Share This Page
Drug Price Trends for PROMETHAZINE-DM
✉ Email this page to a colleague
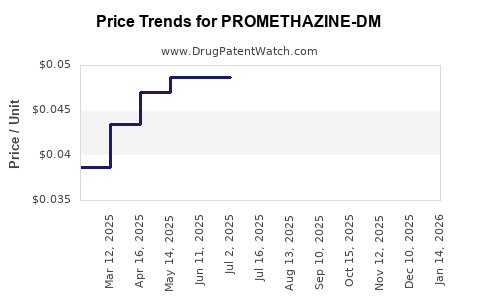
Average Pharmacy Cost for PROMETHAZINE-DM
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| PROMETHAZINE-DM 6.25-15 MG/5 ML SOLUTION | 62559-0758-16 | 0.03887 | ML | 2026-04-22 |
| PROMETHAZINE-DM 6.25-15 MG/5 ML SOLUTION | 00713-0780-75 | 0.03887 | ML | 2026-04-22 |
| PROMETHAZINE-DM 6.25-15 MG/5 ML SOLUTION | 27808-0057-01 | 0.03887 | ML | 2026-04-22 |
| PROMETHAZINE-DM 6.25-15 MG/5 ML SYRUP | 00116-4024-16 | 0.03887 | ML | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Promethazine-DM: Market Landscape and Price Projections
Market Overview
Promethazine-DM is a combination medication comprising promethazine hydrochloride, an antihistamine, and dextromethorphan hydrobromide, a cough suppressant. It is primarily indicated for the temporary relief of cough and upper respiratory symptoms associated with the common cold or allergies. The market for cough and cold remedies is substantial, driven by seasonal demand and the prevalence of respiratory illnesses. Promethazine-DM competes within this broad category, facing competition from single-ingredient products and other multi-symptom formulations.
The primary active ingredients, promethazine and dextromethorphan, have long histories of use and are available as generics, contributing to price competition. Regulatory scrutiny of cough and cold medications, particularly regarding their use in pediatric populations, has influenced market dynamics. The U.S. Food and Drug Administration (FDA) has issued advisories on the use of certain cough and cold products in children under two years old, though Promethazine-DM is typically not recommended for this age group.
Market size for the broader cough and cold segment is influenced by factors such as seasonal variations in illness, public health campaigns, and the availability of over-the-counter (OTC) and prescription options. The global cough and cold remedies market was valued at approximately USD 50 billion in 2023 and is projected to grow at a compound annual growth rate (CAGR) of 3% to 4% over the next five years, according to industry reports. Promethazine-DM's specific market share within this segment is difficult to isolate due to its classification as a combination drug and the presence of numerous generic alternatives for its individual components.
Competitive Landscape
Promethazine-DM operates in a highly fragmented market characterized by a significant number of generic manufacturers and a wide array of product offerings. Its key competitors can be categorized as follows:
- Direct Combination Competitors: Other formulations that combine an antihistamine with a cough suppressant, or other symptomatic relief agents like decongestants or expectorants. Examples include products containing diphenhydramine and dextromethorphan, or chlorpheniramine and dextromethorphan.
- Single-Ingredient Competitors:
- Antihistamines: Products containing only promethazine (prescription strength) or other first-generation antihistamines like diphenhydramine or chlorpheniramine, often used for allergy symptoms.
- Cough Suppressants: Products containing only dextromethorphan, which is widely available OTC in various formulations (syrups, lozenges, capsules).
- Other Cough and Cold Remedies: Broader spectrum products that may include decongestants (e.g., phenylephrine, pseudoephedrine), expectorants (e.g., guaifenesin), and pain relievers (e.g., acetaminophen, ibuprofen).
The competitive advantage for Promethazine-DM formulations typically rests on brand recognition, specific dosing convenience, and marketing. However, the availability of inexpensive generic alternatives for both promethazine and dextromethorphan significantly pressures pricing across the entire market. The market entry barriers are relatively low for generic cough and cold formulations.
Major pharmaceutical companies and numerous smaller generic manufacturers produce products containing either promethazine or dextromethorphan, or combinations thereof. Key players in the broader cough and cold market include Sanofi, GlaxoSmithKline, Bayer, and numerous generic pharmaceutical firms such as Teva Pharmaceuticals, Mylan (now Viatris), and Amneal Pharmaceuticals.
Pricing Dynamics and Projections
The pricing of Promethazine-DM is predominantly influenced by its generic status and the competitive landscape. Both promethazine and dextromethorphan are widely available as low-cost generic active pharmaceutical ingredients (APIs).
Current Pricing Factors:
- Generic Equivalents: The vast majority of Promethazine-DM products available are generic. This leads to intense price competition among manufacturers.
- API Costs: The cost of promethazine hydrochloride and dextromethorphan hydrobromide APIs are relatively stable and low, further contributing to affordable finished product pricing.
- Formulation: While the active ingredients are inexpensive, minor variations in excipients, flavoring, or delivery systems (e.g., syrup vs. capsules) can create slight price differences, but these are generally marginal.
- Distribution Channels: Pricing varies based on whether the product is sold through retail pharmacies (OTC), wholesale distributors, or directly to healthcare providers for prescription use.
- Insurance Reimbursement: For prescription formulations, insurance formularies and co-pay structures influence out-of-pocket costs for patients. However, many formulations are also available OTC.
- Regulatory Compliance: Costs associated with Good Manufacturing Practices (GMP), packaging, and labeling contribute to the overall manufacturing cost.
Historical Price Trends:
Over the past decade, the average wholesale price (AWP) for generic cough and cold medications, including formulations containing promethazine and dextromethorphan, has remained relatively stable, with slight downward pressure due to competition. For instance, the average AWP for a standard 4-ounce bottle of generic promethazine DM syrup has historically ranged from $5 to $15. Prescription versions, especially those in higher concentrations or specific formulations, might command higher prices, but the bulk of the market is driven by OTC generics.
Price Projections:
Based on current market dynamics, the pricing for Promethazine-DM is projected to remain stable with a slight downward trend in real terms over the next 3-5 years.
- Short-term (1-2 years): Expect minimal price fluctuations. Retail prices for generic Promethazine-DM are likely to continue to range from $5 to $20 for standard OTC formulations, depending on the retailer and package size. Prescription strengths may see minor adjustments based on payer negotiations but are unlikely to experience significant inflation.
- Mid-term (3-5 years): Continued competitive pressure from generic manufacturers will likely maintain price stability or lead to a marginal decrease in real prices. No significant price increases are anticipated unless there are unforeseen supply chain disruptions or major regulatory changes impacting the production or availability of the active ingredients.
Key factors influencing future pricing:
- Generic Competition: The continued influx of generic products will be the primary driver of price moderation.
- Manufacturing Costs: Stable or slightly decreasing costs for APIs and manufacturing processes will support current pricing.
- Regulatory Landscape: Any new regulations concerning the safety or efficacy of antihistamines or cough suppressants could impact market access and pricing, but major shifts are not currently anticipated.
- Demand: While seasonal demand spikes occur, overall demand for these types of symptom relievers remains consistent, preventing significant price hikes due to surge pricing.
Example Pricing Scenario (OTC Syrup, 4 fl oz):
| Year | Projected Average Retail Price (USD) | Notes |
|---|---|---|
| 2024 | $7.50 - $15.00 | Reflects current generic market variability. |
| 2025 | $7.00 - $14.50 | Slight downward pressure due to ongoing competition. |
| 2026 | $7.00 - $14.00 | Continued price stability. |
| 2027 | $6.80 - $13.80 | Marginal decrease anticipated due to sustained generic pressure. |
| 2028 | $6.80 - $13.50 | Continued market dynamics, slight inflation for specific SKUs possible. |
Note: These projections are for standard OTC formulations and exclude specialty products, bulk hospital supply, or brand-name equivalents if any exist in significant market share.
Regulatory Considerations
The regulatory environment for Promethazine-DM is shaped by the regulations governing its individual active ingredients, promethazine hydrochloride and dextromethorphan hydrobromide.
FDA Oversight:
- Drug Approval and Labeling: Promethazine-DM products are subject to FDA regulations under the Federal Food, Drug, and Cosmetic Act. Manufacturers must obtain approval (e.g., through Abbreviated New Drug Applications for generics) demonstrating safety, efficacy, and quality. Labeling requirements are stringent, detailing indications, contraindications, warnings, dosage, and administration.
- Pediatric Use: The FDA has previously issued warnings and recommendations regarding the use of OTC cough and cold medications, including those containing antihistamines and cough suppressants, in children under two years old due to the risk of serious side effects. While Promethazine-DM is typically not recommended for this age group, manufacturers must ensure clear labeling to this effect. [1]
- Controlled Substance Status: Dextromethorphan has faced increasing scrutiny regarding its potential for abuse. While not a Schedule I-V controlled substance, some states have enacted age restrictions for its purchase or require it to be sold from behind the pharmacy counter to curb misuse. [2] Promethazine is a prescription-only medication in many jurisdictions, although its use in combination OTC products is common. Regulatory bodies monitor its diversion and misuse.
- Manufacturing Standards: All manufacturing facilities must adhere to Current Good Manufacturing Practices (cGMP) to ensure product quality, purity, and consistency. [3]
Impact on Market:
- Labeling Compliance: Manufacturers must ensure their labeling aligns with current FDA guidance, particularly regarding pediatric use and potential abuse. Non-compliance can lead to product recalls and regulatory action.
- Distribution Restrictions: If states implement further restrictions on dextromethorphan sales (e.g., requiring behind-the-counter placement), this could impact distribution channels and consumer accessibility, potentially influencing market dynamics for combination products.
- Post-Market Surveillance: The FDA conducts post-market surveillance to monitor adverse events. Significant safety signals for either active ingredient could lead to label changes, warnings, or even market withdrawal.
- Generic Drug Competition: The regulatory pathway for generic drug approval allows for multiple manufacturers to enter the market once a brand-name drug's patent expires, fostering price competition.
Key Takeaways
Promethazine-DM operates within the competitive and price-sensitive cough and cold remedies market, primarily as a generic combination product. Its pricing is heavily influenced by the availability of low-cost generic alternatives for both promethazine and dextromethorphan. Projections indicate continued price stability with a slight downward trend in real terms over the next 3-5 years, driven by persistent generic competition and stable API costs. Regulatory oversight, particularly concerning pediatric use and the potential for dextromethorphan abuse, shapes market access and labeling requirements.
Frequently Asked Questions
What are the primary indications for Promethazine-DM?
Promethazine-DM is indicated for the temporary relief of cough and upper respiratory symptoms associated with the common cold or allergies.
What is the typical price range for OTC Promethazine-DM?
Over-the-counter (OTC) formulations of Promethazine-DM, such as a 4-ounce syrup, typically retail between $7.00 and $15.00, varying by brand and retailer.
What regulatory concerns impact Promethazine-DM?
Key regulatory concerns include FDA guidelines on pediatric use of cough and cold medications and state-level regulations addressing the potential misuse of dextromethorphan.
How does generic availability affect Promethazine-DM pricing?
The widespread availability of generic versions of both promethazine and dextromethorphan leads to intense price competition, keeping prices for combination products low.
Are there any projected significant price increases for Promethazine-DM?
Significant price increases are not anticipated in the near to mid-term; market stability is expected due to ongoing generic competition and predictable manufacturing costs.
Citations
[1] U.S. Food and Drug Administration. (2008, July 16). FDA requests label changes to warn about cough and cold drug risks in children younger than 6. U.S. Food and Drug Administration. Retrieved from https://www.fda.gov/drugs/drug-safety-and-availability/fda-requests-label-changes-warn-about-cough-and-cold-drug-risks-children-younger-6
[2] National Association of Boards of Pharmacy. (n.d.). Dextromethorphan (DXM) State Laws & Regulations. Retrieved from https://nabp.pharmacy/programs/consumer-health/dextromethorphan/state-laws-regulations/
[3] U.S. Food and Drug Administration. (2021, April 22). Current Good Manufacturing Practice (CGMP) for Drugs. U.S. Food and Drug Administration. Retrieved from https://www.fda.gov/drugs/pharmaceutical-quality-and-compliance/current-good-manufacturing-practice-cgmp-drugs
More… ↓
