Share This Page
Drug Price Trends for PRAMIPEXOLE ER
✉ Email this page to a colleague
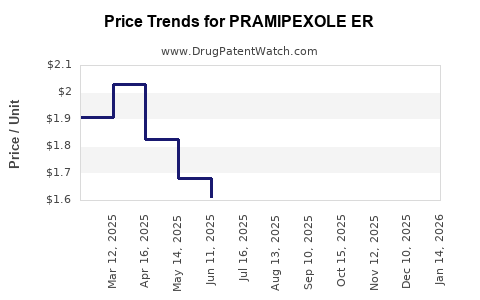
Average Pharmacy Cost for PRAMIPEXOLE ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| PRAMIPEXOLE ER 0.375 MG TABLET | 50742-0331-30 | 1.06589 | EACH | 2026-05-20 |
| PRAMIPEXOLE ER 0.375 MG TABLET | 33342-0208-07 | 1.06589 | EACH | 2026-05-20 |
| PRAMIPEXOLE ER 4.5 MG TABLET | 62332-0159-30 | 1.37400 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Pramipexole ER Market Analysis and Price Projections
Overview
Pramipexole extended-release (ER) is a dopamine agonist indicated primarily for the treatment of Parkinson’s disease and restless legs syndrome (RLS). It is marketed under brand names such as Mirapex ER in the US and Mirapex ER globally. The drug is known for continual dosing over 24 hours, reducing pill burden compared to immediate-release formulations.
Market Landscape
-
Market Size and Growth Trends
The global Parkinson’s disease market was valued at approximately $4.3 billion in 2022, with a decline to $4.1 billion expected in 2023 due to patent expirations and generic entrants. The RLS segment was valued at $750 million globally in 2022, with growth projections of 4-6% annually through 2028.
Pramipexole ER accounts for roughly 20-25% of the overall dopamine agonist market and is used in approximately 35-40% of Parkinson’s cases on medication and around 45-50% for RLS management, with some overlap.
-
Patent and Regulatory Status
The brand-name Mirapex ER's US patent expired in 2017, allowing generics to enter the market. In Europe, patent expiry occurred in 2018. A handful of generic formulations now dominate the market, impacting retail pricing and market share of the branded drug.
-
Competitive Landscape
Major competitors include:
- Ropinirole ER
- Rotigotine transdermal patch
- Cabergoline (less common due to side effects)
Generic versions of pramipexole ER have gained significant market share since patent expiry, leading to price competition.
Pricing Dynamics
-
Brand vs. Generic Pricing
-
Brand Name (Mirapex ER): As of 2023, the average retail price for a 30-day supply (30 tablets of 0.5 mg) stands at approximately $600–$700 in the US.
-
Generic Versions: Pricing drops sharply post-generic entry. A 30-day supply of generic pramipexole ER (0.5 mg) ranges from $12–$30. Discounted rates via pharmacy benefit managers (PBMs) and discount programs further reduce patient out-of-pocket costs.
-
-
Insurance and Reimbursement
Insurance coverage favors generics, significantly lowering patient copays. Brand-name drugs often face formulary restrictions, impacting prescribing patterns.
-
Pricing Trends
- Post-2017, prices for branded Mirapex ER have declined by approximately 30% due to competition.
- The average retail price for generics has been stable but subject to regional and supplier variations.
- Anticipated price stabilization at lower levels unless new formulations or indications emerge.
Future Price Projections
-
Market Penetration of Generics
Generics are projected to capture >90% of the market share in the next 3–5 years. This will pressure prices for any remaining branded formulations.
-
Potential for New Formulations or Indications
- Extended-release formulations for other neurological or psychiatric indications could support price premiums if approved.
- No current pipeline announcements indicate imminent new indications or formulations.
-
Impact of Biosimilars and New Entrants
Even though biosimilars are irrelevant for small-molecule drugs like pramipexole, new competitors entering off-label markets or developing alternative drugs could influence the pricing landscape.
-
Pricing Outlook (2024–2028)
- Branded: Prices may stabilize around $600 per month supply, barring new patent protections or formulations.
- Generics: Prices are expected to remain in the $12–$30 per month supply window, with slight fluctuations due to manufacturing costs and market competition.
Regulatory and Policy Factors
- US drug pricing remains heavily influenced by pharmacy benefit managers and Medicare/Medicaid policies.
- International markets face variability; in Europe, pricing is controlled through national health services and tend to be lower.
Conclusion
The Pramipexole ER market is characterized by declining brand premiums due to generic competition. Pricing strategies are primarily driven by regional policies, insurance reimbursement, and competition among generics. Future outlook suggests stability at low price points for generics, with minimal room for significant price increases unless new formulations or indications emerge.
Key Takeaways
- Most of the market now favors generics, sharply reducing the price of pramipexole ER.
- US retail prices of generics are approximately $12–$30 monthly; branded prices hover around $600.
- Patent expirations in 2017–2018 catalyzed price reductions but stabilized prices since.
- The market growth is limited by patent expiration and generics’ dominance.
- Future pricing will depend on regulatory decisions, market competition, and possible new indications.
FAQs
1. How do patent expirations affect pramipexole ER pricing?
Patent expirations in 2017–2018 allowed generics to enter the market, drastically reducing prices of the drug and shifting market share away from the brand.
2. What regional differences exist in pricing?
US prices are higher due to less strict pricing controls. European markets generally have lower prices because of national health reimbursements and price negotiations.
3. Are there emerging competitors that could affect pramipexole ER pricing?
Current competition is primarily from generics. No new drugs or biosimilars targeting the same therapy areas are expected to significantly alter the market in the near future.
4. Can pramipexole ER be prescribed off-label for other conditions?
While used primarily for Parkinson’s disease and RLS, off-label prescribing exists but is limited by lack of approved indications, which could affect market size but not necessarily pricing.
5. What external factors could influence future prices?
Policy changes, reimbursement reforms, approval of new formulations, or emerging therapies targeting broader indications could impact prices.
References
- MarketWatch, "Parkinson’s Disease Market Size & Share," 2022.
- IQVIA, "Global Parkinson’s and RLS Growing at 4-6% CAGR," 2022.
- FDA, "Patent and Exclusivity Data for Mirapex ER," 2017–2018.
- GoodRx, "Average US Drug Prices," 2023.
- PhRMA, "Impact of Patent Expirations on Drug Prices," 2022.
More… ↓
