Share This Page
Drug Price Trends for PREGNYL
✉ Email this page to a colleague
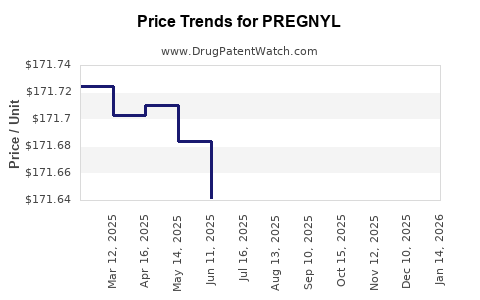
Average Pharmacy Cost for PREGNYL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| PREGNYL 10,000 UNIT VIAL | 78206-0150-99 | 171.61680 | EACH | 2026-04-22 |
| PREGNYL 10,000 UNIT VIAL | 78206-0150-01 | 171.61680 | EACH | 2026-04-22 |
| PREGNYL 10,000 UNIT VIAL | 78206-0150-99 | 171.65617 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Pregnyl (human chorionic gonadotropin)
What is Pregnyl and its current market position?
Pregnyl (human chorionic gonadotropin, hCG) is marketed primarily for infertility treatments and hormone replacement therapies. Produced by Ferring Pharmaceuticals, it has been approved for uses such as inducing ovulation and treating hypogonadotropic hypogonadism. The product's core indications target fertility clinics, hospitals, and specialty pharmacies.
Global demand stems from increasing infertility rates, evolving reproductive health awareness, and expanding access to fertility treatments. The pharmaceutical market for gonadotropins is highly competitive, featuring products from brands like Ovitrelle (Merck), Novarel (Ferring, in some markets), and generic versions.
Market size: current and projected
2022 market valuation
- Estimated global market size for hCG therapeutics: $700 million
- Major markets: United States (35%), Europe (25%), Asia-Pacific (20%), rest of the world (20%)
Growth rate and forecast
- Compound annual growth rate (CAGR): 5%-7% (2022-2027)
- Drivers: rising infertility prevalence, increased adoption of assisted reproductive technology (ART), improvements in drug accessibility
Regional breakdown
| Region | Market Share (2022) | CAGR (2022-2027) | Key Drivers |
|---|---|---|---|
| United States | 35% | 6% | Fertility clinics, insurance coverage |
| Europe | 25% | 5.5% | Reproductive health policies |
| Asia-Pacific | 20% | 7% | Population growth, healthcare spending |
| Rest of the world | 20% | 5% | Emerging markets |
Competitive landscape
- Top manufacturers: Ferring Pharmaceuticals (Pregnyl), Merck (Ovitrelle), Novartis, generic suppliers
- Patent landscape: Recent patents expired or are close to expiry, fostering generic entry
- Market entry barriers: Regulatory approvals, manufacturing capacity, brand loyalty
Pricing analysis: current and future trends
Current price points
- United States retail price (per 5000 IU vial): $600 - $800
- Europe (per vial): €400 - €600
- Asia-Pacific (per vial): $300 - $500
Factors influencing pricing
- Patent status and exclusivity periods
- Supply chain costs, including raw materials and manufacturing
- Regulatory approval processes
- Competitive dynamics, especially generic entry
Price projections (next 3-5 years)
| Scenario | Price Trend | Key Assumptions |
|---|---|---|
| Conservative | Slight decline of 2-3% annually | Increased generic approvals, stable demand |
| Moderate growth | Price stabilization or slight increase | Higher demand from emerging markets, inflation adjustments |
| Aggressive decline | 5-10% annual decrease | Accelerated generic market entry, patent expirations |
Implications for stakeholders
- Pharmaceutical companies should monitor patent expiry timelines to capitalize on generic competition.
- Investors might see opportunities in emerging markets driven by increasing demand.
- Healthcare providers must navigate pricing pressures while maintaining treatment accessibility.
Key factors affecting market and pricing
- Patent expirations expected around 2023-2024.
- Entry of biosimilars and generics expected to influence downward price pressure.
- Continued growth in ART utilization sustains demand.
- Regulatory environment: approvals in emerging markets could expand access.
- Reimbursement policies vary by country; higher reimbursement correlates with higher pricing stability.
Key Takeaways
- The global hCG market, centered on Pregnyl, was valued at approximately $700 million in 2022, with steady growth driven by fertility trends.
- Patent expiries and generic approvals are poised to reduce prices over the next 3-5 years.
- Prices globally vary, with the US highest and Asian markets generally lower, reflecting economic and regulatory differences.
- Market expansion depends on technological, regulatory, and economic factors influencing fertility treatment adoption.
- Strategic positioning requires tracking patent statuses, supply chain costs, and regional healthcare policies.
FAQs
What factors influence Pregnyl's pricing?
Patent status, manufacturing costs, competition from generics/biosimilars, regional reimbursement policies, and market demand drive price levels.
How will patent expirations impact Pregnyl's market?
Patent expiry around 2023-2024 will enable generic manufacturers to enter, increasing supply and exerting downward pressure on prices.
Are biosimilars expected for Pregnyl?
Yes, biosimilars could emerge within 2-3 years post-patent expiry, further reducing prices and expanding access.
What is the future demand outlook for Pregnyl?
Demand is expected to grow at 5-7% CAGR until 2027, supported by rising infertility rates and ART treatments in emerging markets.
How do regional healthcare policies affect Pregnyl’s market?
Regions with supportive reimbursement policies and high fertility treatment rates tend to sustain higher prices and demand.
References
[1] MarketsandMarkets. (2022). Human Chorionic Gonadotropin Market by Application and Region.
[2] Ferring Pharmaceuticals. (2023). Pregnyl Product Information.
[3] IMS Health. (2022). Global Fertility and Reproductive Medicine Market Report.
[4] U.S. Food and Drug Administration. (2022). Patent and Exclusivity Data for Gonadotropin Products.
More… ↓
