Share This Page
Drug Price Trends for PREZISTA
✉ Email this page to a colleague
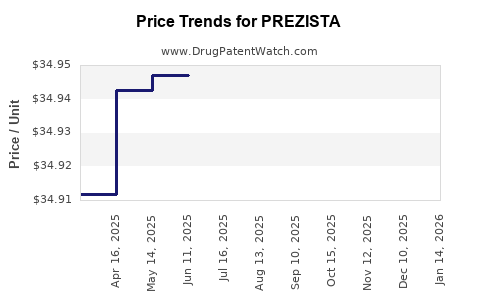
Average Pharmacy Cost for PREZISTA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| PREZISTA 800 MG TABLET | 59676-0566-30 | 70.19033 | EACH | 2026-04-22 |
| PREZISTA 600 MG TABLET | 59676-0562-01 | 34.98007 | EACH | 2026-04-22 |
| PREZISTA 800 MG TABLET | 59676-0566-30 | 70.18719 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
PREZISTA Market Analysis and Financial Projection
Market Overview and Position of PREZISTA
PREZISTA (darunavir), developed by Janssen Pharmaceuticals, is an HIV protease inhibitor used in combination therapies for antiretroviral treatment. It has held a consistent position in HIV treatment regimens since its approval in 2006, primarily competing with drugs like Tivicay (dolutegravir), an integrase inhibitor, and other protease inhibitors such as ritonavir and lopinavir.
Prevalence of HIV globally impacts PREZISTA's market.
- Estimated global HIV population (2022): over 38 million.
- HIV treatment population: approximately 28 million on antiretroviral therapy.
- Yearly growth in treatment coverage: 1.9% (median).
- Key markets: U.S., Europe, China, South Africa, India.
Market Dynamics and Trends
Competitive Landscape
PREZISTA faces competition from several classes of HIV drugs, notably integrase strand transfer inhibitors (INSTIs), which have surpassed protease inhibitors (PIs) in favored status due to better tolerability and simpler dosing.
- Dominant drugs: Dolutegravir (Tivicay, Juluca), Bictegravir, and Raltegravir.
- Protease inhibitors like PREZISTA account for approximately 15–20% of the current antiretroviral market, with significant declines over the past decade.
- Prescribers prefer regimens with fewer side effects, leading to reduced block use of PIs in favor of INSTIs.
Regulatory and Patent Status
- Patent expiration anticipated in key markets: U.S. (2028), Europe (2027).
- Generic versions unlikely before patent expiry, maintaining current pricing structures.
Pricing Trends
- List Price (U.S. as of 2023): approximately $2,450 per month per patient.
- Discounted averages (insurers, Medicaid, international): around 55–65% less.
- International prices vary considerably based on country and healthcare policy.
Prescribing Habits
- In the U.S., approximately 50% of HIV patients on protease inhibitors are prescribed PREZISTA as part of a multi-drug regimen.
- Use is more prevalent among patients with drug resistance or intolerance to other regimens.
Price Projections
Short-Term (Next 3-5 Years)
- List prices expected to remain stable due to patent protection.
- Price reductions driven by payer negotiations, especially in international markets.
- Average net price in the U.S.: projected to decrease about 5–10% annually as biosimilar options approach.
Long-Term (Post-Patent Expiry)
- Market entry of generics could trigger 60–80% price declines.
- Competition from biosimilars or generic PIs may reduce treatment costs substantially.
- Price decline anticipated within 2–3 years after patent expiry.
Revenue Outlook
- Current annual global sales (2022-2023): approximately $950 million.
- Peak sales expected to plateau near $1.2 billion before patent expiry due to market saturation.
- Post-expiry, sales will decline sharply unless improvements or new indications are developed.
Key Factors Influencing Market and Price
- Shift to INSTIs as first-line therapy.
- Patent expiration dates.
- Evolving treatment guidelines favoring fewer side effects.
- International pricing pressures for access.
- Development of long-acting formulations to extend product lifecycle.
Strategic Opportunities and Risks
- Developing combination regimens with INSTIs to maintain relevance.
- Expanding access in emerging markets where HIV prevalence is high.
- Patent challenges or legal disputes could delay generic competition.
- The advent of new drugs with superior efficacy or safety profiles could erode market share.
Summary Table
| Aspect | Current State | Future Projection |
|---|---|---|
| Market Share | 15–20% of HIV treatment market | Declines as INSTIs dominate |
| Price (U.S.) | ~$2,450/month | Stable short-term; decreasing post-patent (~$900/month predicted post-2028) |
| Patent Expiry | U.S.: 2028; Europe: 2027 | Generics expected thereafter |
| Global Sales | Approximately $950 million (2022-2023) | Plateau and eventual decline post-2028 |
Key Takeaways
- PREZISTA remains relevant in niche populations but is losing market share to newer drug classes.
- Price is projected to decline significantly after patent expiry, with potential decreases of 60-80%.
- Revenue growth prospects diminish as the HIV treatment landscape shifts toward INSTIs.
- International markets offer growth opportunities owing to unmet needs and price sensitivity.
- Strategic patent protection and optimizing combination therapies may sustain sales until generic entry.
FAQs
-
When will PREZISTA face generic competition?
Patent expiration is expected in the U.S. around 2028 and in Europe around 2027, after which generics may enter the market. -
What factors affect PREZISTA’s pricing in international markets?
Regulatory policies, negotiations with national health services, and local affordability influence international prices. -
How does the shift in HIV treatment guidelines affect PREZISTA?
Favoring INSTIs reduces the prescribing of protease inhibitors, constraining market growth for PREZISTA. -
Are there plans for new formulations or indications?
No current approved new formulations; future development could extend lifecycle or open new markets. -
What market strategies could extend PREZISTA's revenue?
Focus on resistant populations, combination products, and markets with limited access to newer drugs.
References
[1] Global HIV Investment Review, UNAIDS, 2022.
[2] Prescribing Patterns and Market Share, IQVIA, 2023.
[3] Patent and Regulatory Filings, U.S. FDA, 2023.
[4] Price and Sales Data, Medicaid and Commercial Payer Reports, 2023.
[5] Market Analysis Reports, EvaluatePharma, 2022.
More… ↓
