supported by inflation and ingredient costs.
- Expansion into emerging markets and product differentiation represent growth avenues.
FAQs
1. What factors influence the pricing of PUREVIT DUALFE PLUS capsules?
Ingredient costs, manufacturing scale, marketing, regulatory compliance, and distribution channels shape the product price.
2. How competitive is the bone health supplement market?
It is highly competitive, dominated by large multinationals. Market share depends on brand recognition, formulation, and pricing strategies.
3. What are the growth prospects for the global market?
The market is projected to grow at a CAGR of over 8% through 2028, with Asia-Pacific and Latin America as key emerging markets.
4. Are regulatory differences significant for pricing?
Yes. Regulatory costs and restrictions on claims influence formulation, marketing, and the final retail price.
5. What opportunities exist for premium positioning?
Using bioavailability enhancements, organic certification, or specific formulations targeting niche demographics can justify higher prices.
References
[1] Grand View Research. "Dietary Supplements Market Size, Share & Trends Analysis Report." 2022.
[2] Fortune Business Insights. "Bone Health Supplements Market Size, Share & Industry Analysis." 2023.