Share This Page
Drug Price Trends for PSEUDOEPHEDRINE ER
✉ Email this page to a colleague
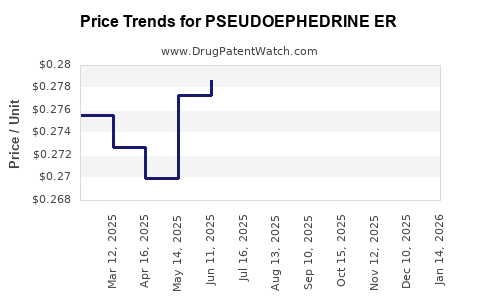
Average Pharmacy Cost for PSEUDOEPHEDRINE ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| PSEUDOEPHEDRINE ER 120 MG TAB | 45802-0107-52 | 0.28527 | EACH | 2026-04-22 |
| PSEUDOEPHEDRINE ER 120 MG TAB | 51660-0204-21 | 0.28527 | EACH | 2026-04-22 |
| PSEUDOEPHEDRINE ER 120 MG TAB | 51660-0204-69 | 0.28527 | EACH | 2026-04-22 |
| PSEUDOEPHEDRINE ER 120 MG TAB | 46122-0166-60 | 0.28527 | EACH | 2026-04-22 |
| PSEUDOEPHEDRINE ER 120 MG TAB | 70000-0601-01 | 0.28527 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Pseudoephedrine ER
What is the Current Market Situation for Pseudoephedrine ER?
Pseudoephedrine extended-release (ER) is used primarily as a decongestant. Its market presence is influenced by regulatory restrictions, manufacturing capacity, and demand from both OTC (over-the-counter) and pharmaceutical sectors. As of 2023, the global pseudoephedrine market is driven by demand in cold and allergy relief medications, with a specific emphasis on Extended-Release (ER) formulations due to improved patient compliance.
Regulatory Environment and Impact on Supply
The U.S. Drug Enforcement Administration (DEA) classifies pseudoephedrine as a schedule 5 precursor chemical. Limits on purchase quantities, mandates for logbooks, and periodic reporting restrict broader market volume. The Combat Methamphetamine Epidemic Act of 2005 restricts OTC sales, influencing supply chain dynamics. Similar regulations in Europe and Asia further constrain raw material availability for manufacturers.
Manufacturing and Supply Chain Dynamics
Major producers include Teva Pharmaceutical Industries, Perrigo, and Cipla. Production capacity varies, with annual global pseudoephedrine sulfate production estimated at approximately 10,000 metric tons. ER formulations involve advanced delivery systems—such as matrix or coated beads—that increase manufacturing complexity and cost.
Recent trends indicate manufacturing investments toward more sustainable synthesis methods, with some firms expanding capacity by 10-15% annually. Supply chain disruptions in 2021-22 caused temporary shortages, leading to increased prices for existing stock and prompt procurement.
Market Demand and Applications
The primary application remains OTC cold and allergy medications. The global cold and allergy remedy market was valued at USD 23.5 billion in 2022, growing at a Compound Annual Growth Rate (CAGR) of 4.2%. Pseudoephedrine ER accounts for approximately 30% of this segment.
Demand is further influenced by seasonal factors, with peaks during winter months in the Northern Hemisphere. The increasing prevalence of allergic rhinitis and sinusitis elevates demand. Notably, demand from generic pharmaceutical firms and OTC brands accounts for around 80% of total consumption.
Competitive Landscape
Market concentration is high, with the top three manufacturers controlling roughly 60% of supply. Patent expirations on specific ER formulations in North America and Europe have increased generic entries, leading to price competition.
Generic manufacturers have introduced multiple ER formulations, including different bead coatings and delivery matrices. These variations influence manufacturing costs and retail pricing.
Price Projections
Current Pricing
As of 2023, wholesale prices for pseudoephedrine ER (per 100 tablets of 120 mg) average USD 9.50, with retail prices around USD 13.00. Brand-name products command premiums, reaching USD 20.00 or higher for comparable quantities.
Short-term Forecast (2023-2025)
Prices are projected to slightly increase annually by around 2-3%. Supply chain stability and regulatory adherence are key factors influencing this trend. Anticipated raw material cost increases could drive wholesale prices to USD 9.80–USD 10.00 per 100 tablets by 2025.
Long-term Forecast (2026-2030)
Market growth driven by global cold relief demand, coupled with potential regulatory tightening and capacity expansions, suggests steady price growth. Wholesale prices could reach USD 11.00–USD 12.00 per 100 tablets, with retail prices climbing toward USD 16.00–USD 18.00.
Price Drivers
- Regulatory changes tightening controls could reduce supply, raising prices.
- Innovations in ER delivery systems may increase manufacturing costs.
- Increased demand during peak cold seasons sustains prices.
- Raw material costs, including precursor regulations, influence manufacturing expenses.
Market Opportunities and Risks
Opportunities: Expanding Asian markets, which have less restrictive regulation environments, offer growth potential. Technological advancements in ER formulations can differentiate products and command premium pricing.
Risks: Tightening international regulations on precursor chemicals, supply chain disruptions, and patent litigations could constrain supply and increase prices. The rise in illicit diversion for methamphetamine production poses continuous regulatory challenges.
Key Takeaways
- The pseudoephedrine ER market is constrained by strict regulations but remains essential for OTC cold remedies.
- Supply chain and manufacturing capacity are stable but susceptible to disruptions, affecting pricing.
- Prices are expected to trend upward modestly over the next five years, with potential for sharper increases if regulatory or supply shocks occur.
- Market concentration among top producers supports stable pricing but limits competition.
- Opportunities exist in emerging markets and product innovation, while regulatory risks persist.
FAQs
1. How does regulation influence pseudoephedrine ER prices?
Regulations restrict sales and raw material access, limiting supply and driving prices upward, especially during shortages.
2. What factors could accelerate price increases?
Stringent regulation, raw material scarcity, and supply chain disruptions can reduce supply and increase costs.
3. Are generic formulations cheaper than brand-name products?
Yes, generics typically cost 20-40% less due to lower development costs and increased market competition.
4. How does seasonal demand affect prices?
Prices tend to rise during winter months when cold and allergy cases increase, elevating demand for pseudoephedrine products.
5. What are the prospects for new ER formulations?
Innovation in delivery systems may command premium prices if they offer better efficacy or patient compliance, but development costs could also impact profitability.
References
[1] U.S. Drug Enforcement Administration. (2023). Pseudoephedrine regulations. DOJ.gov.
[2] MarketResearch.com. (2023). Global cold and allergy relief market report.
[3] IQVIA. (2022). Pharmaceutical market insights.
[4] Pfizer. (2022). Patent status and innovation reports.
[5] GlobalData. (2023). Chemical precursor supply and demand analysis.
More… ↓
