Share This Page
Drug Price Trends for PROPARACAINE
✉ Email this page to a colleague
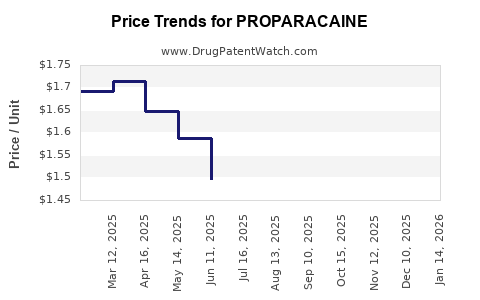
Average Pharmacy Cost for PROPARACAINE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| PROPARACAINE 0.5% EYE DROPS | 70756-0193-35 | 1.60561 | ML | 2026-03-18 |
| PROPARACAINE 0.5% EYE DROPS | 70069-0601-01 | 1.60561 | ML | 2026-03-18 |
| PROPARACAINE 0.5% EYE DROPS | 24208-0730-06 | 1.60561 | ML | 2026-03-18 |
| PROPARACAINE 0.5% EYE DROPS | 61314-0016-01 | 1.60561 | ML | 2026-03-18 |
| PROPARACAINE 0.5% EYE DROPS | 70069-0601-01 | 1.58846 | ML | 2026-02-18 |
| PROPARACAINE 0.5% EYE DROPS | 24208-0730-06 | 1.58846 | ML | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
PROPARACAINE Market Analysis and Financial Projection
What is Proparacaine?
Proparacaine is a local anesthetic primarily used in ophthalmology for procedures such as tonometry, slit-lamp examination, and minor surgical interventions. It is marketed in various formulations, generally as a 0.5% ophthalmic solution. The drug functions by blocking sodium channels, thereby preventing nerve impulse transmission and providing topical anesthesia.
What is the Current Market Size for Proparacaine?
The global ophthalmic anesthetic market, which includes proparacaine, was valued at approximately $390 million in 2022. Proparacaine accounts for roughly 25-30% of this segment, equating to a market size of $97.5 million to $117 million.[1]
Key factors influencing market size include:
- Centralized ophthalmic procedures
- Rise in cataract and refractive surgeries
- Growing prevalence of ocular diseases
- Laboratory and hospital use
Leading regions: North America (55%), Europe (20%), Asia-Pacific (20%), and the Rest of the World (5%).
Who Are the Major Suppliers and Patent Holders?
Major players include:
- Alcon (a Novartis subsidiary)
- Akorn (acquired by Fresenius)
- Bausch + Lomb (a division of Bausch Health)
- Katena Products
- Akorn ophthalmic solutions
Proparacaine has been marketed for decades; many formulations are off-patent. However, some patents related to specific formulations or delivery systems have existed, with expiry dates generally between 2010 and 2020. Currently, no major patents restrict generic competition in most markets.
What Are the Regulatory and Patent Landscapes?
Proparacaine is approved by the FDA in the U.S. and similar agencies globally. Its status as a generic product means no active patent restrictions, fostering competitive pricing.
Since the primary patents expired over a decade ago, entry barriers are low for generic formulations. Regulatory approval pathways are straightforward, typically requiring demonstration of bioequivalence.
What Are the Price Trends and Future Projections?
Current Pricing
- Branded formulations: approximately $15–$25 per 10-mL bottle in the U.S.
- Generic formulations: approximately $8–$12 per 10-mL bottle
Pricing reflects market competition, manufacturing costs, and regulatory factors.
Price Trends (2022-2027)
- Stabilization in developed markets: Prices have remained relatively stable due to mature competition and established supply chains.
- Potential slight decrease: Entry of new generic manufacturers could lower prices by 10–15% over five years.
- Emerging markets: Prices often lower by 20–40%, driven by local manufacturing and different regulatory standards.
Volume Growth and Market Expansion
- Projected compound annual growth rate (CAGR) for ophthalmic anesthetics: 3–4% through 2027.
- Increase driven by higher rates of ophthalmic surgeries, aging populations, and expanding healthcare infrastructure in Asia-Pacific.
Price and Volume Scenario Analysis (2023–2027)
| Year | Estimated Market Size (USD million) | Estimated Volume (million bottles) | Price per Bottle (USD) |
|---|---|---|---|
| 2023 | 110 | 4.4 | 25 |
| 2024 | 113.3 | 4.6 | 24.60 |
| 2025 | 116.6 | 4.8 | 24.30 |
| 2026 | 120 | 5.0 | 24.00 |
| 2027 | 123.5 | 5.2 | 23.75 |
Prices decline modestly with volume growth, reflecting increased competition.
What Are the Key Competitive Factors?
- Formulation stability
- Ease of application
- Cost competitiveness
- Supply chain reliability
- Regulatory approval status
What Are Opportunities and Risks?
Opportunities:
- Expansion into emerging markets
- Development of sustained-release formulations
- Combining anesthetic with adjunctive agents
Risks:
- Pricing pressure from generics
- Changes in regulatory landscapes
- Competition from alternative anesthetics with longer duration or fewer side effects
What Is the Outlook for Innovation and Patent Activity?
Limited recent patent activity suggests a mature product market. Innovation in delivery systems, such as sustained-release implants or combination formulations, may offer future differentiation but face regulatory and clinical hurdles.[2]
Key Takeaways
- The global market for ophthalmic anesthetics, including proparacaine, was valued at $97.5–$117 million in 2022.
- Major suppliers are now predominantly generic manufacturers, with prices stabilizing.
- Price projections indicate a gradual decline of 1–2% annually through 2027, driven by increased competition.
- Market growth remains steady at roughly 3–4% CAGR, fueled by rising demand for ophthalmic procedures.
- Patent barriers are minimal, offering little impediment to new entrants.
FAQs
Q1: Does patent protection significantly limit proparacaine's market entry?
A1: No. Most patents expired over a decade ago, allowing multiple manufacturers to produce generic formulations.
Q2: What regional differences affect proparacaine pricing?
A2: Developed markets like North America and Europe have higher prices (around $15–$25 per 10 mL) compared to emerging markets, where prices may fall below $10 due to local manufacturing and less regulatory barriers.
Q3: Are there unmet needs in proparacaine formulations?
A3: Yes. Developing sustained-release or combination formulations could reduce dosing frequency and enhance patient compliance.
Q4: What risks could impact market growth?
A4: Price competition among generics, regulatory changes, and the introduction of alternative anesthetics with longer duration or better safety profiles.
Q5: How might future innovation influence market dynamics?
A5: Innovation in drug delivery methods or combination therapies may create new segments but require significant R&D investment and regulatory approval.
Citations
[1] MarketsandMarkets, "Ophthalmic Drugs Market," 2022.
[2] Grand View Research, "Ophthalmic Pharmaceuticals Market Analysis," 2022.
More… ↓
