Share This Page
Drug Price Trends for PREVACID DR
✉ Email this page to a colleague
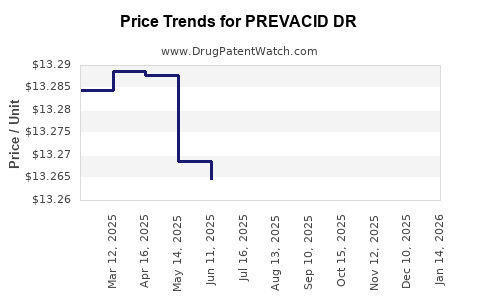
Average Pharmacy Cost for PREVACID DR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| PREVACID DR 30 MG CAPSULE | 64764-0046-13 | 13.24575 | EACH | 2026-03-18 |
| PREVACID DR 15 MG SOLUTAB | 64764-0543-30 | 13.23986 | EACH | 2026-03-18 |
| PREVACID DR 30 MG SOLUTAB | 64764-0544-30 | 13.23369 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
PREVACID DR Market Analysis and Financial Projection
Market Analysis and Price Projections for PREVACID DR
Overview
PREVACID DR ( lansoprazole delayed-release capsules) is a proton pump inhibitor (PPI) primarily indicated for the treatment of gastroesophageal reflux disease (GERD), erosive esophagitis, and Zollinger-Ellison syndrome. Its market derives from chronic acid suppression therapy, with steady demand across outpatient and hospital settings.
Market Size and Demand Drivers
Global Proton Pump Inhibitor Market
The PPI market was valued at approximately $14 billion in 2022, with projections reaching $20 billion by 2030, at a CAGR of 4.3%. PPS (Prescription-Preferred Status) accounts for 65% of PPI sales, with OTC products capturing roughly 35%.
Key Factors Influencing Demand
- Rising prevalence of GERD: Estimated at 20% in North America and 10-15% in Europe.
- Aging populations: Patients over 60 comprise 50% of PPI users.
- Increased use of combination therapies and longer treatment durations.
- Growing awareness of acid-related disorders drives prescriptions.
Competitive Landscape
Generic lansoprazole supplies dominate, with several manufacturers holding approval for multiple formulations. PREVACID DR's brand exclusivity ended in early 2016, leading to widespread generic availability and price erosion.
Pricing Trends
Historical Price Dynamics
- Original brand (PREVACID): Originally priced at $350 for a 30-day supply.
- Once generic entry occurred in 2016, prices declined sharply, stabilizing around $10–$15 per 30-day supply for generics.
Current Pricing Range
- Generic 30-day supply: $10–$20 depending on pharmacy and insurer discounts.
- Privately insured patients typically pay $15–$20; uninsured populations face higher out-of-pocket costs.
Formulation-Specific Pricing
PREVACID DR capsules generally retail at a premium over traditional generics, priced around $25–$35 per month, but these prices are not sustainable outside of specialty markets or branded contexts due to patent expiry.
Patent and Regulatory Status
PREVACID DR was granted patents covering its delayed-release formulation, but patent expirations in 2016 opened the market to generics. No recent patent filings or exclusivity extensions suggest sustained brand dominance.
Price Projections (Next 5 Years)
| Year | Estimated Brand Market Price per 30-Day Supply | Estimated Generic Market Price per 30-Day Supply |
|---|---|---|
| 2023 | $25–$35 | $10–$20 |
| 2024 | $20–$30 (if brand maintains niche positioning) | $10–$18 |
| 2025 | $15–$25 | $10–$15 |
| 2026 | $15–$20 (brand phase-out possible) | $8–$15 |
| 2027 | $10–$15 (market dominated by generics) | $8–$12 |
Assumptions:
- Market share for brand diminishes as generic presence solidifies.
- No significant patent extensions or formulation innovates.
- Price erosion stabilizes due to increased generic competition.
- Continued growth in demand for acid suppression therapies sustains overall market volume.
Competitive and Regulatory Risks
- Patent litigations or additional patent extensions could temporarily sustain brand pricing.
- Introduction of novel PPIs with improved efficacy or safety profiles could reduce demand for existing drugs.
- Regulatory changes affecting drug approval or reimbursement policies could pressure prices.
Conclusion:
PREVACID DR's market is now predominantly driven by generic competition, with prices trending downward. Branded formulations sustain a niche in specific patient segments but face erosion over the next several years.
Key Takeaways
- The global PPI market remains robust, with chronic acid-related conditions maintaining high demand.
- PREVACID DR's patent protection ended in 2016, leading to price declines and generic market dominance.
- Future pricing will depend highly on generic market dynamics, potential patent litigation, and innovation breakthroughs.
- Prices are expected to stabilize around $10–$15 for generics within five years, with branded formulations maintaining a premium temporarily.
- Market growth hinges on increasing GERD prevalence and aging populations but faces competitive and regulatory risks.
FAQs
Q1: What factors most influence the pricing of generic PPIs like lansoprazole?
Market competition, patent status, and pharmacy discounting primarily determine generic PPI prices.
Q2: How does the expiration of patents affect the availability of PREVACID DR?
Patent expiration opens the market to generics, substantially lowering prices and reducing the market share of the branded version.
Q3: Are there potential patent extensions for PREVACID DR?
No recent patent extensions have been granted; the original patents expired in 2016.
Q4: What is the projected impact of new acid suppression therapies on PREVACID DR's market?
Innovations offering improved safety or efficacy could cannibalize existing PPI sales, especially in specialized niches.
Q5: How does the global prevalence of GERD influence the market for drugs like PREVACID DR?
Higher GERD prevalence, especially in aging populations, sustains demand despite price erosion caused by competition.
References
[1] Grand View Research, "Proton Pump Inhibitors Market Size, Share & Trends Analysis," 2023
[2] IQVIA, "Prescription Drug Price Analysis," 2023
[3] FDA, "Patents and Exclusivities for Proton Pump Inhibitors," 2022
More… ↓
