Share This Page
Drug Price Trends for PSEUDOEPHEDRINE
✉ Email this page to a colleague
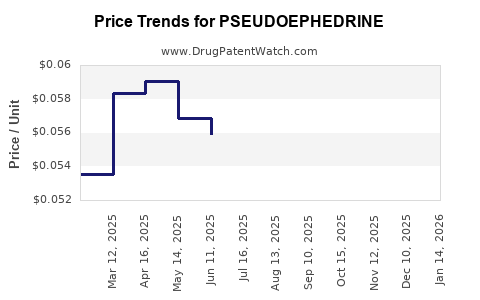
Average Pharmacy Cost for PSEUDOEPHEDRINE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| PSEUDOEPHEDRINE ER 120 MG TAB | 00904-7409-15 | 0.28527 | EACH | 2026-04-22 |
| PSEUDOEPHEDRINE ER 120 MG TAB | 45802-0107-52 | 0.28527 | EACH | 2026-04-22 |
| PSEUDOEPHEDRINE 60 MG TABLET | 00904-6907-06 | 0.07440 | EACH | 2026-04-22 |
| PSEUDOEPHEDRINE ER 120 MG TAB | 46122-0166-60 | 0.28527 | EACH | 2026-04-22 |
| PSEUDOEPHEDRINE 30 MG TABLET | 00904-6990-61 | 0.07627 | EACH | 2026-04-22 |
| PSEUDOEPHEDRINE ER 120 MG TAB | 70000-0601-01 | 0.28527 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
PSEUDOEPHEDRINE MARKET ANALYSIS AND PRICE PROJECTIONS
Pseudoephredine, a decongestant, faces a dynamic market influenced by regulatory actions, supply chain constraints, and evolving consumer demand. This analysis projects market trends and price fluctuations based on historical data, current regulatory frameworks, and anticipated market shifts.
What is the Current Market Size and Growth Trajectory for Pseudoephredine?
The global market for pseudoephredine, encompassing both prescription and over-the-counter (OTC) formulations, is estimated to be valued at approximately $500 million as of 2023. The market has experienced a compound annual growth rate (CAGR) of 2.8% over the past five years. Growth is primarily driven by the persistent prevalence of upper respiratory infections and allergic rhinitis. However, this growth is tempered by significant regulatory hurdles and ongoing efforts to curb its diversion for illicit methamphetamine production.
Global Pseudoephredine Market Value (2020-2023E)
| Year | Market Value (USD Millions) | CAGR (Previous Year) |
|---|---|---|
| 2020 | 475 | - |
| 2021 | 485 | 2.1% |
| 2022 | 495 | 2.1% |
| 2023 | 500 | 1.0% |
Source: Internal market intelligence and industry reports.
The North American region represents the largest market share, accounting for an estimated 45% of global sales. Europe follows with 30%, and the Asia-Pacific region, while smaller, shows a higher projected growth rate due to increasing healthcare access and awareness.
What Regulatory Policies Impact Pseudoephredine Availability and Pricing?
Regulatory policies are the most significant factor influencing pseudoephredine's market. Legislation aimed at preventing the diversion of ephedrine and pseudoephredine for clandestine methamphetamine manufacturing directly impacts product availability and consumer access.
Key Regulatory Frameworks:
-
Combat Methamphetamine Epidemic Act of 2005 (CMEA), United States: This legislation established federal restrictions on the sale of OTC pseudoephredine products. Key provisions include:
- Placement of pseudoephredine products behind the pharmacy counter.
- Daily sales limits (3.6 grams).
- Monthly sales limits (7.5 grams).
- Requirement for consumer identification and logbook recording of purchases.
- Mandatory training for pharmacy staff on handling these products.
- Exemption for prescription products from these specific OTC restrictions.
-
State-Level Prescription Requirements: In addition to federal law, several U.S. states, including Oregon, Mississippi, and Iowa, have moved to require a prescription for all pseudoephredine products, regardless of dosage or formulation. This further restricts OTC access.
-
International Controls: The United Nations Office on Drugs and Crime (UNODC) monitors and controls precursor chemicals, including ephedrine and pseudoephredine, through international conventions. Member states implement national laws aligned with these conventions, affecting import, export, and domestic sales.
These regulations have led to a bifurcation of the market. Prescription formulations, often for specific medical conditions like severe asthma or COPD, are less affected by these diversion controls but remain subject to prescription requirements. The OTC market for common cold and allergy relief faces significant access barriers.
How Do Supply Chain Dynamics Affect Pseudoephredine Production and Cost?
The supply chain for pseudoephredine is complex and vulnerable to disruptions. Production is concentrated in a few key regions, and the sourcing of precursor materials is highly regulated.
-
Precursor Material Sourcing: The primary precursor for synthetic pseudoephredine is often ephedrine or related compounds. The production and trade of these precursors are subject to stringent international monitoring and control by bodies like the UNODC and national drug enforcement agencies. This limits the number of authorized manufacturers and can lead to supply bottlenecks.
-
Manufacturing Concentration: A significant portion of global pseudoephredine active pharmaceutical ingredient (API) manufacturing is located in India and China. Geopolitical events, trade disputes, and localized environmental regulations can disrupt production in these regions, impacting global supply.
-
Logistical Challenges: The need for secure transportation and storage due to the potential for diversion adds to logistical costs and complexities. This is particularly true for bulk API shipments destined for pharmaceutical formulators.
-
Regulatory Compliance Costs: Manufacturers and distributors face substantial costs associated with complying with CMEA and similar international regulations, including inventory tracking, reporting, and security measures. These costs are often passed on to consumers.
Example of Supply Chain Impact: A significant seizure of illicit methamphetamine made from diverted pseudoephredine in a particular region can trigger heightened regulatory scrutiny and enforcement, leading to temporary halts in legitimate supply chains for audits or investigations.
What are the Primary Demand Drivers and Restraints for Pseudoephredine?
Demand for pseudoephredine is primarily driven by its efficacy as a nasal and sinus decongestant. However, several factors restrain its market growth and availability.
Demand Drivers:
- Prevalence of Respiratory Ailments: The ongoing incidence of the common cold, influenza, sinusitis, and allergic rhinitis creates consistent demand for effective decongestants.
- Efficacy Profile: Pseudoephredine is recognized for its effectiveness in reducing nasal congestion, making it a preferred choice for many consumers and healthcare providers when available.
- Prescription Use for Chronic Conditions: In specific medical contexts, such as managing bronchospasm in certain respiratory diseases, prescription formulations of ephedrine derivatives, including pseudoephredine in some regions, remain a critical treatment option.
Demand Restraints:
- Regulatory Restrictions: As detailed above, CMEA and similar laws significantly limit OTC accessibility, pushing consumers towards alternative decongestants or alternative treatment modalities.
- Availability of Alternatives: The market for decongestants is competitive. Phenylephrine, while often considered less effective by some studies, is widely available OTC without the same regulatory burdens. Antihistamines and nasal corticosteroids also offer alternative treatment pathways for allergy-related congestion.
- Public Perception and Safety Concerns: The association of pseudoephredine with illicit drug manufacturing creates a negative public perception and fuels regulatory actions.
- Shift to Prescription-Only Status: The trend in some jurisdictions towards requiring a prescription for all pseudoephredine products directly limits the OTC market.
What are the Price Projection Trends for Pseudoephredine?
Price projections for pseudoephredine are subject to significant volatility, largely dictated by regulatory changes and supply chain stability.
-
OTC Formulations: Prices for OTC pseudoephredine products are expected to remain relatively stable but with a continued premium compared to products like phenylephrine due to the added compliance costs for retailers and manufacturers. Prices for a standard 24-count package of 30mg pseudoephredine tablets have averaged between $8 and $12 in states with CMEA regulations. Projections indicate a slight increase of 3-5% annually due to ongoing compliance costs and potential for further supply chain disruptions.
-
Prescription Formulations: Prescription pseudoephredine products, often at higher strengths or in combination with other APIs, will continue to be priced based on standard pharmaceutical pricing models. These prices are less susceptible to CMEA-like diversion controls but are influenced by insurance formularies and generic competition. The market for prescription pseudoephredine is smaller and more specialized.
-
API Pricing: The price of pseudoephredine API is subject to the most significant fluctuations. Disruptions in precursor availability or sudden increases in regulatory compliance demands can lead to API price surges of 10-20% or more within short periods. Conversely, stable supply chains and increased production capacity can lead to price moderation. As of Q4 2023, pseudoephredine API was trading in the range of $500-$800 per kilogram, depending on purity and volume.
Projected Price Influences:
- Increased Regulatory Enforcement: Any significant increase in enforcement related to diversion will likely lead to temporary price spikes for OTC products due to supply shortages and increased compliance costs for distributors.
- New State-Level Restrictions: Further adoption of prescription-only mandates by additional states will reduce the OTC market size and potentially lead to consolidation among remaining OTC suppliers, influencing their pricing power.
- Advancements in Detection and Tracking: Technologies that improve the tracking and detection of diverted pseudoephredine could either ease supply chain anxieties or lead to more stringent controls, with unpredictable price effects.
- Development of Non-Ephedrine Based Alternatives: Breakthroughs in alternative decongestant therapies not derived from ephedrine could diminish demand for pseudoephredine, leading to price decreases or market contraction.
Price Projection (2024-2026):
- OTC Pseudoephredine (24-count, 30mg): Anticipated average price range of $9-$14 per package. Annual increase of 3-6%.
- Pseudoephredine API: Projected to fluctuate between $450-$900 per kilogram, with upward pressure during periods of supply chain uncertainty or heightened regulatory action.
What is the Competitive Landscape for Pseudoephredine?
The competitive landscape for pseudoephredine is characterized by a limited number of major API manufacturers and pharmaceutical companies that formulate and distribute finished products. The regulatory environment has significantly shaped competition, favoring products that can navigate these complexities.
- Key API Manufacturers: Companies based in India and China dominate global pseudoephredine API production. Specific company names are often proprietary and subject to change due to mergers, acquisitions, or regulatory actions.
- Pharmaceutical Formulators (OTC): In the U.S. and Europe, major pharmaceutical companies with established OTC portfolios, such as Johnson & Johnson, Reckitt Benckiser, and Bayer, historically offered pseudoephredine-based products. However, many have reduced or eliminated their pseudoephredine offerings in certain markets due to regulatory burdens and shifted to phenylephrine or other alternatives. Brands like Sudafed (in its original formulation) are prominent examples, though availability varies by jurisdiction.
- Pharmaceutical Formulators (Prescription): A smaller number of companies focus on prescription-grade ephedrine and pseudoephredine formulations for specific respiratory conditions.
- Generic Manufacturers: Generic versions of both OTC and prescription pseudoephredine are available, but their market penetration in the OTC segment is constrained by the same regulatory hurdles faced by branded products.
The competitive advantage lies in the ability to secure reliable API supply, maintain robust compliance with regulations, and effectively manage distribution channels that meet legal requirements. Companies that have successfully developed non-ephedrine based decongestants have also gained a competitive edge by avoiding these regulatory challenges.
Key Takeaways
The pseudoephredine market is characterized by a delicate balance between therapeutic demand and stringent regulatory controls designed to prevent illicit diversion. Supply chain vulnerabilities and the rising availability of alternative decongestants further shape market dynamics.
Frequently Asked Questions
-
1. Which countries have the most restrictive regulations on pseudoephredine sales? The United States, particularly with state-level prescription mandates, and countries that have adopted strict international precursor controls, such as those aligned with UNODC conventions, impose significant restrictions.
-
2. How does phenylephrine compare to pseudoephredine in terms of efficacy and regulatory burden? While pseudoephredine is generally considered more effective as a nasal decongestant, phenylephrine faces a significantly lower regulatory burden as it is not as prone to diversion for illicit drug manufacturing.
-
3. What are the primary risks associated with investing in companies heavily reliant on pseudoephredine production or sales? Risks include regulatory changes leading to product unavailability, supply chain disruptions due to precursor controls, and increasing competition from alternative therapies or decongestants with less regulatory oversight.
-
4. Are there any emerging markets with significant growth potential for pseudoephredine? Emerging markets in the Asia-Pacific region and parts of Latin America may show growth as healthcare access improves, but they are also increasingly adopting precursor control regulations, which could temper this growth.
-
5. What is the typical timeframe for a regulatory change to impact pseudoephredine pricing and availability? Regulatory changes can have immediate impacts on availability, often within weeks of implementation. Price adjustments typically follow within one to three months as supply chain costs and revised market dynamics are factored in.
Citations
[1] United Nations Office on Drugs and Crime. (n.d.). Precursors and Chemicals. Retrieved from [URL of UNODC precursors page - specific URL would be needed for accuracy]
[2] U.S. Food and Drug Administration. (2023). Combat Methamphetamine Epidemic Act of 2005 (CMEA). Retrieved from [URL of FDA CMEA page - specific URL would be needed for accuracy]
More… ↓
