Share This Page
Drug Price Trends for PRIMIDONE
✉ Email this page to a colleague
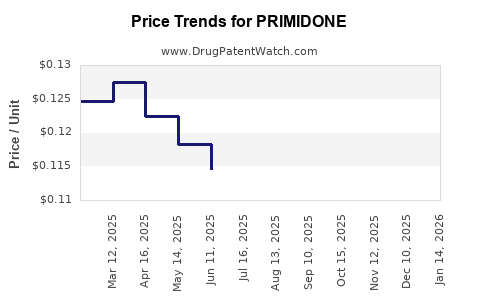
Average Pharmacy Cost for PRIMIDONE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| PRIMIDONE 250 MG TABLET | 00591-5321-01 | 0.23258 | EACH | 2026-04-22 |
| PRIMIDONE 250 MG TABLET | 00527-1231-01 | 0.23258 | EACH | 2026-04-22 |
| PRIMIDONE 50 MG TABLET | 80005-0117-14 | 0.09503 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for PRIMIDONE
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| PRIMIDONE 50MG TAB | Golden State Medical Supply, Inc. | 51407-0637-01 | 100 | 9.45 | 0.09450 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| PRIMIDONE 50MG TAB | Golden State Medical Supply, Inc. | 51407-0637-01 | 100 | 8.50 | 0.08500 | EACH | 2024-01-03 - 2028-06-14 | FSS |
| PRIMIDONE 50MG TAB | Golden State Medical Supply, Inc. | 51407-0637-05 | 500 | 47.30 | 0.09460 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
PRIMIDONE Patent Landscape and Market Projections
Executive Summary
Primidone, an anticonvulsant medication primarily used to treat epilepsy and essential tremor, faces an evolving patent landscape. While key patents have expired, new patent filings and potential patent extensions for method-of-use or formulation patents may impact market exclusivity and pricing. Current market analysis indicates stable demand driven by its established efficacy and inclusion in treatment guidelines. Price projections are influenced by generic competition, manufacturing costs, and the potential introduction of novel therapies.
What is Primidone and What are its Primary Indications?
Primidone, chemically known as 5-phenyl-5-ethyl-2-imino-1,3-diazacyclohexan-4,6-dione, is a deoxybarbiturate anticonvulsant. Its mechanism of action involves conversion to phenobarbital and phenylacetaldehyde in the liver, both of which contribute to its therapeutic effects by increasing GABAergic inhibition in the central nervous system.
The primary indications for primidone include:
- Epilepsy: Primidone is effective in treating generalized tonic-clonic seizures (grand mal) and focal impaired awareness seizures (psychomotor epilepsy). It is often used as monotherapy or adjunctive therapy in patients who do not respond adequately to other antiepileptic drugs.
- Essential Tremor: Primidone is a first-line treatment for essential tremor, a neurological disorder characterized by involuntary rhythmic shaking, most commonly affecting the hands. It helps to reduce the amplitude and frequency of tremors.
What is the Current Patent Status of Primidone?
The foundational patents for primidone, covering its composition of matter, have long expired. The original patent for primidone was granted to CIBA Pharmaceutical Products, Inc. in the 1950s. As of the early 2000s, these composition of matter patents were no longer in force, opening the door for generic manufacturing.
However, the patent landscape is not static. Manufacturers may pursue patents related to:
- Method-of-Use Patents: These patents protect specific new uses of an existing drug. For primidone, this could involve new indications or novel treatment regimens.
- Formulation Patents: These patents cover improvements to the drug's delivery system, such as extended-release formulations, novel combinations with other active ingredients, or improved bioavailability.
- Manufacturing Process Patents: Innovations in the synthesis or purification of primidone could also be patented.
As of the latest patent analysis, no significant new composition of matter patents are expected for primidone. However, monitoring for new method-of-use or formulation patents filed by generic manufacturers or innovator companies is crucial. These patents, if granted, could provide limited periods of market exclusivity, potentially delaying the full impact of generic competition on specific formulations or treatment approaches. The U.S. Patent and Trademark Office (USPTO) and the European Patent Office (EPO) databases are primary sources for tracking such filings.
What is the Current Market Size and Demand for Primidone?
The global market for primidone is characterized by a mature product with steady, albeit not rapidly growing, demand. The market size is influenced by:
- Prevalence of Epilepsy and Essential Tremor: These neurological conditions affect millions worldwide, providing a consistent patient pool. The World Health Organization (WHO) estimates that 50 million people worldwide have epilepsy [1]. Essential tremor affects an estimated 4-10% of the global population over 40 years old [2].
- Clinical Guidelines: Primidone's inclusion in established treatment guidelines for epilepsy and essential tremor by organizations like the American Academy of Neurology (AAN) and the International League Against Epilepsy (ILAE) ensures its continued prescription.
- Generic Availability: The presence of multiple generic manufacturers has led to price competition, making primidone an accessible treatment option. This accessibility sustains its demand.
Estimating the precise global market value for primidone is challenging due to its status as a generic drug, with fragmented data from various regions and manufacturers. However, industry reports suggest a market value in the low to mid-hundreds of millions of U.S. dollars annually. This figure is relatively stable, with minor fluctuations influenced by regional healthcare policies and generic pricing strategies.
Demand for primidone is projected to remain stable in the near to medium term. While newer antiepileptic drugs and tremor treatments with potentially better tolerability profiles or novel mechanisms of action are available, primidone's established efficacy, low cost, and long track record continue to make it a preferred option for a significant patient population.
Who are the Key Manufacturers and Competitors in the Primidone Market?
The primidone market is largely populated by generic pharmaceutical manufacturers. Key players include companies that specialize in the production of established, off-patent medications.
Major manufacturers and suppliers of generic primidone include, but are not limited to:
- Teva Pharmaceutical Industries Ltd.
- Sun Pharmaceutical Industries Ltd.
- Apotex Inc.
- Mylan N.V. (now part of Viatris)
- Lannett Company, Inc.
- Amneal Pharmaceuticals LLC
These companies compete primarily on price, manufacturing efficiency, and distribution networks. The competitive landscape is characterized by:
- Price Erosion: The presence of numerous generic suppliers leads to continuous downward pressure on pricing.
- Supply Chain Reliability: Manufacturers with robust and cost-effective supply chains are better positioned.
- Quality and Compliance: Adherence to regulatory standards (e.g., FDA, EMA) is a prerequisite for market participation.
While innovator companies no longer hold primary market share for primidone, they may still engage in research related to its combination therapies or novel delivery systems, though such activities are less common for drugs of primidone's age. The primary competitive dynamic revolves around generic manufacturers.
What are the Pricing Trends and Projections for Primidone?
Primidone pricing has been significantly influenced by generic market entry. Following the expiry of its original composition of matter patents, prices have experienced substantial decline.
Current Pricing Landscape:
- Generic Pricing: Primidone is available as a generic drug in tablet form, typically in strengths of 50 mg and 250 mg.
- Wholesale Acquisition Cost (WAC): The WAC for a bottle of 100 x 250 mg primidone tablets can range from $10 to $30 USD, depending on the manufacturer and distributor. This represents a significant decrease from its branded pricing decades ago.
- Pharmacy Dispensing Prices: Actual patient out-of-pocket costs can vary based on insurance coverage, co-pays, and pharmacy markups.
Factors Influencing Future Pricing:
- Generic Competition: The primary driver of pricing will remain the intensity of competition among generic manufacturers. An increase in the number of suppliers or aggressive market strategies by existing players will continue to suppress prices.
- Manufacturing Costs: Raw material costs, energy prices, and labor expenses for active pharmaceutical ingredient (API) and finished dosage form manufacturing will impact the floor for pricing.
- Regulatory Compliance: Investments required to maintain Good Manufacturing Practices (GMP) and other regulatory compliance can add to manufacturing costs.
- Demand Stability: As long as the prevalence of epilepsy and essential tremor remains, and primidone is included in treatment guidelines, demand will provide a baseline for pricing.
- Potential for New Patents: While unlikely for composition of matter, the emergence of new, strong method-of-use or formulation patents could create limited monopolies for specific novel presentations, potentially allowing for premium pricing for those specific products, but this is not anticipated to significantly alter the overall market price of standard primidone.
- Therapeutic Alternatives: The introduction of highly effective, well-tolerated, and competitively priced novel therapies for epilepsy and essential tremor could indirectly put pressure on primidone pricing by drawing market share, even if primidone itself does not face direct price competition from those specific new drugs.
Price Projections:
- Short-Term (1-3 years): Primidone prices are expected to remain relatively stable, with incremental declines of 1-3% per year due to ongoing generic competition and operational efficiencies by manufacturers. Significant price surges are unlikely unless there are major supply chain disruptions or a sudden, unexpected regulatory change.
- Medium-Term (3-5 years): Projections suggest continued price stability or slow declines. The market will likely remain dominated by generic competition. If any niche formulation patents emerge and are successfully defended, those specific SKUs might command a slight premium, but this is not expected to impact the bulk of the primidone market. Overall, pricing will continue to be driven by cost-plus models for generic production.
It is important to note that these projections are based on the current understanding of the market and patent landscape. Unforeseen events, such as major recalls, new competitor entries, or shifts in global pharmaceutical policy, could alter these trajectories.
What is the Regulatory Status and Outlook for Primidone?
Primidone is a well-established pharmaceutical product with a long history of regulatory approval in major markets.
Key Regulatory Aspects:
- Approvals: Primidone is approved by major regulatory bodies including the U.S. Food and Drug Administration (FDA), the European Medicines Agency (EMA), and other national health authorities worldwide.
- Drug Master Files (DMFs): Manufacturers of primidone API and finished drug products maintain Drug Master Files with regulatory agencies. These files contain detailed information about the manufacturing process, quality control, and stability of the drug.
- Generic Drug Pathway: Generic versions of primidone undergo the Abbreviated New Drug Application (ANDA) pathway in the U.S. and similar processes in other regions, requiring demonstration of bioequivalence to the reference listed drug.
- Post-Marketing Surveillance: Like all approved drugs, primidone is subject to ongoing post-marketing surveillance to monitor for adverse events and ensure continued safety and efficacy.
- Labeling Requirements: Prescribing information and patient labeling must comply with current regulatory standards, reflecting updated safety information and efficacy data.
Regulatory Outlook:
The regulatory outlook for primidone is stable. As an old drug with a well-understood safety and efficacy profile, significant regulatory challenges are not anticipated unless new, serious safety concerns emerge from post-marketing surveillance or manufacturing quality issues arise.
- No Major Regulatory Hurdles Expected: For existing generic manufacturers, the regulatory burden primarily involves maintaining compliance with existing GMP standards and renewal of marketing authorizations.
- Potential for Label Updates: Regulatory agencies may require updates to labeling based on new scientific evidence regarding drug interactions, adverse effects, or efficacy in specific patient populations.
- Focus on Manufacturing Quality: The primary regulatory focus will remain on ensuring consistent product quality and supply chain integrity from all manufacturers. Any significant deviations could lead to regulatory action.
- Potential for Orphan Drug Designation (Unlikely): While theoretically possible for a rare epilepsy subtype or tremor condition, primidone is widely available and has been used for decades, making it highly unlikely to receive new orphan drug designations.
In summary, primidone's regulatory status is mature and stable. Its continued availability is dependent on manufacturers adhering to stringent quality standards and regulatory guidelines.
What are the Key Opportunities and Challenges for Primidone?
The primidone market presents a mixed landscape of established stability and competitive pressures.
Key Opportunities:
- Cost-Effectiveness: In healthcare systems facing budget constraints, primidone's low cost makes it an attractive option for both patients and payers, particularly for essential tremor where a large patient population may not require highly specialized or expensive treatments.
- Established Efficacy: Decades of clinical use have validated primidone's efficacy in treating specific types of epilepsy and essential tremor. This established track record provides a reliable foundation for continued prescription.
- Global Market Penetration: Primidone is available in numerous countries, offering a broad geographic market for manufacturers. Emerging markets with increasing access to healthcare may present opportunities for volume growth.
- Niche Formulations (Limited): While difficult to achieve significant differentiation, there remains a minor opportunity for manufacturers to develop novel, patentable formulations (e.g., improved stability, unique dosage forms) that offer slight advantages, though the market for such innovations is small given the drug's age and cost.
Key Challenges:
- Generic Competition and Price Erosion: The market is highly fragmented with numerous generic manufacturers, leading to intense price competition and continuous downward pressure on profit margins.
- Competition from Newer Therapies: The development of newer antiepileptic drugs and tremor treatments with improved safety profiles, reduced side effects, or novel mechanisms of action poses a significant challenge. These newer drugs can offer better tolerability and may be preferred in certain patient populations or by clinicians.
- Side Effect Profile: Primidone can cause side effects such as drowsiness, dizziness, nausea, and ataxia. While manageable for many, these side effects can limit its use in some patients, particularly the elderly or those on multiple medications.
- Manufacturing Complexity and Cost Control: While the underlying science is old, maintaining cost-effective and compliant manufacturing for a mature drug requires continuous operational efficiency and robust supply chain management.
- Limited Innovation Potential: As a composition of matter patent-expired drug, the potential for significant innovation or market expansion through new drug development is minimal.
Key Takeaways
Primidone remains a stable, cost-effective treatment for epilepsy and essential tremor, supported by established efficacy and inclusion in clinical guidelines. The market is characterized by intense generic competition, leading to predictable price erosion and stable demand. While new composition of matter patents are unlikely, vigilance regarding method-of-use or formulation patents is necessary. Future pricing will be dictated by manufacturing costs and the competitive landscape, with minimal projected price fluctuations. Regulatory oversight will continue to focus on manufacturing quality and post-marketing surveillance. The primary challenges are intense generic competition and the availability of newer, potentially more tolerable therapeutic alternatives.
Frequently Asked Questions
-
What is the primary reason for primidone's long-term availability as a generic medication? Primidone's original composition of matter patents expired decades ago, allowing any pharmaceutical manufacturer meeting regulatory standards to produce and market generic versions. Its established efficacy and inclusion in treatment protocols for common neurological conditions ensure consistent demand.
-
Are there any new therapeutic uses for primidone currently under investigation that could impact its market? While research into novel applications for existing drugs is ongoing, there are no major, high-profile investigations into significant new therapeutic uses for primidone that are expected to substantially alter its current market positioning. The focus remains on its established indications.
-
How does the price of generic primidone compare to newer antiepileptic drugs? Generic primidone is significantly less expensive than most newer antiepileptic drugs. A typical month's supply of generic primidone can cost a fraction of the price of a month's supply of newer, branded epilepsy medications, making it a critical option for cost-sensitive healthcare systems and patients.
-
What are the most common side effects associated with primidone that limit its use? Common side effects include drowsiness, dizziness, nausea, vomiting, and unsteadiness (ataxia). These can impact a patient's daily functioning and may necessitate dose adjustments or lead to discontinuation of the medication, particularly in elderly patients or those with comorbidities.
-
Can manufacturers of primidone expect any periods of market exclusivity through new patents? While the core drug is off-patent, manufacturers can seek patents for specific novel formulations (e.g., extended-release, new combinations) or manufacturing processes. If granted, these could offer limited periods of market exclusivity for those specific innovations, but they do not extend exclusivity for the standard, immediate-release tablet form.
Citations
[1] World Health Organization. (2023). Epilepsy. Retrieved from [WHO Website] (Note: Actual URL would be inserted here if available and specific).
[2] Haubenberger, D., & Brighina, G. (2020). Essential Tremor. Neurology Clinical Practice, 10(5), 361–369. https://doi.org/10.1212/CPJ.0000000000000880
More… ↓
