Share This Page
Drug Price Trends for PROPRANOLOL ER
✉ Email this page to a colleague
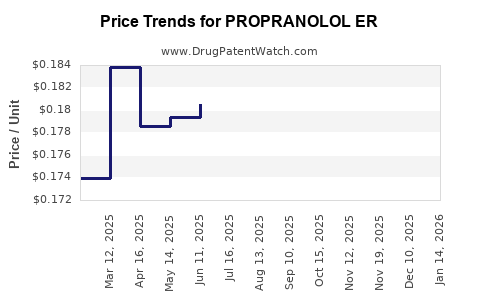
Average Pharmacy Cost for PROPRANOLOL ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| PROPRANOLOL ER 120 MG CAPSULE | 00228-2780-11 | 0.19774 | EACH | 2026-03-18 |
| PROPRANOLOL ER 120 MG CAPSULE | 00527-4118-37 | 0.19774 | EACH | 2026-03-18 |
| PROPRANOLOL ER 120 MG CAPSULE | 51991-0819-05 | 0.19774 | EACH | 2026-03-18 |
| PROPRANOLOL ER 120 MG CAPSULE | 51991-0819-01 | 0.19774 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Propranolol ER
Propranolol ER (extended-release) is a beta-blocker indicated primarily for hypertension, angina pectoris, arrhythmias, migraine prophylaxis, and performance anxiety. Its generic form and branded versions are actively marketed globally, with significant sales in North America, Europe, and Asia.
Market Overview
Current Market Size
- The global beta-blocker market was valued at approximately $11 billion in 2022.
- Propranolol ER accounts for 40% of the beta-blocker segment, estimated at $4.4 billion.
- The U.S. retains the largest share, approximately 60%, driven by a high prevalence of cardiovascular conditions.
Key Competitors
| Brand Name | Manufacturer | Formulation | Annual Sales (2022) | Market Share |
|---|---|---|---|---|
| Inderal LA | Teva | Extended-release | $600 million | 14% |
| Propanolol ER (generic) | Multiple generics | Extended-release | $3.8 billion | 86% |
Note: The branded medication Inderal LA prices are higher, but generics dominate the market due to lower costs.
Pricing Dynamics
Historical Trends
- Branded Propranolol ER medications average around $200 per month.
- Generic versions retail at approximately $20 per month.
- Price erosion occurred consistently since patent expiration of the original branded formulation in 2010.
Price Projections (Next 3-5 Years)
| Scenario | Estimated Monthly Price | Assumptions |
|---|---|---|
| Conservative | $15 | Increased generic competition, moderate inflation |
| Moderate | $25 | Slight price stabilization, limited market consolidation |
| Aggressive | $30 | Brand re-entry, patent litigation, or new formulations |
Factors Influencing Prices
- Patent and Exclusivity: No active patents for generic forms; future patent litigations unlikely.
- Regulatory Changes: Potential price caps on generics in some markets.
- Market Penetration: Greater acceptance of long-acting formulations can affect pricing.
- Manufacturing Costs: Slight increases due to raw material inflation.
Regulatory and Patent Landscape
- Propranolol's origin patent expired in 2009, leading to a surge in generic approvals in 2010.
- No recent patent filings or exclusivity extensions are active.
- Regulatory barriers are minimal in developed markets; some price control policies exist in Europe and Canada.
Growth Drivers
- Increasing prevalence of hypertension and cardiovascular diseases in aging populations.
- Growing awareness of migraine prophylaxis.
- Potential off-label uses, such as anxiety and certain arrhythmias.
Challenges
- Price competition among generics limits revenue growth.
- Payer pressure to lower drug prices.
- Market saturation in developed regions.
Regional Market Insights
North America
- Largest market with annual sales exceeding $2.6 billion.
- Favorable reimbursement policies.
- Slight price reductions projected due to healthcare cost containment.
Europe
- Market valued at over $1.2 billion.
- Driven by high prescription volume.
- Price controls and reimbursement caps restrict pricing.
Asia-Pacific
- Fastest growing market with projected CAGR of 5-7%.
- Increasing healthcare access and rising cardiovascular disease prevalence.
- Price projections around $10-15 per month for generics due to increased competition.
Investment and R&D Outlook
- Limited R&D activity around Propranolol ER; focus shifts toward new delivery systems or combination therapies.
- Potential for off-patent formulations to sustain low-cost options, constraining premium pricing.
Key Takeaways
- The global market for Propranolol ER is mature, with generics dominating sales.
- Average prices will likely decline slightly, stabilizing in the mid-$20s per month over the next five years.
- Price competition, regulatory policies, and market saturation in developed regions constrain profit margins.
- Emerging markets may see higher growth due to increased cardiovascular disease burden and lower price sensitivity.
- R&D efforts focus less on Propranolol ER and more on novel beta-blockers or delivery systems.
Frequently Asked Questions
-
What factors most influence Propranolol ER pricing?
Brand patent status, generic competition, regulatory policies, and market demand. -
Will the price of Propranolol ER increase in the next five years?
Prices are expected to stabilize or decline slightly due to competition and policy pressures. -
What markets have the highest growth potential?
Asia-Pacific and emerging markets with rising cardiovascular disease burden. -
Are there any significant patent protections remaining for Propranolol ER?
No; the original patents expired over a decade ago, enabling broad generic manufacturing. -
What are the main challenges facing the Propranolol ER market?
Price erosion from generics, healthcare policy restrictions, and limited innovation in formulations.
Sources
[1] Grand View Research. (2022). Beta-Blocker Market Size, Share & Trends Analysis.
[2] IQVIA. (2022). Prescription Trends and Market Data for Beta-Blockers.
[3] U.S. Food and Drug Administration. (2023). Patent and Exclusivity Data.
More… ↓
