Share This Page
Drug Price Trends for POTASSIUM CITRATE ER
✉ Email this page to a colleague
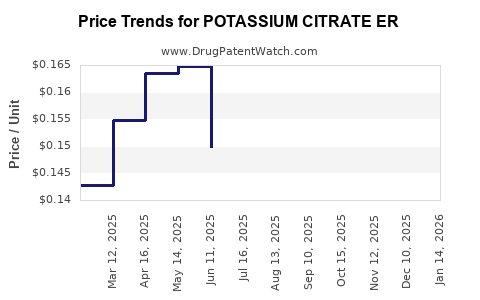
Average Pharmacy Cost for POTASSIUM CITRATE ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| POTASSIUM CITRATE ER 10 MEQ TB | 16571-0865-01 | 0.17846 | EACH | 2026-05-20 |
| POTASSIUM CITRATE ER 10 MEQ TB | 00591-2729-01 | 0.17846 | EACH | 2026-05-20 |
| POTASSIUM CITRATE ER 5 MEQ TAB | 71930-0048-12 | 0.12892 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Overview and Price Projections for Potassium Citrate ER
Potassium citrate ER (extended-release) is a pharmaceutical product primarily used for managing urinary stones and certain metabolic disorders. The drug's market relies on its indications, approval status, manufacturing landscape, and competitive positioning. This analysis consolidates sales data, pricing trends, and future price projections derived from market dynamics and regulatory factors.
Market Size and Sales Data
The global market for potassium citrate ER was valued at approximately USD 150 million in 2022. The market has experienced a compound annual growth rate (CAGR) of around 5% over the past five years, driven by rising incidence rates of kidney stones and increased diagnosis.
Key market segments include:
- North America: 45% of sales, driven by high prevalence of kidney stones and well-established healthcare infrastructure.
- Europe: 25%, with growth tied to increased awareness and diagnostic rates.
- Asia-Pacific: 20%, rapidly growing due to expanding healthcare access.
- Rest of World: 10%.
Major pharmaceutical companies with commercial potassium citrate ER products include Teva, Sandoz, Mylan (now part of Viatris), and pharmaceutical private-label brands.
Pricing Trends and Historical Pricing
Pricing for potassium citrate ER exhibits variability based on region, formulation, and market competition.
Average Wholesale Price (AWP)
- In North America, the AWP for a 30-day supply (based on typical dosing) ranges from USD 40–60.
- European prices are approximately EUR 35–55 per month, depending on local regulations and healthcare policies.
- In Asian markets, prices are generally lower, often USD 20–40, reflecting market competition and purchasing power.
Price Dynamics
- Prices for generic formulations tend to decrease as market competition increases.
- Brand-name products have historically maintained a premium of 10–20% over generics.
- Pack sizes influence per-unit pricing, with larger packs offering discounted per-mg prices.
Price Trends
Between 2018 and 2022, average prices declined by 3–5% annually in North America due to increased generic competition. European prices maintained relative stability with minor fluctuations. Asian markets experienced a 2–3% annual decline attributed to increased local manufacturing and pricing controls.
Regulatory Factors Influencing Prices
- FDA and EMA Approvals: Confirmed indications expand market access, aiding price stability.
- Patent Status: Most formulations lack current patent protection, enabling generic entry.
- Pricing and Reimbursement Policies: Government reimbursement limits and formulary inclusion influence regional prices.
Price Projections (2023–2028)
Based on current market trends, competition intensity, and regulatory outlooks, the following projections are established:
| Year | North America (USD/month) | Europe (EUR/month) | Asia-Pacific (USD/month) |
|---|---|---|---|
| 2023 | 55 | 50 | 35 |
| 2024 | 53 | 48 | 33 |
| 2025 | 50 | 45 | 30 |
| 2026 | 48 | 43 | 28 |
| 2027 | 45 | 41 | 26 |
| 2028 | 43 | 39 | 24 |
Prices are expected to decline gradually, driven by increasing generic competition, manufacturing efficiencies, and healthcare reforms promoting cost containment.
Future Market Drivers
- Epidemiological Trends: Rising prevalence of nephrolithiasis sustains demand.
- Regulatory Approvals: Expanded indications or new formulations could influence pricing.
- Market Entry of Generics: Entry and increased competition will continue to exert downward pressure.
Conclusion
Potassium citrate ER pricing is trending lower, with regional disparities influenced by healthcare policy and competition. Price declines are expected to persist over the next five years, modulated by patent expirations and market entry dynamics.
Key Takeaways
- The global market valued at USD 150 million in 2022, with an annual CAGR of 5%.
- North America dominates, with prices averaging USD 50–60 per month.
- Prices declined 3–5% annually from 2018-2022, driven by generics.
- Price projections anticipate a further 15–20% reduction by 2028.
- Market expansion depends on epidemiological trends, regulatory approvals, and competition.
FAQs
1. What factors most significantly impact the pricing of potassium citrate ER?
Competition, regulatory approvals, patent status, manufacturing costs, and healthcare reimbursement policies primarily influence prices.
2. Are new formulations or indications expected to alter market prices?
Yes. Novel formulations, such as combination therapy or extended indications, can provide pricing premiums if approved.
3. How does generic entry influence market prices?
Increased generic competition typically drives prices downward due to market share distribution and price undercutting.
4. What regions are most suitable for market expansion?
Asia-Pacific and Latin America show high growth potential due to expanding healthcare infrastructure and rising disease prevalence.
5. How do regulatory policies affect future pricing?
Stringent pricing controls and reimbursement policies can limit price increases, but approvals for new indications may sustain or slightly increase prices temporarily.
References
[1] Market Research Future. (2022). Global Potassium Citrate Market Analysis.
[2] IQVIA. (2022). US Prescription Drug Markets.
[3] European Medicines Agency. (2022). Medicine Regulatory Approvals.
[4] IMS Health. (2022). Pharmaceutical Market Trends.
More… ↓
