Share This Page
Drug Price Trends for PROMETRIUM
✉ Email this page to a colleague
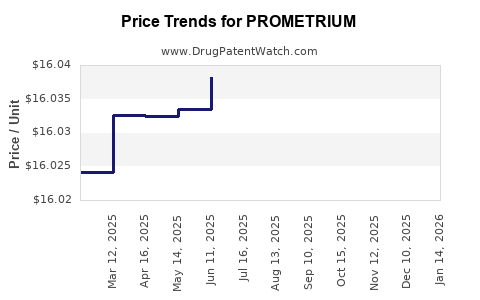
Average Pharmacy Cost for PROMETRIUM
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| PROMETRIUM 200 MG CAPSULE | 72989-0373-30 | 30.38679 | EACH | 2026-04-22 |
| PROMETRIUM 100 MG CAPSULE | 72989-0372-30 | 16.01755 | EACH | 2026-04-22 |
| PROMETRIUM 200 MG CAPSULE | 72989-0373-30 | 30.32705 | EACH | 2026-03-18 |
| PROMETRIUM 100 MG CAPSULE | 72989-0372-30 | 16.04409 | EACH | 2026-03-18 |
| PROMETRIUM 200 MG CAPSULE | 72989-0373-30 | 30.28539 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for PROMETRIUM
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| PROMETRIUM 100 MG CAPSULE | Acertis Pharmaceuticals, LLC | 72989-0372-30 | 30 | 260.48 | 8.68267 | EACH | 2023-09-29 - 2028-09-28 | FSS |
| PROMETRIUM 100 MG CAPSULE | Acertis Pharmaceuticals, LLC | 69543-0372-30 | 30 | 260.48 | 8.68267 | EACH | 2023-09-29 - 2028-09-28 | FSS |
| PROMETRIUM 100 MG CAPSULE | Acertis Pharmaceuticals, LLC | 72989-0372-30 | 30 | 198.49 | 6.61633 | EACH | 2024-01-01 - 2028-09-28 | FSS |
| PROMETRIUM 100 MG CAPSULE | Acertis Pharmaceuticals, LLC | 69543-0372-30 | 30 | 198.49 | 6.61633 | EACH | 2024-01-01 - 2028-09-28 | FSS |
| PROMETRIUM 200 MG CAPSULE | Acertis Pharmaceuticals, LLC | 72989-0373-30 | 30 | 494.83 | 16.49433 | EACH | 2023-09-29 - 2028-09-28 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
PROMETRIUM Market Analysis and Price Projections
PROMETRIUM (progesterone) is a progesterone hormone replacement therapy used to treat conditions such as irregular menstruation, abnormal uterine bleeding, and amenorrhea. It is also used in combination with conjugated estrogens for hormone replacement therapy to treat menopausal symptoms. The market for PROMETRIUM is influenced by factors including its therapeutic applications, competition from alternative treatments, and patent expirations.
What is the Current Market Landscape for PROMETRIUM?
The global market for progesterone hormone replacement therapy, including PROMETRIUM, is characterized by established use in gynecological treatments and hormone therapy. Key market segments include the treatment of infertility, menopausal symptom management, and gynecological disorders. The demand is driven by an aging female population experiencing menopause and a rising prevalence of reproductive health issues.
-
Therapeutic Applications:
- Menopause Management: PROMETRIUM is often prescribed as part of hormone replacement therapy (HRT) to alleviate menopausal symptoms like hot flashes, night sweats, and vaginal dryness. This segment is substantial due to the significant number of women entering or in menopause.
- Gynecological Disorders: It addresses conditions such as irregular menstruation, abnormal uterine bleeding, and amenorrhea, which are common reproductive health concerns.
- Infertility Treatment: Progesterone is crucial for supporting pregnancy. PROMETRIUM and other progesterone formulations are used in assisted reproductive technologies (ART) and for women with luteal phase defects.
-
Key Market Drivers:
- Aging Population: The global increase in life expectancy leads to a larger population of postmenopausal women, driving demand for HRT.
- Rising Prevalence of Gynecological Issues: Factors like lifestyle changes and increasing awareness contribute to a higher diagnosis rate of conditions requiring progesterone therapy.
- Growth in ART: The expanding market for in vitro fertilization (IVF) and other fertility treatments directly boosts demand for progesterone.
-
Market Size and Growth: The global progesterone market was valued at approximately $1.7 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of around 5-7% from 2023 to 2030 [1]. PROMETRIUM, as a branded product, holds a significant share within this market.
Who are the Primary Competitors to PROMETRIUM?
PROMETRIUM faces competition from both generic progesterone products and alternative treatment modalities. The competitive landscape is dynamic, with new formulations and therapies emerging.
-
Generic Progesterone Products:
- Numerous generic manufacturers offer bioequivalent progesterone capsules and suppositories. These products often compete on price, impacting PROMETRIUM's market share, particularly after patent expirations. Examples include products from Mylan, Teva, and Sun Pharmaceutical Industries.
- Price Difference: Generic versions are typically priced 20-50% lower than branded PROMETRIUM, making them a preferred option for cost-sensitive healthcare systems and patients.
-
Other Branded Progesterone Products:
- CrinoGest® (Progesterone): While less prevalent in the US market, other branded progesterone products exist globally, offering similar therapeutic profiles.
- Vaginal Progesterone Gels and Inserts: Formulations specifically designed for vaginal administration are gaining traction for HRT and infertility, offering localized delivery and potentially fewer systemic side effects.
-
Alternative Treatment Modalities:
- Non-Hormonal Therapies for Menopause: For menopausal symptoms, non-hormonal options like SSRIs, SNRIs, and gabapentin are gaining traction, particularly for women with contraindications to HRT.
- Other Estrogen Therapies: Different forms of estrogen, including transdermal patches and gels, are used in HRT, often in combination with progesterone.
- Management of Gynecological Disorders: Treatment strategies for abnormal uterine bleeding and irregular menstruation can also include non-hormonal medications (e.g., tranexamic acid, oral contraceptives) or surgical interventions.
What is the Patent Status of PROMETRIUM and its Impact on the Market?
The patent landscape for PROMETRIUM is critical to understanding its pricing and market exclusivity. The original patents for PROMETRIUM have expired, leading to the entry of generic competition.
- Original Patent Expiration: The key patents protecting the original formulation of PROMETRIUM have expired. The primary patents were granted in the late 1980s and early 1990s [2].
- Generic Entry: Following patent expiry, multiple generic manufacturers have launched their progesterone products. This has led to a significant price reduction in the overall progesterone market.
- Potential for New Patents: While the foundational patents have expired, there could be patents related to specific manufacturing processes, new formulations, or novel therapeutic uses of progesterone that might offer some degree of extended protection or market differentiation for certain products. However, for the established PROMETRIUM formulation, generic erosion is the dominant factor.
- Impact on Pricing: The influx of generic competitors has driven down the average selling price of progesterone. Branded PROMETRIUM can still command a premium due to brand recognition and established physician trust, but its market share and pricing power are significantly constrained by generics.
What are the Projected Price Trends for PROMETRIUM?
Price projections for PROMETRIUM are heavily influenced by the sustained competition from generic alternatives and the overall market dynamics of hormone replacement therapy.
-
Current Pricing Environment:
- Branded PROMETRIUM: A typical prescription of 90 capsules (100mg) of branded PROMETRIUM can range from $150 to $300 USD or higher, depending on the pharmacy and insurance coverage [3].
- Generic Progesterone: The same quantity of generic progesterone can cost between $20 and $70 USD [3]. This represents a substantial price differential.
-
Projected Price Trends (Next 5-10 Years):
- Continued Price Erosion for Branded PROMETRIUM: While PROMETRIUM may retain a segment of the market due to brand loyalty and physician preference, its price is expected to continue declining gradually. This erosion will be driven by increasing market share of generics and pressure from payers to reduce healthcare costs. A modest annual decline of 2-5% for the branded product is anticipated.
- Stable to Declining Generic Prices: Generic progesterone prices are expected to remain competitive and may even see slight further reductions as more manufacturers enter the market or as economies of scale are achieved. Price stability or a slight decrease of 0-2% annually is projected for generics.
- Influence of Payer Policies: Insurance formularies and government healthcare programs will continue to exert downward pressure on prices. Preferred placement on formularies for generics will accelerate the shift away from branded options.
- Innovation vs. Generics: Unless there is a significant patentable innovation in a new PROMETRIUM formulation or delivery method that offers distinct advantages, the pricing trend will remain dominated by generic competition.
-
Factors Affecting Price Projections:
- Volume of Generic Penetration: The higher the market share captured by generics, the greater the downward pressure on branded prices.
- Manufacturing Costs: Fluctuations in the cost of raw materials and production can influence base pricing for all progesterone products.
- Regulatory Environment: Changes in FDA regulations or pricing oversight could impact market dynamics.
- Physician Prescribing Habits: A continued reliance on established brands by physicians could slow generic uptake, but this trend is weakening.
What is the Future Market Outlook for PROMETRIUM?
The future market outlook for PROMETRIUM is one of mature product status facing significant generic competition. Its role is likely to become increasingly niche, focused on specific patient populations or situations where brand loyalty or physician preference outweighs cost considerations.
-
Market Segmentation:
- Menopausal Hormone Therapy: This segment will continue to see pressure from both generics and non-hormonal alternatives. PROMETRIUM's share will likely decrease as cost-effectiveness becomes a more dominant factor.
- Infertility Treatments: This area might offer more stable demand for progesterone, given its critical role in ART. However, even here, cost pressures will encourage the use of generics.
- Gynecological Disorders: Treatment protocols for these conditions are evolving, and PROMETRIUM will compete with other pharmaceutical interventions and potentially minimally invasive procedures.
-
Challenges:
- Generic Competition: This remains the most significant challenge, eroding market share and limiting pricing power.
- Safety Concerns and Alternatives: Lingering concerns about the long-term risks associated with HRT, although often debated and specific to formulations, can influence prescribing patterns and drive adoption of alternatives.
- Cost Containment: Healthcare systems globally are focused on reducing drug expenditures, favoring generics and lower-cost alternatives.
-
Opportunities:
- Established Efficacy: PROMETRIUM has a long history of use and a well-understood efficacy profile, which can be leveraged in specific patient cases.
- Physician Familiarity: Long-term physician familiarity with the drug can maintain some level of prescribing.
- Potential for New Indications (Low Probability): While unlikely for an older drug, discovery of new, patentable indications for progesterone could theoretically revive market interest, though this is speculative.
-
Overall Outlook: The market for PROMETRIUM is expected to experience a slow decline in volume and a more significant decline in revenue over the next decade. Its position will likely shift from a primary branded option to one of several progesterone choices, with generics dominating the volume and driving overall market price trends.
Key Takeaways
- PROMETRIUM's market is mature, driven by its use in menopausal symptom management, gynecological disorders, and infertility treatments.
- The primary competitive threat comes from a large number of generic progesterone products that offer significant cost savings.
- Original patents for PROMETRIUM have expired, leading to widespread generic availability and intense price competition.
- Price projections indicate continued gradual decline for branded PROMETRIUM and stable to declining prices for generic progesterone, largely due to payer policies and market competition.
- The future market outlook for PROMETRIUM is characterized by a shrinking market share and an increasingly challenging pricing environment, with generics set to dominate volume.
Frequently Asked Questions
1. What is the primary driver for the price difference between branded PROMETRIUM and its generic counterparts?
The primary driver for the price difference is the expiration of patents held by the original manufacturer of PROMETRIUM. This allows multiple pharmaceutical companies to produce and sell generic versions of progesterone, leading to increased supply and competitive pricing, significantly undercutting the branded product's price.
2. How do payer policies influence PROMETRIUM's market price and availability?
Payer policies, such as insurance formulary placement and preferred drug lists, strongly influence PROMETRIUM's market. Insurers often prioritize generics due to their lower cost, requiring higher co-pays or prior authorization for branded PROMETRIUM, thereby limiting its prescribing volume and reinforcing downward price pressure.
3. Are there any new formulations or therapeutic uses for PROMETRIUM that could impact its future market position?
While the original PROMETRIUM formulation faces generic competition, research into novel drug delivery systems or specific therapeutic applications for progesterone could emerge. However, for the established PROMETRIUM product, significant innovation that would dramatically alter its market trajectory and pricing power is not currently anticipated. Existing R&D focuses more on next-generation hormone therapies or alternative treatments.
4. What is the typical prescription volume for PROMETRIUM annually?
Estimating precise annual prescription volumes for branded PROMETRIUM is challenging as this data is proprietary and fluctuates. However, given the market size of progesterone therapeutics, branded PROMETRIUM likely accounts for millions of prescriptions annually in key markets like the United States, though this volume is steadily being eroded by generics.
5. What is the expected impact of the Women's Health Initiative (WHI) study on the current and future market for PROMETRIUM?
The Women's Health Initiative (WHI) study, published in 2002, raised safety concerns regarding hormone replacement therapy (HRT), leading to a significant decrease in HRT use. While subsequent analyses have provided a more nuanced understanding of HRT risks and benefits, the study's initial findings influenced physician prescribing habits and patient perceptions, contributing to a more cautious approach to HRT and a greater emphasis on non-hormonal alternatives or individualized risk-benefit assessments. This caution continues to influence the market for PROMETRIUM, particularly in the menopausal symptom management segment, by encouraging consideration of alternatives and potentially limiting demand for HRT.
Citations
[1] Grand View Research. (2023). Progesterone Market Size, Share & Trends Analysis Report By Product (Micronized Progesterone, Synthetic Progesterone), By Application (Menopause Management, Contraceptive, Infertility Treatment, Gynecological Disorders), By Route Of Administration (Oral, Injectable, Topical, Vaginal), By Region, And Segment Forecasts, 2023 - 2030.
[2] U.S. Food and Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from https://www.fda.gov/drugs/drug-approvals-and-databases/orange-book-approved-drug-products-therapeutic-equivalence-evaluations
[3] GoodRx. (n.d.). Progesterone Prices, Coupons, and Patient Assistance Programs. Retrieved from https://www.goodrx.com/progesterone
More… ↓
