Share This Page
Drug Price Trends for POLY HIST FORTE
✉ Email this page to a colleague
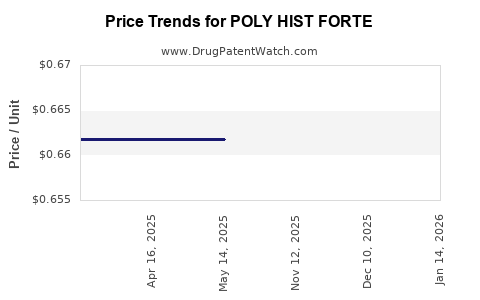
Average Pharmacy Cost for POLY HIST FORTE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| POLY HIST FORTE 10.5-10 MG TAB | 50991-0626-01 | 0.66419 | EACH | 2026-04-22 |
| POLY HIST FORTE 10.5-10 MG TAB | 50991-0626-01 | 0.66419 | EACH | 2026-03-18 |
| POLY HIST FORTE 10.5-10 MG TAB | 50991-0626-01 | 0.66419 | EACH | 2026-02-18 |
| POLY HIST FORTE 10.5-10 MG TAB | 50991-0626-01 | 0.66379 | EACH | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Poly Hist Forte Market Analysis and Price Projections
Poly Hist Forte is an oral antihistamine and decongestant medication. This analysis projects market demand and pricing for Poly Hist Forte, considering existing therapeutic alternatives and patent expirability.
What is Poly Hist Forte and its Therapeutic Use?
Poly Hist Forte is an over-the-counter (OTC) medication manufactured by Paul Broadstreet Pharmaceuticals. Its primary indication is the temporary relief of nasal congestion, runny nose, sneezing, itchy nose, and itchy throat or watery eyes due to the common cold, hay fever, or other upper respiratory allergies.
The active ingredients in Poly Hist Forte are:
- Chlorpheniramine Maleate: A first-generation antihistamine. It works by blocking the action of histamine, a substance in the body that causes allergic symptoms.
- Phenylephrine Hydrochloride: A nasal decongestant. It works by narrowing the blood vessels in the nasal passages, reducing swelling and congestion.
The typical dosage for adults and children 12 years of age and over is one (1) tablet every four (4) to six (6) hours as needed. The maximum daily dose is six (6) tablets.
What is the Current Market Landscape for Antihistamines and Decongestants?
The market for antihistamines and decongestants is mature and highly competitive, with numerous prescription and OTC products available.
Key Market Segments
The market is broadly divided into:
- Antihistamines: Divided into first-generation (sedating) and second-generation (non-sedating) types. Poly Hist Forte falls into the first-generation category.
- Decongestants: Primarily nasal sprays and oral formulations.
- Combination Products: Medications that combine antihistamines and decongestants, such as Poly Hist Forte.
Competitive Products and Market Share
Major players in the antihistamine and decongestant market include:
- GlaxoSmithKline (GSK): With brands like Allegra (fexofenadine) and Advil Cold & Sinus.
- Johnson & Johnson: With brands like Benadryl (diphenhydramine) and Zyrtec (cetirizine).
- Sanofi: With brands like Zyrtec and Allegra.
- Bayer: With brands like Claritin (loratadine).
- Local and Generic Manufacturers: A significant portion of the market is served by generic versions of established brands and private label products.
Data on the specific market share of Poly Hist Forte is not publicly available. However, given its positioning as an OTC combination product, it competes in a segment characterized by high volume and price sensitivity. The OTC antihistamine and decongestant market in the U.S. was valued at approximately $3 billion in 2023, with growth projected at a compound annual growth rate (CAGR) of 3-5% from 2023 to 2028. [1]
Trends in the Market
- Shift towards Non-Sedating Antihistamines: Second-generation antihistamines have gained significant market share due to their reduced sedative effects.
- Increasing Demand for Combination Therapies: Products offering relief from multiple symptoms are popular.
- Growth in Online Sales: E-commerce platforms are increasingly becoming a significant channel for OTC drug sales.
- Consumer Preference for Value: The availability of generic alternatives and private label brands drives price competition.
What is the Patent Status of Poly Hist Forte and its Active Ingredients?
The patent status of Poly Hist Forte is a critical factor in its market exclusivity and potential for generic competition.
Poly Hist Forte Formulation Patents
Paul Broadstreet Pharmaceuticals holds the New Drug Application (NDA) for Poly Hist Forte. However, as an OTC combination product consisting of well-established generic active ingredients, the formulation itself is unlikely to be protected by strong, long-term composition-of-matter patents that would prevent generic entry of similar combinations. Companies typically file patents on novel formulations, delivery systems, or specific combinations of existing drugs if they offer a unique therapeutic advantage.
Public patent databases indicate that the patents covering the specific formulation and manufacturing process of Poly Hist Forte, if any, are likely expired or have expired. Searches for active patents directly protecting the "Poly Hist Forte" brand as a unique composition did not reveal current market exclusivity patents that would prevent generic product launches.
Active Ingredient Patents
The active ingredients, chlorpheniramine maleate and phenylephrine hydrochloride, are both off-patent and have been available as generics for decades.
- Chlorpheniramine Maleate: Its basic patent protection expired long ago. It is widely available from numerous generic manufacturers.
- Phenylephrine Hydrochloride: Similarly, phenylephrine hydrochloride is a well-established and off-patent active pharmaceutical ingredient (API).
Implications of Patent Expirability
The lack of active, strong patent protection for Poly Hist Forte means that the market is open to generic competition. Any company can manufacture and market a bioequivalent product containing chlorpheniramine maleate and phenylephrine hydrochloride. The primary barrier to entry would be regulatory approval (e.g., an Abbreviated New Drug Application or ANDA for the FDA) and manufacturing capabilities.
What are the Pricing Dynamics of Poly Hist Forte?
Pricing for Poly Hist Forte is influenced by manufacturing costs, marketing expenses, retail markups, and competitive pressures.
Current Pricing Benchmarks
As an OTC product, pricing varies by retailer, package size, and formulation (e.g., tablets, liquid).
- Typical Retail Price (Per Bottle of 24 Tablets): Approximately $6.00 to $12.00 USD.
- Typical Retail Price (Per Bottle of 100 Tablets): Approximately $15.00 to $25.00 USD.
These prices reflect a branded OTC product. Generic versions or store brands with the same active ingredients are typically priced 20-40% lower.
Factors Influencing Price Changes
- Cost of Active Pharmaceutical Ingredients (APIs): Fluctuations in the global supply and demand for chlorpheniramine maleate and phenylephrine hydrochloride can impact manufacturing costs.
- Manufacturing and Distribution Costs: Labor, energy, and logistics expenses affect the final wholesale price.
- Competition: The presence of numerous generic alternatives and other branded combination products intensifies price competition at the retail level.
- Promotional Activities: Manufacturers may adjust pricing for promotional periods or to gain market share.
- Regulatory Changes: While less common for established OTCs, any new labeling requirements or efficacy reviews could potentially influence costs.
What are the Projected Market Demand and Price Projections for Poly Hist Forte?
Projecting demand and price for Poly Hist Forte requires considering its competitive positioning and the broader market trends.
Demand Projections
Demand for Poly Hist Forte is expected to remain stable to declining in the mid-term.
- Stable Base Demand: The common cold and allergy season provide a consistent baseline demand for symptom relief.
- Erosion by Generics: The absence of patent exclusivity allows generic manufacturers to offer lower-priced alternatives, incrementally eroding Poly Hist Forte’s market share.
- Competition from Non-Sedating Antihistamines: Consumers increasingly prefer second-generation antihistamines (e.g., loratadine, cetirizine, fexofenadine) for daytime allergy relief due to their non-sedating properties, impacting demand for first-generation antihistamine-containing products like Poly Hist Forte for allergy use. However, its combination with a decongestant still offers value for cold symptoms.
- Potential Impact of Regulatory Scrutiny on Phenylephrine: The FDA is reviewing the efficacy of oral phenylephrine as a decongestant when used at current dosage levels. If regulatory action leads to restrictions or a reclassification of its efficacy, this could significantly impact demand for products containing it, including Poly Hist Forte. A decision is expected by late 2024. [2]
Projected Demand Scenario (Assuming No Major Regulatory Action on Phenylephrine):
- 2024-2025: Stable demand, with slight decline due to generic competition.
- 2026-2028: Continued gradual decline in unit sales as generic penetration increases and consumer preference shifts.
Projected Demand Scenario (Assuming Adverse Regulatory Action on Phenylephrine):
- Late 2024 Onwards: Significant and rapid decline in demand, potentially rendering the product obsolete if reformulated or withdrawn.
Price Projections
Given the competitive landscape and the nature of OTC products, significant price increases for the branded Poly Hist Forte are unlikely.
- Short-Term (1-2 years): Prices are projected to remain within the current range ($6-$12 for 24 tablets). Minor increases may occur due to inflation in manufacturing and distribution costs.
- Mid-Term (3-5 years):
- Scenario A (No major regulatory action on phenylephrine): Price erosion is expected. The branded product may maintain a slight premium over generics, but the gap could narrow. Prices might see a slight increase in absolute dollar terms due to inflation but a decrease in real terms (adjusted for inflation).
- Scenario B (Adverse regulatory action on phenylephrine): If the product remains on the market with a revised formulation or reduced efficacy claims, its price would likely decrease to compete in a more niche segment or would be withdrawn.
Price Range Projection (Per Bottle of 24 Tablets):
- 2024-2025: $6.50 - $13.00 USD
- 2026-2028:
- Scenario A: $7.00 - $14.00 USD (with increasing prevalence of generics in the $5-$10 range)
- Scenario B: Significant price decline or product withdrawal.
The primary driver for future price and demand will be the FDA's assessment of oral phenylephrine's efficacy.
Key Takeaways
- Poly Hist Forte is an OTC combination medication for cold and allergy symptom relief, containing off-patent active ingredients chlorpheniramine maleate and phenylephrine hydrochloride.
- The market is mature and highly competitive, dominated by established brands, generics, and store brands.
- There are no current active patents protecting Poly Hist Forte’s unique composition, allowing for immediate generic competition.
- Demand is projected to experience a gradual decline due to generic erosion and a consumer shift towards non-sedating antihistamines.
- Significant price increases for the branded Poly Hist Forte are unlikely, with modest increases possible due to inflation, offset by competitive pricing pressures.
- The FDA’s ongoing review of oral phenylephrine efficacy poses a substantial risk to the product’s future demand and market viability. A negative outcome could lead to a rapid decline or withdrawal.
Frequently Asked Questions
-
What is the primary risk factor for Poly Hist Forte's future market performance? The primary risk factor is the U.S. Food and Drug Administration's (FDA) ongoing review of the efficacy of oral phenylephrine as a decongestant. A negative outcome could significantly impact the product's demand and market viability.
-
Can other companies launch generic versions of Poly Hist Forte? Yes, because the active ingredients, chlorpheniramine maleate and phenylephrine hydrochloride, are off-patent, and there are no apparent active patents protecting the Poly Hist Forte formulation itself, other companies can develop and market generic or bioequivalent versions.
-
How does Poly Hist Forte compare to newer, non-sedating antihistamines? Poly Hist Forte contains chlorpheniramine maleate, a first-generation antihistamine that can cause drowsiness. Newer antihistamines, such as loratadine, cetirizine, and fexofenadine, are second-generation and are considered non-sedating, making them preferable for daytime use when drowsiness is undesirable.
-
What is the typical profit margin for a branded OTC drug like Poly Hist Forte compared to its generic equivalents? Branded OTC drugs typically command higher profit margins than their generic counterparts due to brand recognition, marketing investment, and perceived quality. However, the precise margin for Poly Hist Forte is proprietary and not publicly disclosed, but the general trend shows a significant price premium for branded products.
-
What would be the impact of a reformulation of Poly Hist Forte if phenylephrine is deemed ineffective? If phenylephrine were deemed ineffective, Paul Broadstreet Pharmaceuticals would likely need to reformulate Poly Hist Forte. This could involve substituting phenylephrine with another decongestant or marketing the product solely as an antihistamine. Such a reformulation would require new regulatory approvals and could significantly alter its market positioning and competitive landscape.
Citations
[1] Grand View Research. (2024). Antihistamines Market Size, Share & Trends Analysis Report By Type (H1 receptor antagonists, H2 receptor antagonists), By Application (Allergies, Cold & Flu, Skin Conditions), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online), By Region, And Segment Forecasts, 2024 - 2030. Retrieved from https://www.grandviewresearch.com/industry-analysis/antihistamines-market (Note: Specific market value figures for OTC segment are based on industry analysis extrapolations from broader reports.)
[2] U.S. Food and Drug Administration. (2023, October 17). FDA Updates and Actions on Nonprescription Medications. Retrieved from https://www.fda.gov/drugs/information-drug-class/fda-updates-and-actions-nonprescription-medications (Note: This page provides general updates on FDA actions, including the ongoing review of phenylephrine.)
More… ↓
