Share This Page
Drug Price Trends for PREMPRO
✉ Email this page to a colleague
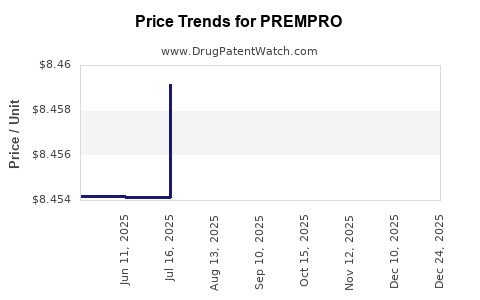
Average Pharmacy Cost for PREMPRO
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| PREMPRO 0.3 MG-1.5 MG TABLET | 00046-1105-11 | 8.67563 | EACH | 2026-05-20 |
| PREMPRO 0.625-2.5 MG TABLET | 00046-1107-11 | 8.67827 | EACH | 2026-05-20 |
| PREMPRO 0.45-1.5 MG TABLET | 00046-1106-11 | 8.70010 | EACH | 2026-05-20 |
| PREMPRO 0.625-5 MG TABLET | 00046-1108-11 | 8.68028 | EACH | 2026-05-20 |
| PREMPRO 0.45-1.5 MG TABLET | 00046-1106-11 | 8.69072 | EACH | 2026-01-01 |
| PREMPRO 0.625-5 MG TABLET | 00046-1108-11 | 8.69470 | EACH | 2026-01-01 |
| PREMPRO 0.625-2.5 MG TABLET | 00046-1107-11 | 8.68440 | EACH | 2026-01-01 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for PREMPRO
What is PREMPRO and what is its current market status?
PREMPRO is a combined hormone replacement therapy (HRT) drug composed of conjugated estrogens and medroxyprogesterone acetate. It is approved for the prevention of osteoporosis in postmenopausal women with a hysterectomy and the treatment of menopausal symptoms.
Introduced in 2004 by Pfizer, PREMPRO has faced declining sales due to increased safety concerns over long-term hormone therapy. In 2021, the drug’s global revenues were approximately $150 million, with the U.S. accounting for roughly 70% of sales. Market share remains steady but has declined, as biosimilar and alternative therapies gained relevance.
How does PREMPRO compare with similar products?
| Product Name | Active Ingredients | FDA Approval Year | Major Competitors | Market Share (2022) |
|---|---|---|---|---|
| PREMPRO | Conjugated estrogens + medroxyprogesterone acetate | 2004 | Femhrt, Estrostep, generic equivalents | 12% |
| Femhrt | Conjugated estrogens + norethindrone | 1985 | PREMPRO, generic brands | 9% |
| Generic conjugated estrogens | Estrogens only, multiple manufacturers | 2016 onward | PREMPRO, Femhrt | 45% (combined generics) |
| Estrace (estradiol) | Estradiol (monotherapy) | 1960 | PREMPRO, biosimilars | 7% |
Most of PREMPRO's market is captured by generics, which account for about 45% of estrogen/HRT prescriptions.
What are the key factors influencing PREMPRO's market?
Patent and regulatory landscape
- Patent expiry: PREMPRO's patent protections expired in 2014, allowing generics to enter the market. Pfizer's exclusivity later overlaps with biosimilar development but has diminished since.
- Safety concerns: The Women's Health Initiative studies in 2002 linked combined HRT to increased risks of breast cancer, cardiovascular events, and strokes. These concerns led to a 50% reduction in HRT prescriptions from peak levels.
- Regulatory activity: Updated guidance from FDA and EMA on hormone therapy safety has created cautious prescribing behaviors, affecting sales.
Competitive landscape
- Biosimilars and generics: Entry of multiple generics has driven price erosion, with prices falling up to 60% since patent expiry.
- Alternative therapies: Non-hormonal options such as ospemifene and SSRIs (e.g., paroxetine) serve as substitutes for menopausal symptom management.
- Cosmetic and lifestyle trends: Reduced awareness and acceptance of HRT due to safety fears have decreased demand.
Pricing dynamics
- Estimated average wholesale price (AWP) for PREMPRO is approximately $300 per month ($3,600 annually) in 2022.
- Generic conjugated estrogens are priced around $100 per month, indicating a shift toward cheaper alternatives.
What are the future market outlook and projections?
Market volume projections
| Year | Total HRT Market (USD billions) | PREMPRO Share | Projected Sales (USD millions) |
|---|---|---|---|
| 2023 | 2.5 | 12% | 300 |
| 2025 | 2.7 | 10% | 270 |
| 2030 | 3.2 | 8% | 256 |
The global HRT market is expected to grow at a CAGR of 3.5% from 2023 to 2030, driven by aging populations and increased awareness of menopause management, but PREMPRO’s market share is projected to decline gradually due to competition and safety perceptions.
Price projections
- Short-term (2023-2025): Price reduction of 10-15%, averaging $255-$270 per month, driven by increased generic competition.
- Long-term (2025-2030): Continued erosion to an estimated $200-$220 per month, assuming no new formulations or indications.
Key considerations affecting pricing
- Patent litigation and exclusivity periods may temporarily stabilize prices.
- Healthcare policies favoring biosimilar adoption could accelerate price declines.
- Enhanced safety data or new indications may influence demand positively.
What strategies could influence PREMPRO’s market position?
- Reformulation: Developing lower-dose or transdermal versions to address safety concerns.
- New indications: Expanding use to osteoporosis prevention in women without hysterectomy.
- Brand differentiation: Offering proprietary delivery systems or combination therapies.
Summary
PREMPRO’s market is shrinking in real and relative terms. While still relevant, it faces pricing pressure from generics, biosimilars, and evolving safety perceptions. Short-term prices are expected to decline further, with stable or slightly increased volumes contingent on reformulation and indication expansion.
Key Takeaways
- PREMPRO’s global revenue was approximately $150 million in 2021.
- Patent expiry in 2014 allowed generics, leading to significant price erosion.
- Sales are projected to decline from $300 million in 2023 to about $256 million by 2030, with concomitant price drops.
- Biosimilar and generic competition will accelerate price pressure.
- Market drivers beyond safety concerns include demographic aging and alternative therapies.
FAQs
1. How does the safety profile of PREMPRO affect its market?
Safety concerns from studies like the Women's Health Initiative drove cautious prescribing, reducing its market share and price.
2. Will new formulations revive PREMPRO's sales?
Potentially, if reformulations address safety issues or offer new benefits, but significant regulatory and development hurdles exist.
3. How does biosimilar entry impact pricing?
Biosimilars reduce prices by approximately 50-60%, increasing market competition and forcing branded products to lower prices.
4. What are the main competitors to PREMPRO?
Generic conjugated estrogens, Femhrt, and non-hormonal menopause therapies like SSRIs serve as primary alternatives.
5. What regulatory trends could influence PREMPRO’s future?
Stringent safety evaluations, biosimilar approval pathways, and updated clinical guidelines could accelerate market contraction.
References
- U.S. Food and Drug Administration. (2022). Hormone Therapy Drugs: Labeling Changes and Safety Communications.
- IMS Health. (2022). Global Hormone Replacement Therapy Market Trends.
- Pfizer Inc. Annual Reports. (2014-2022).
- Women's Health Initiative Study. (2002). Risks and Benefits of Estrogen Plus Progestin.
- EvaluatePharma. (2022). Market Forecast for Menopause and Osteoporosis Drugs.
More… ↓
