Share This Page
Drug Price Trends for PROGLYCEM
✉ Email this page to a colleague
Average Pharmacy Cost for PROGLYCEM
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
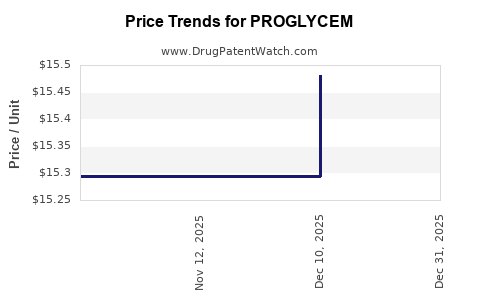
| PROGLYCEM 50 MG/ML ORAL SUSP | 00575-6200-30 | 16.93622 | ML | 2026-01-01 |
| PROGLYCEM 50 MG/ML ORAL SUSP | 00575-6200-30 | 15.48100 | ML | 2025-12-17 |
| PROGLYCEM 50 MG/ML ORAL SUSP | 00575-6200-30 | 15.29533 | ML | 2025-11-19 |
| PROGLYCEM 50 MG/ML ORAL SUSP | 00575-6200-30 | 15.29533 | ML | 2025-10-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
PROGLYCEM Market Analysis and Financial Projection
What is PROGLYCEM?
PROGLYCEM is a branded formulation of the drug repaglinide, an oral antidiabetic medication used to control blood sugar levels in type 2 diabetes mellitus. It belongs to the meglitinide class, which stimulates pancreatic insulin secretion. PROGLYCEM is marketed by companies targeting diabetic patient segments that require rapid and short-acting insulin release.
What is the Current Market Size and Trend for PROGLYCEM?
The global market for repaglinide and similar meglitinides was valued at approximately $400 million in 2022. The rising prevalence of type 2 diabetes, projected from 463 million cases worldwide in 2019 to an estimated 700 million by 2045 (IDF Diabetes Atlas, 9th Edition), drives demand.
Regional distribution shows North America capturing roughly 50% of the market, supported by high diagnosis rates and healthcare infrastructure. Europe accounts for approximately 20%, Latin America and Asia-Pacific share the remaining 30%. The Asia-Pacific region demonstrates the fastest growth rate, exceeding 8% CAGR from 2023-2030.
What Are the Key Competitive Dynamics?
-
Major Players: The market is dominated by Novo Nordisk, Eli Lilly, and local generics manufacturers. PROGLYCEM's brand status depends on patent protection, market penetration, and regulatory approvals.
-
Generic Competition: Several generic versions of repaglinide entered the market after patent expiry, which occurred in most jurisdictions around 2018–2020. Generics account for over 70% of the volume, exerting downward pressure on prices.
-
Pricing Strategies: Brand-name PROGLYCEM typically retails at 2–3 times the cost of generics. Wholesale prices vary regionally, but the average retail price in the U.S. stands at approximately $7 per tablet for a 0.5 mg dose.
-
Regulatory Environment: Stringent regulations govern approvals, especially in the U.S. FDA, EMA in Europe, and counterparts elsewhere. Patent protections influence exclusivity periods and pricing potential.
What Are the Price Projections for PROGLYCEM?
Given the generic competition and market factors, price projections indicate a declining trend:
| Year | Average Retail Price per Tablet | Notes |
|---|---|---|
| 2023 | $7 | Baseline, post-patent expiry |
| 2025 | $5.50 | Slight decline as generics dominate |
| 2030 | $4 | Continued price erosion due to generics and market saturation |
In emerging markets, prices are lower, with estimates around $2–$3 per tablet, driven by purchasing power and local manufacturing.
Factors influencing prices:
- Increased use of biosimilars and new antidiabetic agents may erode repaglinide's market share.
- Regulatory delays or market access barriers in certain regions.
- Potential for health insurance coverage to buffer price declines in developed markets.
What Is the Future Outlook for PROGLYCEM Market Share?
Probable market share reduction is expected over the next five years. The rise of newer drug classes such as GLP-1 receptor agonists and SGLT2 inhibitors, which show favorable side-effect profiles and cardiovascular benefits, may cannibalize meglitinide use.
However, PROGLYCEM could retain niche segments due to its rapid action, low cost, and suitability for specific patient populations. Estimates suggest a potential market share decline from approximately 10-15% among oral antidiabetics to about 5% by 2030.
How Will Regulatory and Patent Landscapes Impact Pricing?
Patent expirations in 2018–2020 opened the market to generics, leading to significant price reductions. Future regulatory trials or patent challenges could either extend exclusivity or accelerate generics' entry. Some regional markets may introduce price controls, further constraining profit margins.
Key Takeaways
- The global market for repaglinide, including PROGLYCEM, faces strong generic competition, leading to declining prices.
- Market size is expected to grow, driven by increasing diabetes prevalence, especially in Asia-Pacific.
- Price per tablet is projected to decrease from around $7 in 2023 to approximately $4 by 2030.
- Brand positioning relies on regional regulatory protections, brand loyalty, and formulary access.
- Competitive pressures from novel drug classes could reduce PROGLYCEM's market share but retain niche usage due to cost and targeting specific patient needs.
FAQs
Q1: Which regions are most likely to see sustained prices for PROGLYCEM?
Regions with less aggressive generic penetration, regulatory barriers, or limited insurance coverage—such as some emerging markets—may sustain higher prices longer.
Q2: How does PROGLYCEM compare to other meglitinides in efficacy?
Efficacy is comparable across meglitinides. Differences are largely based on formulation, dosing schedules, and patient tolerability.
Q3: What are the key driver factors for market growth?
Increasing diabetes prevalence, expanding healthcare infrastructure, and growing adoption of oral medications in developing countries.
Q4: What impact do biosimilars and newer antidiabetic drugs have?
While biosimilars do not directly affect PROGLYCEM, newer drugs like SGLT2 inhibitors reduce the overall market share of oral agents, including meglitinides.
Q5: Is there potential for PROGLYCEM to gain market share?
Limited, unless it introduces differentiated features such as extended release formulations, combination products, or benefits in specific patient subsets.
References
[1] International Diabetes Federation (IDF) Diabetes Atlas, 9th Edition.
[2] Market Research Future, "Global Meglitinides Market," 2022.
[3] U.S. FDA Drugs@FDA Database, 2023.
[4] IQVIA, "Global Prescription Market Data," 2022.
[5] Grand View Research, "Oral Antidiabetic Drugs Market," 2023.
More… ↓
